Matrix metalloproteinases are a family of zinc and calcium dependent endopeptidases with the combined ability to degrade all the components of the extracellular matrix. MMP-1 (interstitial collagenase), can degrade a broad range of substrates including types I, II, III, VII, VIII, and X collagens as well as casein, gelatin,
alpha ‑1 antitrypsin, myelin basic protein, L-Selectin, pro-TNF, IL-1 beta, IGF-BP3, IGF-BP5, pro MMP-2 and pro MMP-9. A significant role of MMP-1 is the degradation of fibrillar collagens in extracellular matrix remodeling, characterized by the cleavage of the interstitial collagen triple helix into ¾, ¼ fragments. However, as the list of substrates above illustrates, the role of MMP-1 is more diverse than originally envisaged, and may involve enzyme cascades, cytokine regulation and cell surface molecule modulation. MMP-1 is expressed by fibroblasts, keratinocytes, endothelial cells, monocytes and macrophages. Structurally, MMP-1 may be divided into several distinct domains; a pro-domain which is cleaved upon activation; a catalytic domain containing the zinc binding site; a short hinge region and a carboxyl terminal (hemopexin-like) domain.


Key Product Details
Validated by
Knockout/Knockdown, Biological Validation
Species Reactivity
Validated:
Human
Cited:
Human, Mouse, Rat, Rabbit
Applications
Validated:
Knockout Validated, Immunohistochemistry, Western Blot, Neutralization, Immunoprecipitation
Cited:
Immunohistochemistry, Immunohistochemistry-Paraffin, Western Blot, Neutralization, Immunocytochemistry, Dot Blot, ELISA Development
Label
Unconjugated
Antibody Source
Monoclonal Mouse IgG1 Clone # 36665
Loading...
Product Specifications
Immunogen
Chinese hamster ovary cell line CHO-derived recombinant human MMP-1
Phe20-Asn469
Accession # P03956
Phe20-Asn469
Accession # P03956
Specificity
Detects pro and active forms of human MMP-1. In Western blots, no cross-reactivity with recombinant human (rh) MMP‑2, rhMMP‑3, or rhMMP‑9 is observed.
Clonality
Monoclonal
Host
Mouse
Isotype
IgG1
Scientific Data Images for Human MMP‑1 Antibody
Detection of Human MMP‑1 by Western Blot.
Western blot shows lysates of PC-3 human prostate cancer cell line. PVDF Membrane was probed with 2 µg/mL of Mouse Anti-Human MMP-1 Monoclonal Antibody (Catalog # MAB901) followed by HRP-conjugated Anti-Mouse IgG Secondary Antibody (Catalog # HAF007). A specific band was detected for MMP-1 at approximately 54 kDa (as indicated). This experiment was conducted under reducing conditions and using Immunoblot Buffer Group 1.MMP‑1 in Human Ovarian Cancer Tissue.
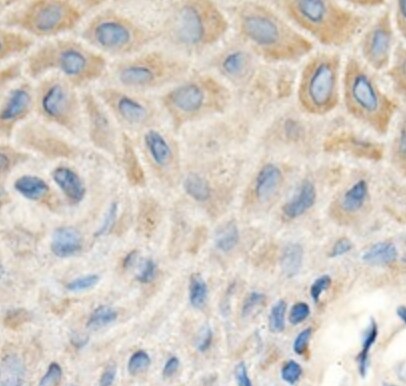
MMP‑1 was detected in immersion fixed paraffin-embedded sections of human ovarian cancer tissue using 25 µg/mL Mouse Anti-Human MMP‑1 Monoclonal Antibody (Catalog # MAB901) overnight at 4 °C. Tissue was stained with the Anti-Mouse HRP-AEC Cell & Tissue Staining Kit (red; Catalog # CTS003) and counterstained with hematoxylin (blue). View our protocol for Chromogenic IHC Staining of Paraffin-embedded Tissue Sections.MMP‑1 in Human Ovarian Array.
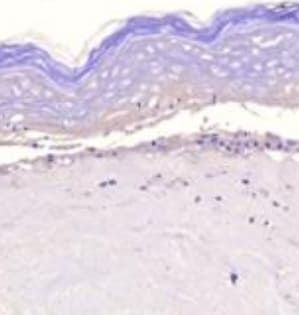
MMP-1 was detected in immersion fixed paraffin-embedded sections of human ovarian array using Mouse Anti-Human MMP-1 Monoclonal Antibody (Catalog # MAB901) at 25 µg/mL overnight at 4 °C. Tissue was stained using the Anti-Mouse HRP-DAB Cell & Tissue Staining Kit (brown; Catalog # CTS002) and counterstained with hematoxylin (blue). Lower panel shows a lack of labeling if primary antibodies are omitted and tissue is stained only with secondary antibody followed by incubation with detection reagents. View our protocol for Chromogenic IHC Staining of Paraffin-embedded Tissue Sections.Western Blot Shows Human MMP‑1 Specificity by Using Knockout Cell Line.
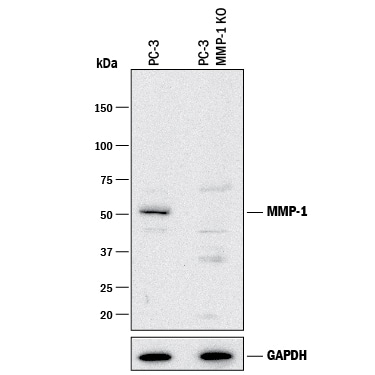
Western blot shows lysates of PC-3 human prostate cancer parental cell line and MMP-1 knockout PC-3 cell line (KO). PVDF membrane was probed with 2 µg/mL of Mouse Anti-Human MMP-1 Monoclonal Antibody (Catalog # MAB901) followed by HRP-conjugated Anti-Mouse IgG Secondary Antibody (Catalog # HAF018). A specific band was detected for MMP-1 at approximately 50 kDa (as indicated) in the parental PC-3 cell line, but is not detectable in knockout PC-3 cell line. GAPDH (Catalog # AF5718) is shown as a loading control. This experiment was conducted under reducing conditions and using Immunoblot Buffer Group 1.Detection of Human MMP-1 by Western Blot
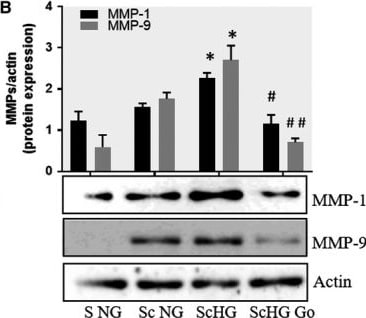
Inhibition of PKC alpha decreases the protein expression of MMP‐1 and MMP‐9 induced by cell cross‐talk in SMC. A, Evaluation of PKC alpha activation in control SMC (S NG) vs SMC previously co‐cultured with MAC in HG (Sc HG). Note that, the phosphorylated form of PKC alpha is significantly increased in SMC that were before exposed to MAC in HG conditions. Silencing of CCR2 or p65 decreases the phosphorylation of PKC alpha to control levels. B, Effect of PKC alpha inhibitor – Go 6976 on the MMP‐1 and MMP‐9 protein expression induced by SMC‐MAC cross‐talk. S NG – control SMC, Sc NG – SMC which were co‐cultured with MAC in normal glucose, Sc HG ‐ SMC which were co‐cultured with MAC in high glucose, Sc HGGo ‐ SMC which were co‐cultured with MAC in high glucose in presence of 1 μmol/L Go 6976. n = 3, *P < .05, **P <.01, #P <.05, ##P <.01 Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/29992758), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human MMP-1 by Western Blot
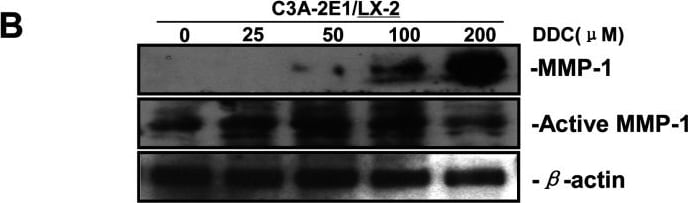
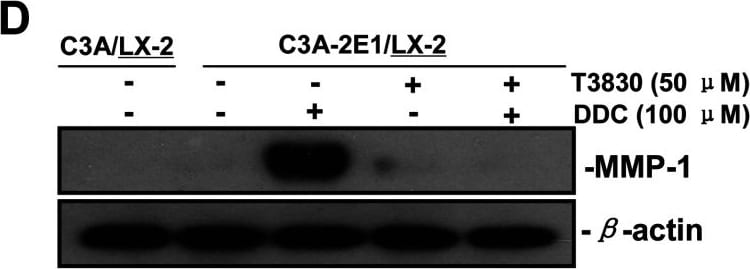
DDC up-regulates MMP-1 through ERK1/2 and Akt activation(A) Co-cultures were treated with 100 μM DDC for the indicated time. Phosphorylation of ERK1/2, p38 and Akt were determined by Western blotting analysis. The corresponding non-phosphorylated ERK1/2, p38, Akt and beta -actin were used for protein loading control. (B) Co-cultures were treated with or without 100 μM DDC for 24 h in the presence or absence of ERK1/2 inhibitor (U0126, 10 μM). MMP-1 protein levels were analysed by Western blotting. (C) Co-cultures were treated with or without 100 μM DDC for 24 h in the presence or absence of p38 inhibitor (SB203580, 10 μM). MMP-1 protein levels were analysed by Western blotting. (D) Co-cultures were treated with or without 100 μM DDC for 24 h in the presence or absence of Akt inhibitor (T3830, 50 μM). MMP-1 protein levels were analysed by Western blotting. Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/23577625), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human MMP-1 by Western Blot
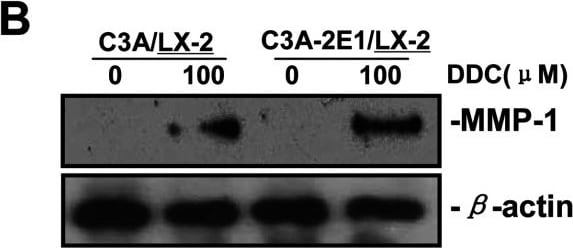
DDC up-regulates MMP-1 expression in a dose-dependent manner and lowered collagen I in LX-2 cellsCo-culture systems were treated with 25, 50, 100 and 200 μM DDC for 24 h. LX-2 cells and the culture medium were collected and analysed. (A) Quantitative RT-PCR results of MMP-1 expression in LX-2 cells. The results were normalized with beta -actin and expressed as fold increase relative to the control in each experiment. (B) 20 μg aliquots of total protein extracts from LX-2 cells were subjected to Western blotting analysis with anti-MMP-1 antibody (MAB 901 and MAB 3223 for active form only) and beta -actin antibody as a loading control, as described in the Materials and Methods section. (C) 30 μg of LX-2 lysates or 20 μl aliquots of 10-fold concentrated culture medium with 5×loading buffer were assayed by Casein Zymography for MMP-1 activity. (D) Levels of collagen I in LX-2 cells were assayed by western blotting. (E) Levels of collagen I in the culture medium were assayed by ELISA. *P<0.05 compared with untreated C3A-2E1/LX-2. Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/23577625), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human MMP-1 by Western Blot
DDC up-regulates MMP-1 through ERK1/2 and Akt activation(A) Co-cultures were treated with 100 μM DDC for the indicated time. Phosphorylation of ERK1/2, p38 and Akt were determined by Western blotting analysis. The corresponding non-phosphorylated ERK1/2, p38, Akt and beta -actin were used for protein loading control. (B) Co-cultures were treated with or without 100 μM DDC for 24 h in the presence or absence of ERK1/2 inhibitor (U0126, 10 μM). MMP-1 protein levels were analysed by Western blotting. (C) Co-cultures were treated with or without 100 μM DDC for 24 h in the presence or absence of p38 inhibitor (SB203580, 10 μM). MMP-1 protein levels were analysed by Western blotting. (D) Co-cultures were treated with or without 100 μM DDC for 24 h in the presence or absence of Akt inhibitor (T3830, 50 μM). MMP-1 protein levels were analysed by Western blotting. Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/23577625), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human MMP-1 by Western Blot
DDC up-regulates MMP-1 expression in a dose-dependent manner and lowered collagen I in LX-2 cellsCo-culture systems were treated with 25, 50, 100 and 200 μM DDC for 24 h. LX-2 cells and the culture medium were collected and analysed. (A) Quantitative RT-PCR results of MMP-1 expression in LX-2 cells. The results were normalized with beta -actin and expressed as fold increase relative to the control in each experiment. (B) 20 μg aliquots of total protein extracts from LX-2 cells were subjected to Western blotting analysis with anti-MMP-1 antibody (MAB 901 and MAB 3223 for active form only) and beta -actin antibody as a loading control, as described in the Materials and Methods section. (C) 30 μg of LX-2 lysates or 20 μl aliquots of 10-fold concentrated culture medium with 5×loading buffer were assayed by Casein Zymography for MMP-1 activity. (D) Levels of collagen I in LX-2 cells were assayed by western blotting. (E) Levels of collagen I in the culture medium were assayed by ELISA. *P<0.05 compared with untreated C3A-2E1/LX-2. Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/23577625), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human MMP-1 by Western Blot
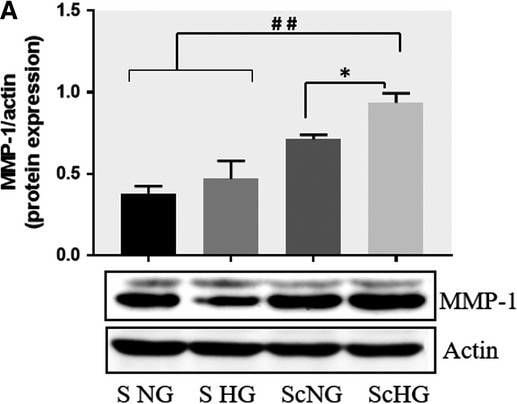
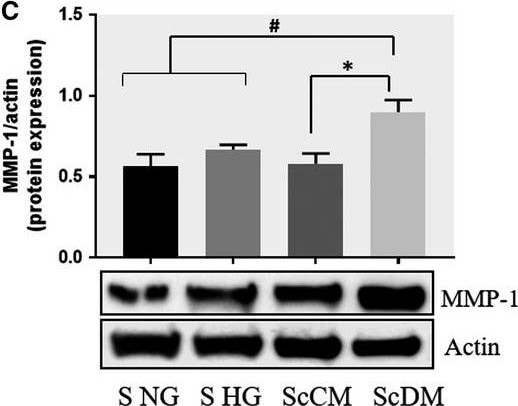
Co‐culture of SMC and MAC in high glucose conditions (24 h) induces a significant increase in the protein expression of MMP‐1 and MMP‐9. A, B: Protein expression of MMP‐1 (A) and MMP‐9 (B) was assessed in SMC after co‐culture with THP‐derived macrophages and evaluated by Western blot assay. C, D: Protein expression of MMP‐1 (C) and MMP‐9 (D) in SMC after co‐culture with human macrophages derived from freshly isolated monocytes from control subjects (CM) or diabetic patients with ACS (DM). S NG –control SMC, S HG – SMC exposed to HG, Sc NG or Sc CM – SMC which were co‐cultured with MAC in normal glucose, Sc HG or Sc DM ‐ SMC which were co‐cultured with MAC in high glucose. n = 3, *P < .05, **P < .01, ***P < .001, #P <.05, ##P <.01, ###P <.001 Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/29992758), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human MMP-1 by Western Blot
Co‐culture of SMC and MAC in high glucose conditions (24 h) induces a significant increase in the protein expression of MMP‐1 and MMP‐9. A, B: Protein expression of MMP‐1 (A) and MMP‐9 (B) was assessed in SMC after co‐culture with THP‐derived macrophages and evaluated by Western blot assay. C, D: Protein expression of MMP‐1 (C) and MMP‐9 (D) in SMC after co‐culture with human macrophages derived from freshly isolated monocytes from control subjects (CM) or diabetic patients with ACS (DM). S NG –control SMC, S HG – SMC exposed to HG, Sc NG or Sc CM – SMC which were co‐cultured with MAC in normal glucose, Sc HG or Sc DM ‐ SMC which were co‐cultured with MAC in high glucose. n = 3, *P < .05, **P < .01, ***P < .001, #P <.05, ##P <.01, ###P <.001 Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/29992758), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human MMP-1 by Western Blot
DDC up-regulates MMP-1 through ERK1/2 and Akt activation(A) Co-cultures were treated with 100 μM DDC for the indicated time. Phosphorylation of ERK1/2, p38 and Akt were determined by Western blotting analysis. The corresponding non-phosphorylated ERK1/2, p38, Akt and beta -actin were used for protein loading control. (B) Co-cultures were treated with or without 100 μM DDC for 24 h in the presence or absence of ERK1/2 inhibitor (U0126, 10 μM). MMP-1 protein levels were analysed by Western blotting. (C) Co-cultures were treated with or without 100 μM DDC for 24 h in the presence or absence of p38 inhibitor (SB203580, 10 μM). MMP-1 protein levels were analysed by Western blotting. (D) Co-cultures were treated with or without 100 μM DDC for 24 h in the presence or absence of Akt inhibitor (T3830, 50 μM). MMP-1 protein levels were analysed by Western blotting. Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/23577625), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human MMP-1 by Western Blot
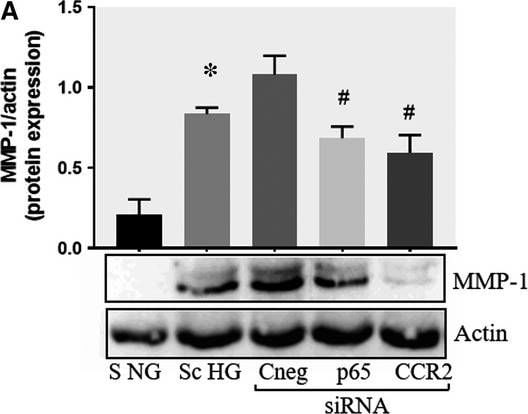
Effect of CCR2 and p65 silencing in the regulation of MMP‐1 and MMP‐9 expression in SMC. Protein expression of MMP‐1 (A) and MMP‐9 (B) determined by Western blot in control SMC (S NG), SMC which were co‐cultured with MAC in HG conditions (Sc HG), and SMC subjected to silencing of CCR‐2 and p65 and co‐cultured with MAC in HG conditions (Cneg, p65, CCR2 siRNA). Note that silencing of CCR2 or p65 decreases significantly the protein expression of MMP‐1 and MMP‐9 induced by SMC‐MAC co‐culture in high glucose conditions. n = 3, *P < .05, #P < .05 Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/29992758), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human MMP-1 by Western Blot
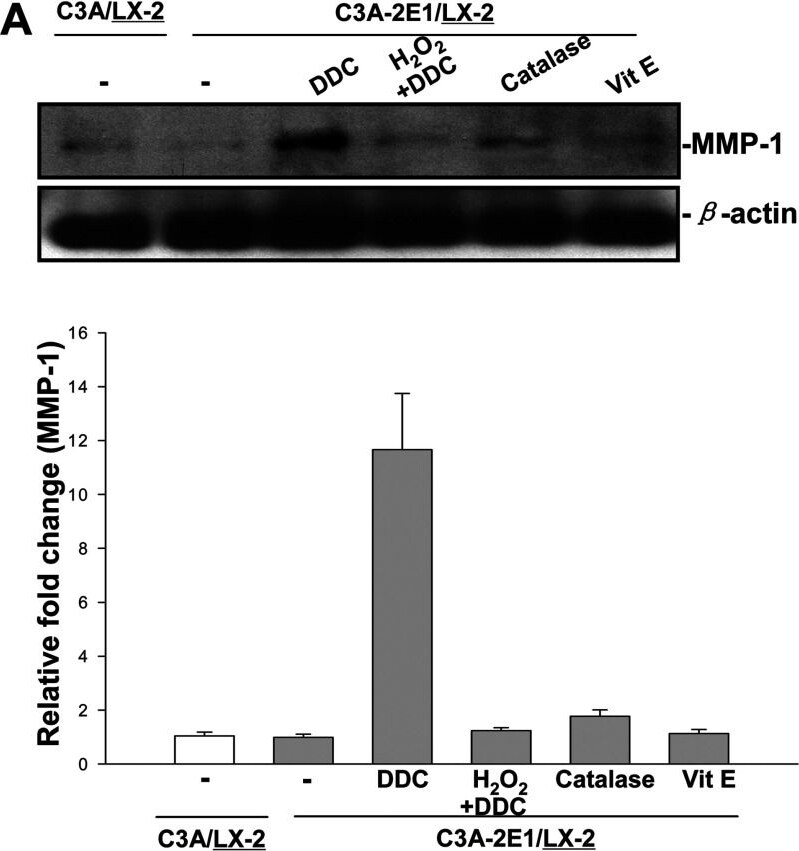
DDC up-regulates MMP-1 expression in an H2O2-associated mannerCo-cultures were treated with 100 μM DDC, 2000 Units catalase, 25 μM vitamin E and 5μM H2O2 combined with 100 μM DDC for 24 h. Cell lysates from the LX-2 cells were collected and analysed. (A) 20 μg aliquots of total protein extracts from LX-2 cells were subjected to Western blotting analysis with anti-MMP-1 antibody (MAB 901) and beta -actin antibody as a loading control, as described in the Materials and Methods section. (B) Levels of collagen I in LX-2 cells were assayed by Western blotting. Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/23577625), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human MMP-1 by Western Blot
DDC up-regulates MMP-1 expression in LX-2 cellsCo-culture systems were treated with 100 μM DDC for 24 h. LX-2 cells and the culture medium were collected and analysed. (A) Quantitative RT-PCR results of MMP-1 expression in LX-2 cells. The results were normalized with beta -actin and expressed as fold increase relative to the control in each experiment. (B) 20 μg aliquots of total protein extracts from LX-2 cells were subjected to western blotting analysis with anti-MMP-1 antibody (MAB 901) and beta -actin antibody as a loading control, as described in the Materials and Methods section. Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/23577625), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of MMP-1 by Western Blot
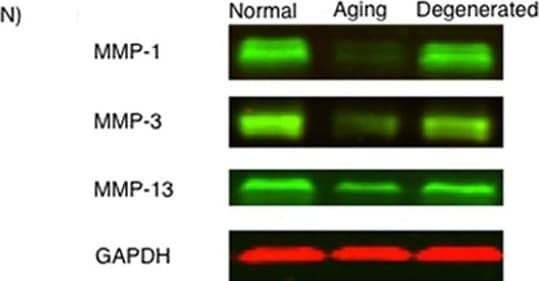
MMP-1, MMP-3, and MMP-13 expression. (A-D) MMP-1; (E-H) MMP-3; (I-L) MMP-13 (A,E,I) ACL from young normal knee; (B,F,J) ACL from aging knee; (C,G,K) fibroblast-like cell aggregates in the degenerated ACL; (D,H,L) chondrocyte-like cell aggregates in the degenerated ACL. (Original magnification ×40). The graph (M) represents the percentage (mean ± SEM) of MMP-1-, MMP-3-, and MMP-13-positive cells in each group. **P < 0.01. (N) Western blotting for MMP-1, MMP-3, and MMP-13 (n = 3 for each category). Image collected and cropped by CiteAb from the following open publication (https://arthritis-research.biomedcentral.com/articles/10.1186/ar4165), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of MMP-1 by Immunohistochemistry
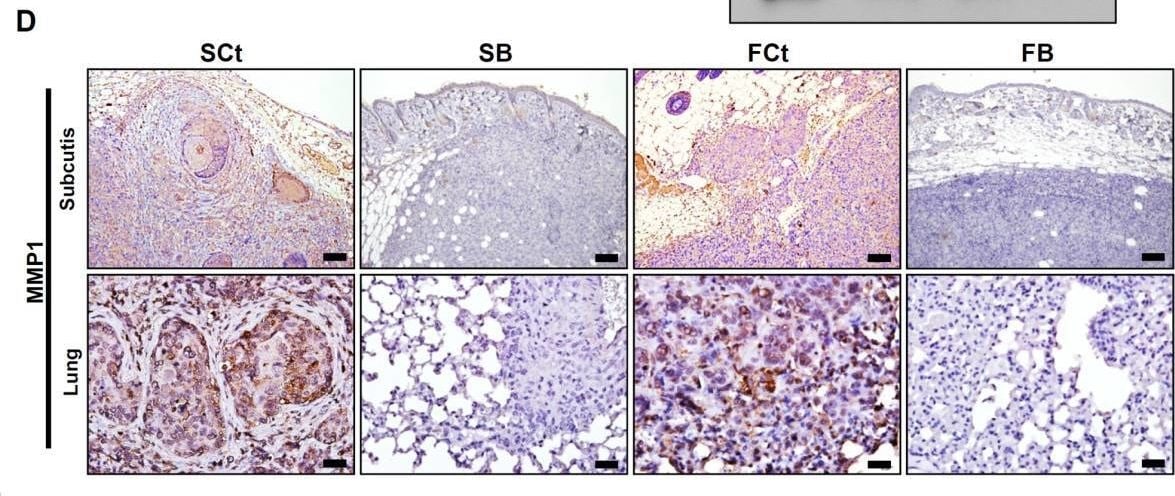
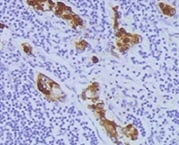
AEG-1 knockdown down-regulated MMP1 expression in HNSCC cells in vitro and in vivo. (A) microarray analysis of gene expression after AEG-1 knockdown in SAS cells. Genes with an absolute fold change greater than 2.5 are shown. SNORD3B-1, Homo sapiens small nucleolar RNA, C/D box 3B-1; EMILIN2, elastin microfibril interfacer 2; FABP4, fatty acid binding protein 4; MMP1, matrix metallopeptidase 1; ANO1, anoctamin 1; RELN, reelin; FPR2, formyl peptide receptor 2; UHMK1, U2AF homology motif kinase 1; RMND5A, required for meiotic nuclear division 5 homolog A; CAPZA1; capping protein (actin filament) muscle Z-line, alpha 1. *, p < 0.05; **, p < 0.01; ***, p < 0.001. (B) RT-QPCR confirmation of gene expression profiles after knockdown of AEG-1 in SAS and FaDu cells. All experiments were performed in triplicate (n = 3) and data were normalized to GAPDH. (C) secreted MMP1 protein in cell-conditioned culture media. Each cell lines were seeded in equal numbers (1 × 106 cells) and were cultured in starving condition (1% fetal bovine serum, FBS) for 24 hr and the media were harvested. (D) immunohistochemical staining of MMP1 in murine subcutaneous xenograft tumors (upper row) and metastatic lesions from in vivo lung metastasis assays (lower row). Scale bar: upper row, 100 μm; lower row, 25 μm. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/24063540), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of MMP-1 by Western Blot
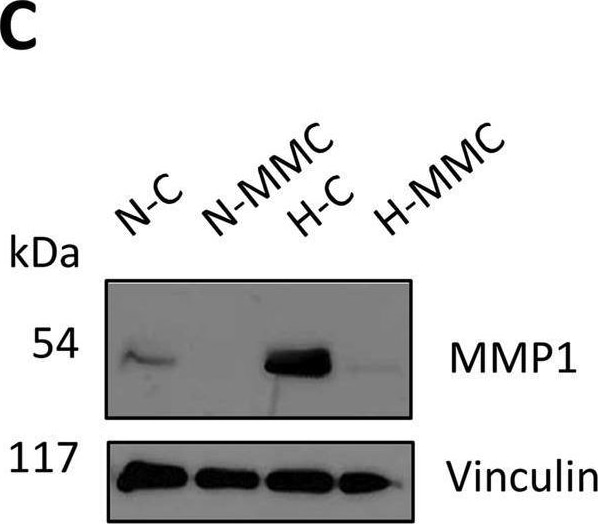
Expression of MMP1 on gene and protein levels upon treatment with mitomycin C (MMC) of SKOV3 cells in normoxia and hypoxia. MMP1 gene expression was measured using (A) HTA 2.0 microarrays and (B) RT-qPCR [normalized to PPIA and GUSB levels and normoxia control (NC) as 1]. (C) Immunoblot of MMP 1 protein (with vinculin as loading control). (D) Quantification of MMP1 protein levels by densitometry. (E) Secretion of pro-MMP1 protein levels measured by ELISA.(F) Secretion of total MMP1 protein levels measured by ELISA. (B,D) Results were normalized to normoxia control as 1. ****p < 0.0001, ***p < 0.001, **p < 0.01, and *p < 0.05; VIN, vinculin. A detailed statistics description is provided in the Supplementary Table S2. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/40584967), licensed under a CC-BY license. Not internally tested by R&D Systems.Applications for Human MMP‑1 Antibody
Application
Recommended Usage
Immunohistochemistry
8-25 µg/mL
Sample: Immersion fixed paraffin-embedded sections of human ovarian cancer tissue, and immersion fixed paraffin-embedded sections of human ovarian array
Sample: Immersion fixed paraffin-embedded sections of human ovarian cancer tissue, and immersion fixed paraffin-embedded sections of human ovarian array
Immunoprecipitation
25 µg/mL
Sample: Conditioned cell culture medium spiked with Recombinant Human MMP‑1 (Catalog # 901‑MP), see our available Western blot detection antibodies
Sample: Conditioned cell culture medium spiked with Recombinant Human MMP‑1 (Catalog # 901‑MP), see our available Western blot detection antibodies
Knockout Validated
MMP-1 is specifically detected in PC‑3 human prostate cancer parental cell line but is not detectable in
MMP-1 knockout PC‑3 cell line.
Western Blot
2 µg/mL
Sample: PC‑3 human prostate cancer cell line
Sample: PC‑3 human prostate cancer cell line
Neutralization
Measured by its ability to neutralize Recombinant Human MMP‑1 (10 µg/mL, Catalog # 901-MP) cleavage of Cultrex Rat Collagen I (250 µg/mL, Catalog # 3440-100-01). The Neutralization Dose (ND50) is typically 200 µg/mL.
Reviewed Applications
Read 5 reviews rated 4.6 using MAB901 in the following applications:
Formulation, Preparation, and Storage
Purification
Protein A or G purified from ascites
Reconstitution
Reconstitute at 0.5 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: MMP-1
References
- Cawston, T.E. (2004) in Interstitial Collagenase. Barrett, A.J. et al. (eds): Handbook of Proteolytic Enzymes, San Diego: Academic Press, p. 472.
Long Name
Matrix Metalloproteinase 1
Alternate Names
MMP1
Gene Symbol
MMP1
UniProt
Additional MMP-1 Products
Product Documents for Human MMP‑1 Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human MMP‑1 Antibody
For research use only
Related Research Areas
Citations for Human MMP‑1 Antibody
Customer Reviews for Human MMP‑1 Antibody (5)
4.6 out of 5
5 Customer Ratings
Have you used Human MMP‑1 Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
Showing
1
-
5 of
5 reviews
Showing All
Filter By:
-
Application: ImmunohistochemistrySample Tested: chronic periodontitis gingival biopsySpecies: HumanVerified Customer | Posted 06/16/2022
-
Application: ImmunohistochemistrySample Tested: Skin tissueSpecies: HumanVerified Customer | Posted 01/24/2022
-
Application: ImmunohistochemistrySample Tested: Gastric cancerSpecies: HumanVerified Customer | Posted 08/13/2021
-
Application: Western BlotSample Tested: HeLa cellsSpecies: HumanVerified Customer | Posted 07/13/2018
-
Application: Western BlotSample Tested: PC-3 human prostate cancer cell lineSpecies: HumanVerified Customer | Posted 05/10/2017
There are no reviews that match your criteria.
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- IHC Sample Preparation (Frozen sections vs Paraffin)
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Immunoprecipitation Protocol
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Immunohistochemistry
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...