Human/Mouse Brachyury Antibody
R&D Systems | Catalog # AF2085

Key Product Details
Species Reactivity
Validated:
Cited:
Applications
Validated:
Cited:
Label
Antibody Source
Product Specifications
Immunogen
Ser2-Glu202
Accession # O15178
Specificity
Clonality
Host
Isotype
Scientific Data Images for Human/Mouse Brachyury Antibody
Detection of Recombinant Human Brachyury by Western Blot.
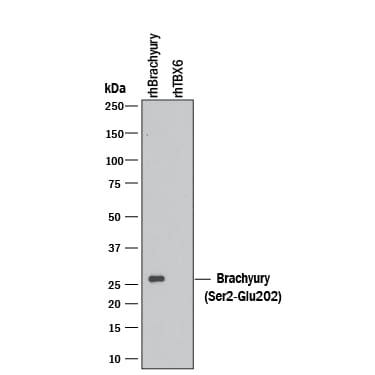
Western blot shows 10 ng of Recombinant Human Brachyury (amino acids Ser2-Glu202) and Recombinant Human TBX6. PVDF Membrane was probed with 0.1 µg/mL of Goat Anti-Human/Mouse Brachyury Antigen Affinity-purified Polyclonal Antibody (Catalog # AF2085) followed by HRP-conjugated Anti-Goat IgG Secondary Antibody (HAF109). A specific band was detected for Brachyury at approximately 26 kDa (as indicated). This experiment was conducted under reducing conditions and using Immunoblot Buffer Group 3.Detection of Human Brachyury by Western Blot.
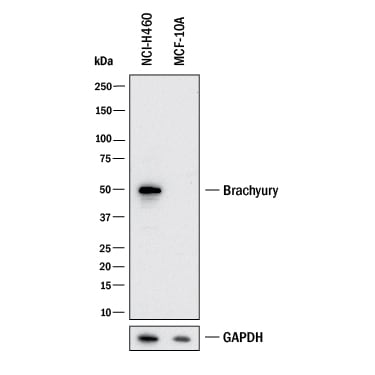
Western blot shows lysates of NCI‑H460 human large cell lung carcinoma cell line and MCF 10A human breast epithelial cell line (negative control). PVDF membrane was probed with 1 µg/mL of Goat Anti-Human/Mouse Brachyury Antigen Affinity-purified Polyclonal Antibody (Catalog # AF2085) followed by HRP-conjugated Anti-Goat IgG Secondary Antibody (HAF017). A specific band was detected for Brachyury at approximately 50 kDa (as indicated). GAPDH (AF5718) is shown as a loading control. This experiment was conducted under reducing conditions and using Western Blot Buffer Group 1.Brachyury in Differentiated Human Embryonic Stem Cells.
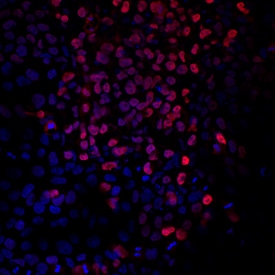
Brachyury was detected in immersion fixed differentiated human embryonic stem cells using 10 µg/mL Goat Anti-Human Brachyury Antigen Affinity-purified Polyclonal Antibody (Catalog # AF2085) for 3 hours at room temperature. Cells were stained (green) and counter-stained with DAPI (blue). View our protocol for Fluorescent ICC Staining of Cells on Coverslips.Brachyury in BG01V Human Stem Cells.
Brachyury was detected in immersion fixed BG01V human embryonic stem cells differentiated into mesoderm using Goat Anti-Human Brachyury Antigen Affinity-purified Polyclonal Antibody (Catalog # AF2085) at 10 µg/mL for 3 hours at room temperature. Cells were stained using the Northern-Lights™ 557-conjugated Anti-Goat IgG Secondary Antibody (red; NL001) and counter-stained with DAPI (blue). Specific staining was localized to nuclei. View our protocol for Fluorescent ICC Staining of Cells on Coverslips.Brachyury in Embryonic Mouse Notochord.
Brachyury was detected in immersion fixed frozen sections of embryonic mouse notochord (E9.5) using 10 µg/mL Goat Anti-Human Brachyury Antigen Affinity-purified Polyclonal Antibody (Catalog # AF2085) overnight at 4 °C. Tissue was stained with the Northern-Lights™ 557-conjugated Anti-Goat IgG Secondary Antibody (red; NL001) and counter-stained with DAPI (blue). View our protocol for Fluorescent IHC Staining of Frozen Tissue Sections.Detection of Brachyury-regulated Genes by Chromatin Immunoprecipitation.
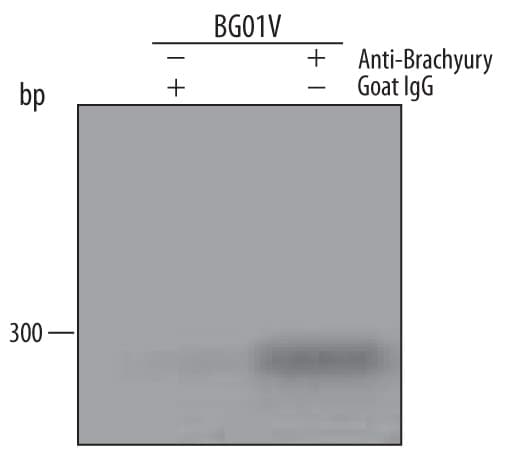
Mesoderm-differentiated BG01V human embryonic stem cells were fixed using formaldehyde, resuspended in lysis buffer, and sonicated to shear chromatin. Brachyury/DNA complexes were immunoprecipitated using 5 µg Goat Anti-Human/Mouse Brachyury Antigen Affinity-purified Polyclonal Antibody (Catalog # AF2085) or control antibody (AB-108-C) for 15 minutes in an ultrasonic bath, followed by Biotinylated Anti-Goat IgG Secondary Antibody (BAF109). Immunocomplexes were captured using 50 µL of MagCellect Streptavidin Ferrofluid (MAG999) and DNA was purified using chelating resin solution. TheVEGFpromoter was detected by standard PCR.Detection of Brachyury in NCI-H460 cells by Flow Cytometry.
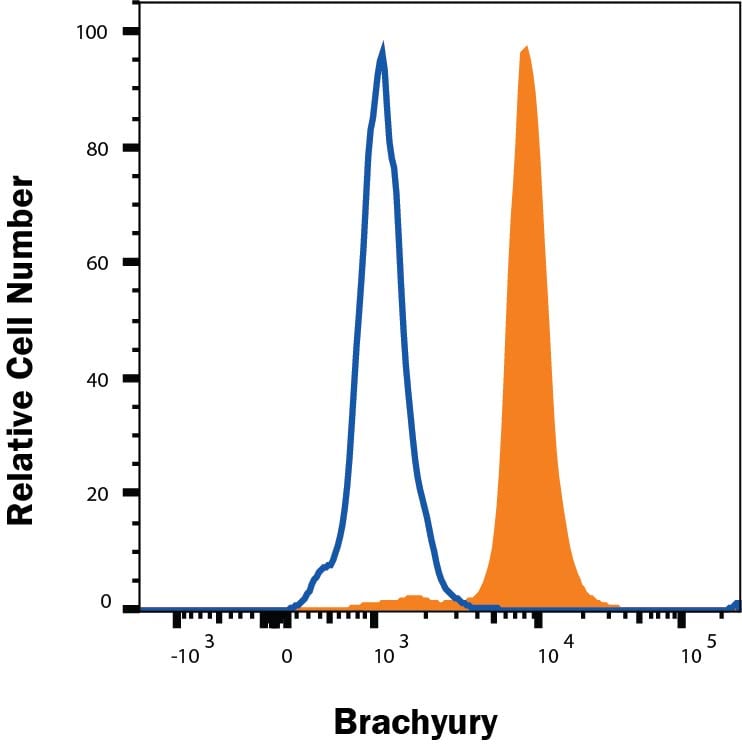
NCI-H460 cells were stained with Goat Anti-Human/Mouse Brachyury Antigen Affinity-purified Polyclonal Antibody (Catalog # AF2085, filled histogram) or isotype control antibody (Catalog # AB-108-C, open histogram), followed by Phycoerythrin-conjugated Anti-Goat IgG Secondary Antibody (Catalog # F0107). To facilitate intracellular staining, cells were fixed and permeabilized with FlowX FoxP3 Fixation & Permeabilization Buffer Kit (Catalog # FC012). View our protocol for Staining Intracellular Molecules.Detection of Human Brachyury by Immunocytochemistry/Immunofluorescence
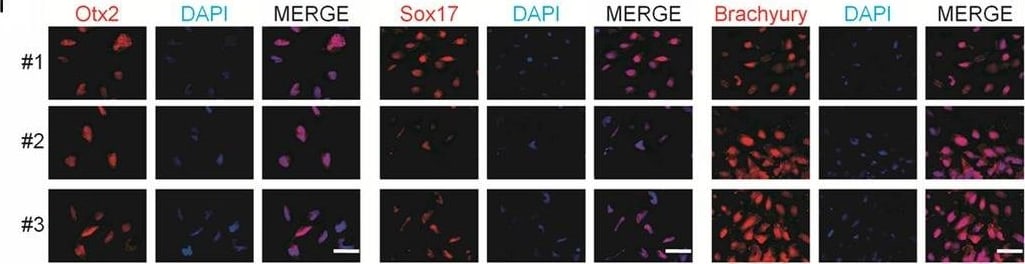
Generation and characterization of systemic lupus erythematosus (SLE)‐specific human‐induced pluripotent stem cells (hiPSCs). (A) Dermal fibroblasts derived from patient with SLE were reprogrammed into iPSCs using Sendai virus vectors and three clones (#1, #2 and #3) were characterized. RT‐PCR confirms the loss of transgenes in hiPSCs‐SLE (lanes 1, 2 and 3), presence (lane 4) in infected fibroblasts (ipF‐SLE) and absence of Sendai viral transgenes in parental fibroblasts (pF‐SLE) (lane 5). Full‐length gels are presented in File S2. (B) The hiPSCs‐SLE colonies expressed alkaline phosphatase. Scale bar, 500 μm. (C) RT‐qPCR analysis of pluripotency genes OCT4, NANOG, SOX2 and REX1 was performed in fibroblasts and in hiPSCs derived from patient with SLE. All expression values are normalized to GAPDH and relative donor fibroblasts. Data are mean ± SEM and all statistical analysis was made between hiPSCs‐SLE clones and relative fibroblasts by Student's t test showing P‐values ≤ .05 in each comparison. (D) PluriTest assays combines novelty score (blue) on x‐axis and pluripotency score (red) on y‐axis. hiPSCs‐SLE localize in the red cloud suggesting the empirical distribution of pluripotent cells compared to non‐pluripotent blue cloud. (E) Representative images of M‐FISH staining show normal karyotypes of hiPSCs‐SLE clones. (F) Immunofluorescence analysis of pluripotent stem cell markers Nanog (green), Oct4 (red) and co‐staining with DAPI (blue) in hiPSCs‐SLE. Scale bar, 50 μm. (G) Representative images of floating and adherent EBs derived from hiPSCs‐SLE at differentiation day 8 and 18, respectively. Scale bar, 500 μm. (H) RT‐qPCR results confirm the capability of hiPSCs‐SLE to differentiate into all three germ layers. The expression levels of GATA4, HAND1 and PAX6 in EBs are relative to undifferentiated hiPSCs. All expression values are normalized to GAPDH and relative hiPSCs. Data are mean ± SEM and all statistical analysis was made between EBs‐SLE and relative hiPSCs‐SLE clones by Student's t test showing P‐values ≤ .05 in each comparison. (I) Immunostaining of Otx2 (ectoderm marker), Sox17 (endoderm marker), Brachyury (mesoderm marker) and co‐staining with DAPI (blue) in EBs derived from hiPSCs‐SLE. Scale bar, 50 μm Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/31536674), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse Brachyury by Immunocytochemistry/ Immunofluorescence
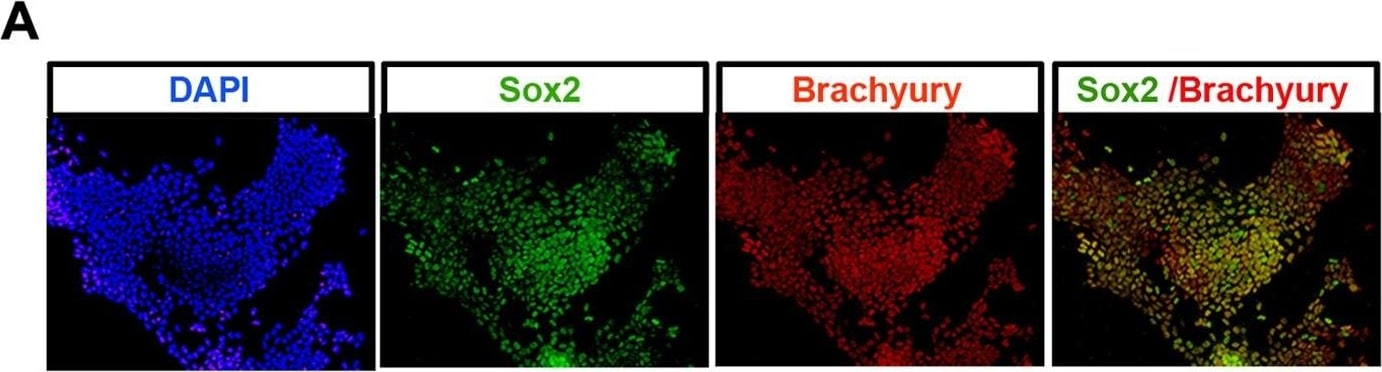
Generation of NMPs from EpiSCs.(A) Brachyury/Sox2 immunocytochemistry in EpiSC cultures treated with FGF/CHIR for 72 h. (B) qPCR analysis for indicated markers in mouse EpiSCs treated with FGF/CHIR. Error bars = s.d. (n = 3). n/d, not determined. Results are represented as log10 ratio of expression versus untreated EpiSCs. The data used to generate the plot can be found in Data S4. (C) Combined fluorescence/brightfield microscopy showing donor cell incorporation of grafted GFP+ EpiSC differentiated for 48 h in FGF/CHIR after 48 h embryo culture. (D) Table summarizing the incorporation of grafted GFP+ EpiSC differentiated for 24 h or 48 h in Fgf/Wnt within host embryos. NT, neural tube; Som, somite; PSM, presomitic mesoderm; n/a, not applicable. (E) Representative examples of donor cell incorporation (green, GFP) and differentiation (red, immunofluorescence for indicated markers). Cell nuclei were stained with DAPI (blue). White boxes indicate the position of magnified images of GFP+ cells. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/25157815), licensed under a CC-BY license. Not internally tested by R&D Systems.Applications for Human/Mouse Brachyury Antibody
Chromatin Immunoprecipitation (ChIP)
Immunocytochemistry
Sample: Immersion fixed differentiated human embryonic stem cells and BG01V human embryonic stem cells differentiated into mesoderm
Immunohistochemistry
Sample: Immersion fixed frozen sections of embryonic mouse notochord (E9.5)
Intracellular Staining by Flow Cytometry
Sample: NCI-H460 cells fixed and permeabilized with FlowX FoxP3 Fixation & Permeabilization Buffer Kit (Catalog # FC012).
Western Blot
Sample: Recombinant Human Brachyury and NCI‑H460 human large cell lung carcinoma cell line
Reviewed Applications
Read 4 reviews rated 4.3 using AF2085 in the following applications:
Flow Cytometry Panel Builder
Bio-Techne Knows Flow Cytometry
Save time and reduce costly mistakes by quickly finding compatible reagents using the Panel Builder Tool.
Advanced Features
- Spectra Viewer - Custom analysis of spectra from multiple fluorochromes
- Spillover Popups - Visualize the spectra of individual fluorochromes
- Antigen Density Selector - Match fluorochrome brightness with antigen density
Formulation, Preparation, and Storage
Purification
Reconstitution
Reconstitute at 0.2 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Formulation
Shipping
Stability & Storage
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: Brachyury
Alternate Names
Entrez Gene IDs
Gene Symbol
UniProt
Additional Brachyury Products
Product Documents for Human/Mouse Brachyury Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human/Mouse Brachyury Antibody
BG01V cells are licensed from ViaCyte, Inc.
For research use only
Related Research Areas
Citations for Human/Mouse Brachyury Antibody
Customer Reviews for Human/Mouse Brachyury Antibody (4)
Have you used Human/Mouse Brachyury Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
-
Application: ImmunohistochemistrySample Tested: Tumor cell lyastesSpecies: MouseVerified Customer | Posted 04/13/2018
-
Application: Immunocytochemistry/ImmunofluorescenceSample Tested: Mesodermal cells differentiated from mouse ESCs and Embryonic stem cellsSpecies: MouseVerified Customer | Posted 09/12/2016Immunohistochemistry on fixed 2D cell culture. Primary antibody was used at 5ug/mL and incubated overnight. Brachyury-positive cells are visualized in red, nuclei are counterstained by Dapi (blue).
-
Application: ImmunofluorescenceSample Tested: See PMID 24319666Species: MouseVerified Customer | Posted 01/06/2015
-
Application: ImmunofluorescenceSample Tested: See PMID 23658023Species: HumanVerified Customer | Posted 01/06/2015
There are no reviews that match your criteria.
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- 7-Amino Actinomycin D (7-AAD) Cell Viability Flow Cytometry Protocol
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- ChIP Protocol Video
- Chromatin Immunoprecipitation (ChIP) Protocol
- Chromatin Immunoprecipitation Protocol
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Extracellular Membrane Flow Cytometry Protocol
- Flow Cytometry Protocol for Cell Surface Markers
- Flow Cytometry Protocol for Staining Membrane Associated Proteins
- Flow Cytometry Staining Protocols
- Flow Cytometry Troubleshooting Guide
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- ICC Cell Smear Protocol for Suspension Cells
- ICC Immunocytochemistry Protocol Videos
- ICC for Adherent Cells
- IHC Sample Preparation (Frozen sections vs Paraffin)
- Immunocytochemistry (ICC) Protocol
- Immunocytochemistry Troubleshooting
- Immunofluorescence of Organoids Embedded in Cultrex Basement Membrane Extract
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Intracellular Flow Cytometry Protocol Using Alcohol (Methanol)
- Intracellular Flow Cytometry Protocol Using Detergents
- Intracellular Nuclear Staining Flow Cytometry Protocol Using Detergents
- Intracellular Staining Flow Cytometry Protocol Using Alcohol Permeabilization
- Intracellular Staining Flow Cytometry Protocol Using Detergents to Permeabilize Cells
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Propidium Iodide Cell Viability Flow Cytometry Protocol
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Liperfluo
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Characterization of Human Th22 Cells
- Protocol for the Characterization of Human Th9 Cells
- Protocol for the Fluorescent ICC Staining of Cell Smears - Graphic
- Protocol for the Fluorescent ICC Staining of Cultured Cells on Coverslips - Graphic
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent ICC Staining of Cells on Coverslips
- Protocol for the Preparation and Fluorescent ICC Staining of Non-adherent Cells
- Protocol for the Preparation and Fluorescent ICC Staining of Stem Cells on Coverslips
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- Protocol for the Preparation of a Cell Smear for Non-adherent Cell ICC - Graphic
- Protocol: Annexin V and PI Staining by Flow Cytometry
- Protocol: Annexin V and PI Staining for Apoptosis by Flow Cytometry
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Fluorokine Flow Cytometry Kits
- Troubleshooting Guide: Immunohistochemistry
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Associated Pathways