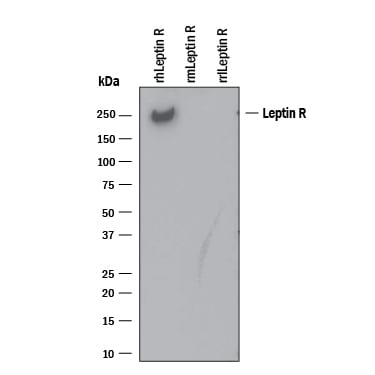
Leptin receptor (OB-R), also named B219, is a type I cytokine receptor family protein with significant amino acid sequence identity with gp130, G-CSF receptor, and the LIF receptor. Multiple isoforms of human and mouse OB-R, including a long form (OB-RL) with a large cytoplasmic domain capable of signal-transduction, and several receptor isoforms with short cytoplasmic domains (OB-Rs) lacking signal-transducing capabilities, have been identified. The extracellular domains of the short and long forms of OB-R are identical. An OB-R transcript, lacking a transmembrane domain and potentially encoding a soluble form of the receptor, has also been described. OB-RL transcripts were reported to be expressed predominantly in regions of the hypothalamus previously thought to be important in body weight regulation. Expression of OB-Rs transcripts have been found in multiple tissues, including the choroid plexus, lung, kidney, and primitive hematopoietic cell populations. OB-R has been shown to be encoded by the mouse diabetes (db) and rat fatty (fa) genes. Rodents homozygous for the db or fa mutations have been known to exhibit an obesity phenotype.
Human OB-R long form encodes a 1165 amino acid (aa) residue precursor protein with a 22 aa residue signal peptide, an 819 aa residue extracellular domain, a 21 aa residue transmembrane domain, and a 303 aa residue cytoplasmic domain. The extracellular domain of OB-R contain two hemopoietin receptor domains, a fibronectin type III domain and the WSXWS domain. Recombinant soluble OB-R has been shown to bind Leptin with high affinity and is a potent Leptin antagonist.