Epithelial (E)‑Cadherin (ECAD), also known as cell-CAM120/80 in the human, uvomorulin in the mouse, Arc-1 in the dog, and L-CAM in the chicken, is a member of the Cadherin family of cell adhesion molecules. Cadherins are calcium-dependent transmembrane proteins which bind to one another in a homophilic manner. On their cytoplasmic side, they associate with the three catenins, alpha, beta, and gamma (plakoglobin). This association links the cadherin protein to the cytoskeleton. Without association with the catenins, the cadherins are non-adhesive. Cadherins play a role in development, specifically in tissue formation. They may also help to maintain tissue architecture in the adult. E‑Cadherin may also play a role in tumor development, as loss of E‑Cadherin has been associated with tumor invasiveness. E‑Cadherin is a classical cadherin molecule. Classical cadherins consist of a large extracellular domain which contains DXD and DXNDN repeats responsible for mediating calcium‑dependent adhesion, a single-pass transmembrane domain, and a short carboxy-terminal cytoplasmic domain responsible for interacting with the catenins. E‑Cadherin contains five extracellular calcium‑binding domains of approximately 110 amino acids each.

Key Product Details
Validated by
Orthogonal Validation
Species Reactivity
Validated:
Human, Mouse
Cited:
Human, Mouse, Canine, Ovine, Primate - Macaca mulatta (Rhesus Macaque), Transgenic Mouse
Applications
Validated:
Immunohistochemistry, Western Blot, Flow Cytometry, Dual RNAscope ISH-IHC Compatible, Immunocytochemistry, Simple Western, CyTOF-ready
Cited:
Immunohistochemistry, Immunohistochemistry-Paraffin, Western Blot, Neutralization, Flow Cytometry, Immunocytochemistry/Immunofluorescence, Immunocytochemistry, IF/IHC, Zymography
Label
Unconjugated
Antibody Source
Polyclonal Goat IgG
Loading...
Product Specifications
Immunogen
Mouse myeloma cell line NS0-derived recombinant human E-Cadherin
Asp155-Ile707
Accession # P12830
Asp155-Ile707
Accession # P12830
Specificity
Detects human E-Cadherin in direct ELISAs and Western blots.
Clonality
Polyclonal
Host
Goat
Isotype
IgG
Scientific Data Images for Human/Mouse E‑Cadherin Antibody
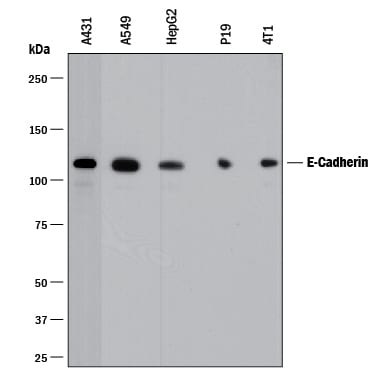
Detection of Human and Mouse E‑Cadherin by Western Blot.
Western blot shows lysates of A431 human epithelial carcinoma cell line, A549 human lung carcinoma cell line, HepG2 human hepatocellular carcinoma cell line, P19 mouse embryonal carcinoma cell line, and 4T1 mouse breast cancer cell line. PVDF membrane was probed with 0.5 µg/mL of Goat Anti-Human/Mouse E-Cadherin Antigen Affinity-purified Polyclonal Antibody (Catalog # AF648) followed by HRP-conjugated Anti-Goat IgG Secondary Antibody (Catalog # HAF017). A specific band was detected for E-Cadherin at approximately 110 kDa (as indicated). This experiment was conducted under reducing conditions and using Immunoblot Buffer Group 1.Detection of Human E‑Cadherin by Simple WesternTM.
Simple Western lane view shows lysates of Exosome Standards (HT‑29) (NBP3-11685) and A431 human epithelial carcinoma cell line, loaded at 0.5 mg/ml. A specific band was detected for E‑Cadherin at approximately 123 kDa (as indicated) using 5 µg/ml of Goat Anti-Human/Mouse E‑Cadherin Antigen Affinity-purified Polyclonal Antibody (Catalog # AF648) followed by HRP-conjugated Donkey Anti-Goat Secondary Antibody (Catalog # 042-206). This experiment was conducted under reducing conditions and using the 12-230kDa separation system.Detection of E‑Cadherin in MCF‑7 Human Cell Line by Flow Cytometry.
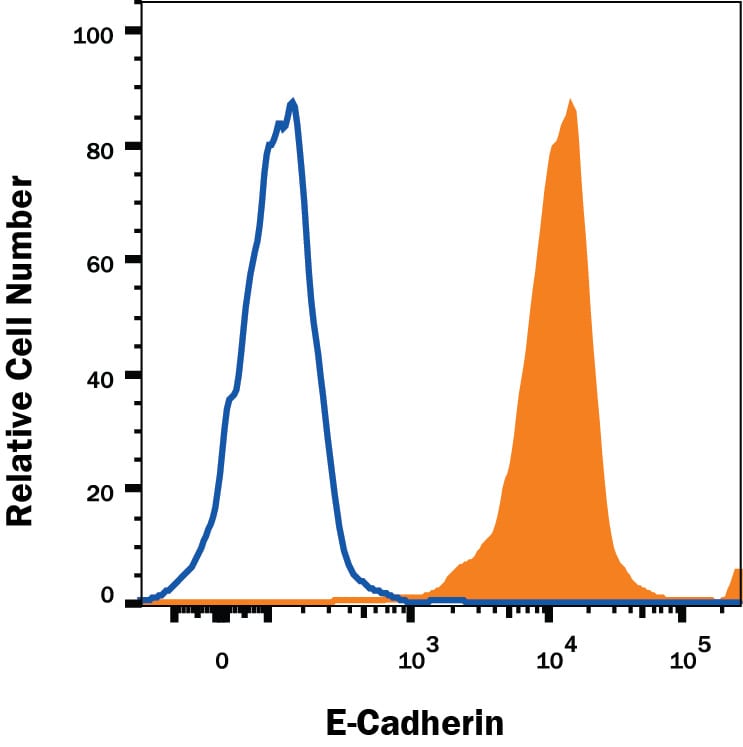
MCF-7 human breast cancer cell line was stained with Goat Anti-Human/Mouse E-Cadherin Antigen Affinity-purified Polyclonal Antibody (Catalog # AF648, filled histogram) or isotype control antibody (Catalog # AB-108-C, open histogram), followed by Allophycocyanin-conjugated Anti-Goat IgG Secondary Antibody (Catalog # F0108). View our protocol for Staining Membrane-associated Proteins.E‑Cadherin in Human Epidermoid Carcinoma Cells.
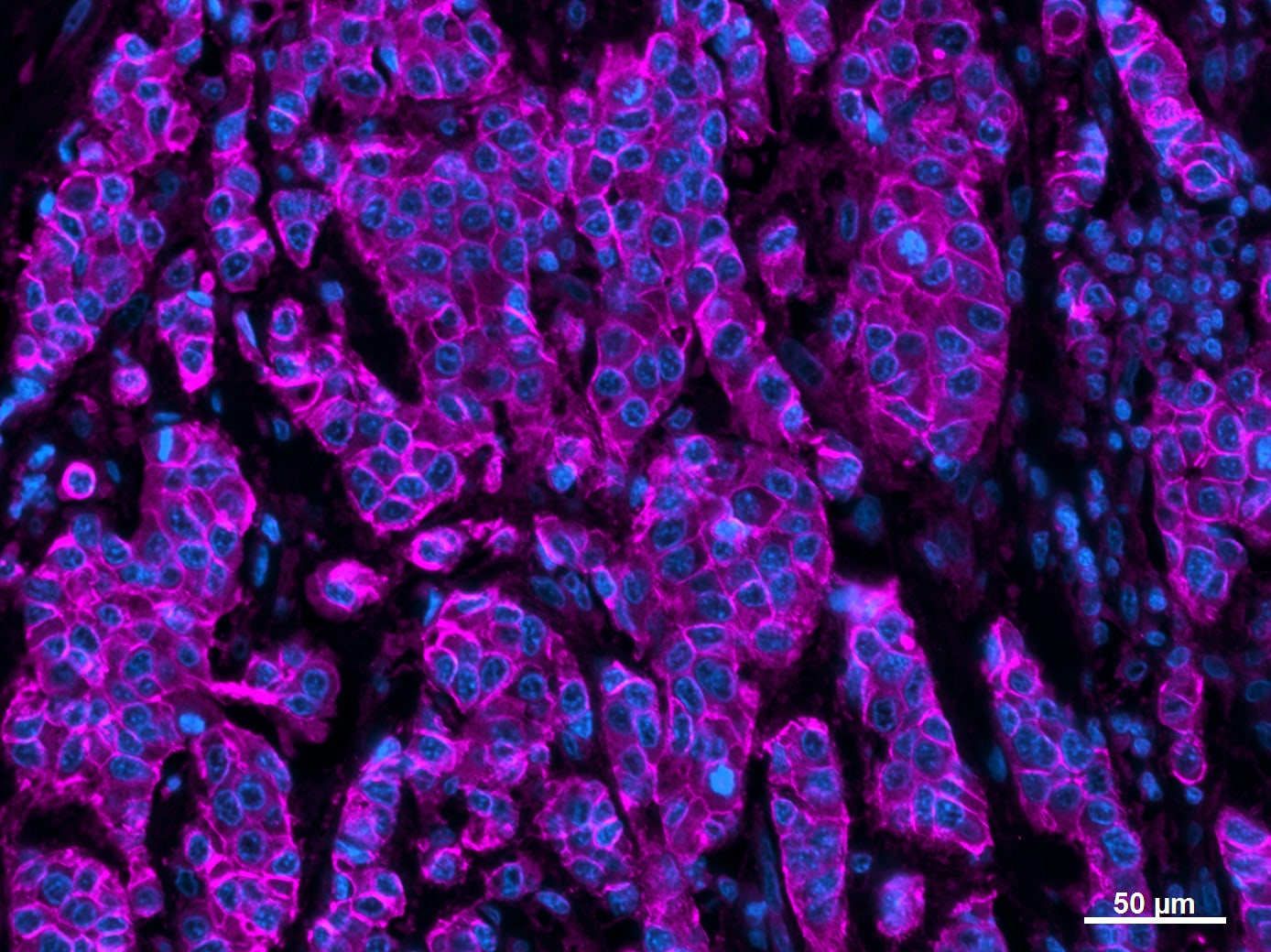
E-Cadherin was detected in immersion fixed human epidermoid carcinoma cells using 10 µg/mL Goat Anti-Human/Mouse E-Cadherin Antigen Affinity-purified Polyclonal Antibody (Catalog # AF648) for 3 hours at room temperature. Cells were stained with the NorthernLights™ 557-conjugated Anti-Goat IgG Secondary Antibody (red; Catalog # NL001) and counterstained with DAPI (blue). View our protocol for Fluorescent ICC Staining of Cells on Coverslips.E‑Cadherin and SOX2 in BG01V Human Stem Cells.
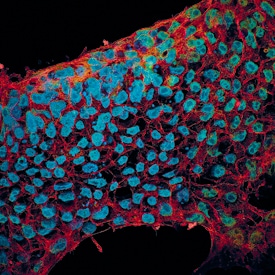
E-Cadherin and SOX2 were detected in BG01V human embryonic stem cells using 10 µg/mL Goat Anti-Human/Mouse E-Cadherin Antigen Affinity-purified Polyclonal Antibody (Catalog # AF648) and 10 µg/mL Human/Mouse SOX2 Monoclonal Antibody (Catalog # MAB2018). Cells were incubated with primary antibodies for 3 hours at room temperature. Cells were stained for E-Cadherin using the NorthernLights™ 557-conjugated Anti-Goat IgG Secondary Antibody (green; Catalog # NL001) and for SOX2 using the NorthernLights 493-conjugated Anti-Mouse Secondary Antibody (red; Catalog # NL009). Cells were counterstained with DAPI (blue). View our protocol for Fluorescent ICC Staining of Cells on Coverslips.E‑Cadherin in Human Stomach.
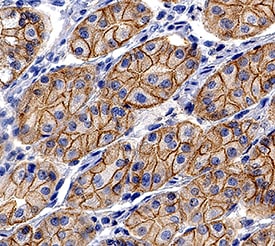
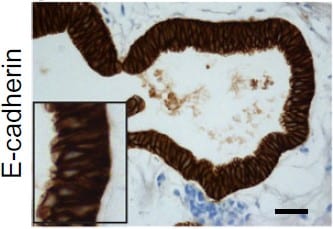
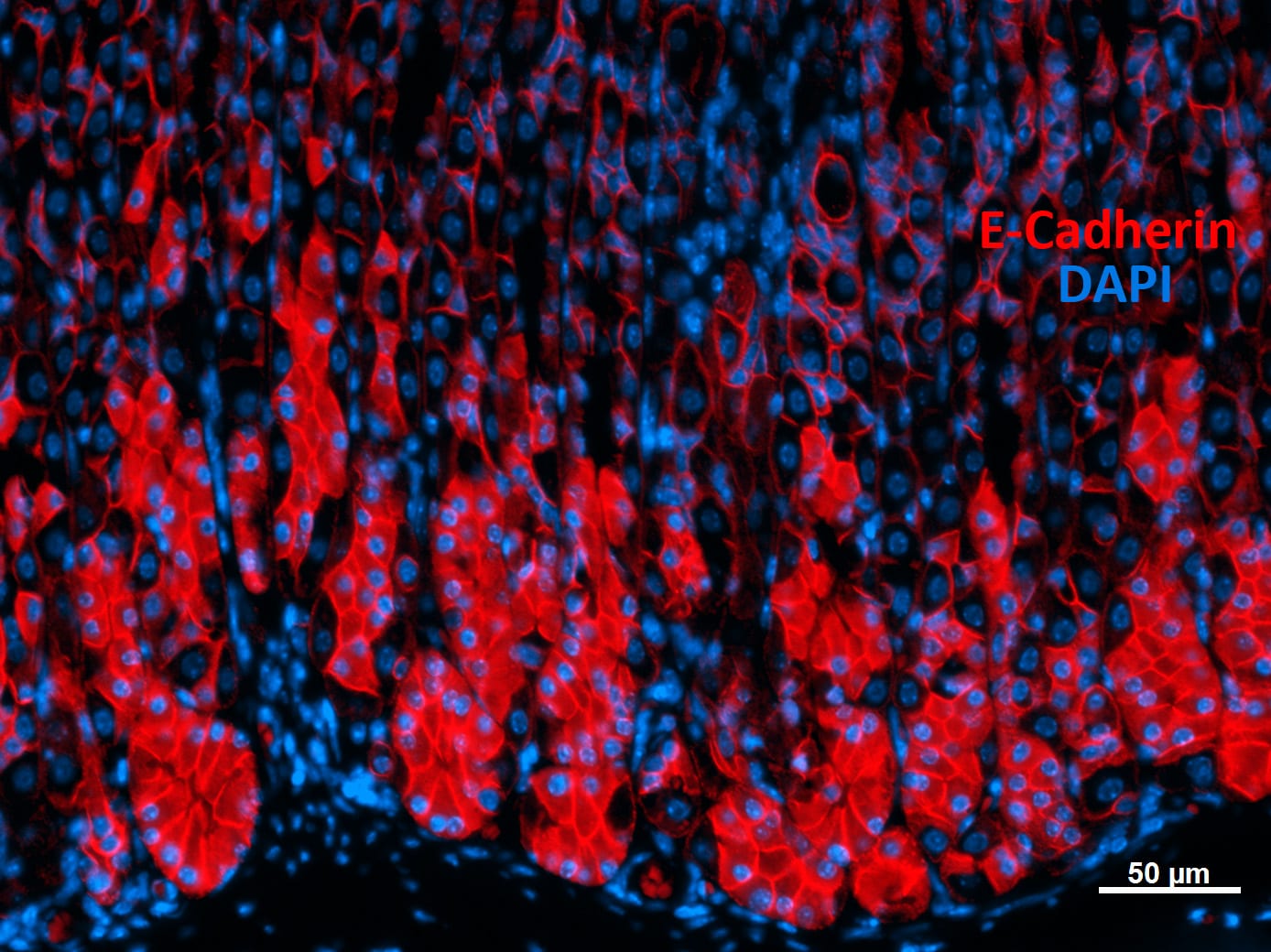
E-Cadherin was detected in immersion fixed paraffin-embedded sections of human stomach using Goat Anti-Human/Mouse E-Cadherin Antigen Affinity-purified Polyclonal Antibody (Catalog # AF648) at 0.3 µg/mL for 1 hour at room temperature followed by incubation with the Anti-Goat IgG VisUCyte™ HRP Polymer Antibody (Catalog # VC004). Tissue was stained using DAB (brown) and counterstained with hematoxylin (blue). Specific staining was localized to cell membrane and cytoplasm in gastric glands. View our protocol for IHC Staining with VisUCyte HRP Polymer Detection Reagents.Detection of Human and Mouse E‑Cadherin by Simple WesternTM.
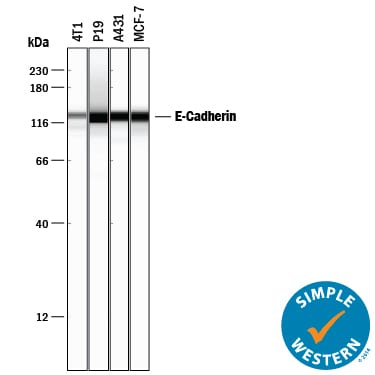
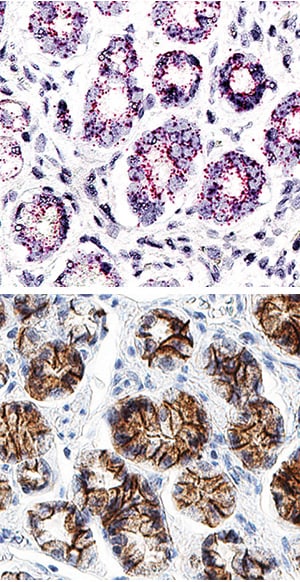
Simple Western lane view shows lysates of 4T1 mouse breast cancer cell line, P19 mouse embryonal carcinoma cell line, A431 human epithelial carcinoma cell line, and MCF-7 human breast cancer cell line, loaded at 0.2 mg/mL. A specific band was detected for E-Cadherin at approximately 128 kDa (as indicated) using 5 µg/mL of Goat Anti-Human/Mouse E-Cadherin Antigen Affinity-purified Polyclonal Antibody (Catalog # AF648) followed by 1:50 dilution of HRP-conjugated Anti-Goat IgG Secondary Antibody (Catalog # HAF017). This experiment was conducted under reducing conditions and using the 12-230 kDa separation system.E-Cadherin in Human Stomach Using Dual RNAscope®ISH and IHC.
E-Cadherin mRNA was detected in formalin-fixed paraffin-embedded tissue sections of human stomach probed with ACD RNAScope®Probe (Catalog # 311091) and stained using ACD RNAscope®2.5 HD Detection Reagents-Red (top image, Catalog # 32260). Adjacent tissue section was processed for immunohistochemistry using R&D Systems Goat Anti-Human E-Cadherin Antigen Affinity-purified Polyclonal Antibody (Catalog # AF648) at 0.3 ug/mL for 1 hour at room temperature followed by incubation with the Anti-Goat IgG VisUCyte HRP Polymer Antibody (R&D Systems, Catalog # VC004) and DAB chromogen (lower image, yellow-brown). Tissues were counterstained with hematoxylin (blue).Detection of Human E-Cadherin by Immunocytochemistry/Immunofluorescence
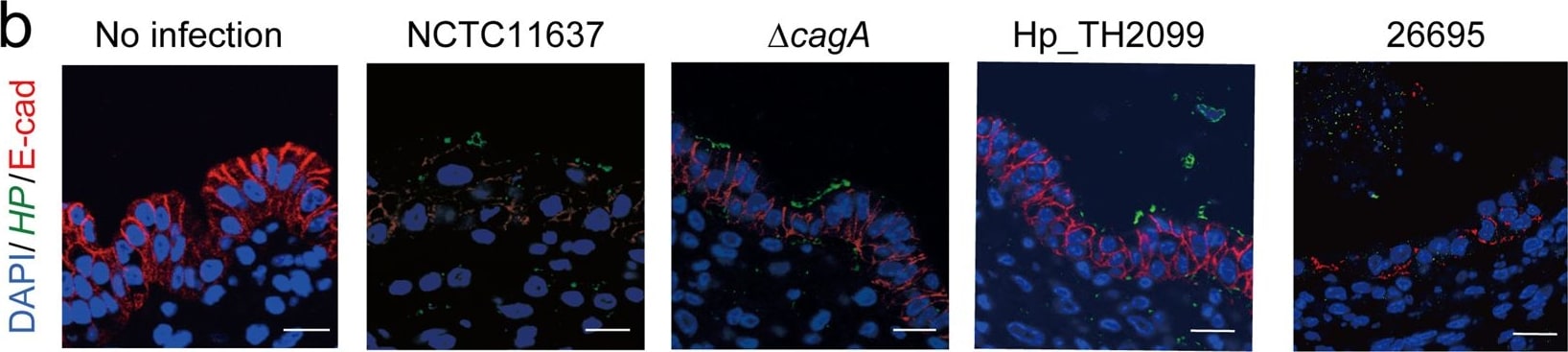
Impact of H. pylori infection on ES cell-derived gastric organoids. (a) HE staining of ES-cell derived gastric organoids infected with H. pylori for 12 hours. Scale bar: 200 µm. (b) Immunostaining of gastric organoids infected with H. pylori for 12 hours. “No infection” indicates that the organoid was injected with bacteria-free Brucella Broth. HP, H. pylori; E-cad, E-cadherin. Scale bar: 10 µm. Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/30374120), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human E-Cadherin by Immunocytochemistry/Immunofluorescence
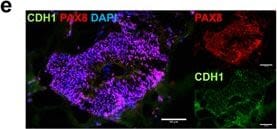
Development and Characterization of an iPSC-Derived Fallopian Tube Organoid. (a) Schematic of factors involved in the differentiation of fallopian tube organoids. (b) Bright field image and H&E staining of FTE organoid at day 14. (c–e) Immunocytochemistry for FTE markers TUBB4A, FOXJ1 and PAX8 and epithelial marker CDH1 (E-Cadherin) at organoid culture day 14. (f) Immunocytochemistry for FTE markers PAX8, TUBB4A, OVGP1 and epithelial marker CDH1 at FTE organoid culture day 45, along with human fallopian tube tissue. (g) Immunocytochemistry for FTE markers TUBB4A and PAX8 at FTE organoid culture day 45. (h) Gene expression of fallopian tube markers OVGP1 (for 2 different primers), FOXJ1, TNFaIP2 and PAX8, as well as kidney markers SALL1 and FOXD1 at organoids culture day 45, human fallopian tube and kidney. The color matrix of the heat map represents the log2(Ratio) of each individual gene relative to its expression at the iPSC stage. Relative gene expression to iPSC stage (day 0) was calculated using delta delta Ct method and normalized to endogenous GAPDH level for 87iCTR-n3 iPSC line (i) H&E staining of FTE organoid at culture day 45 and day 180, and human fallopian tube tissue. Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/28878359), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse E-Cadherin by Immunohistochemistry
Immunohistological visualization of engineered EcN strains in tissue sections.These sections are taken from proximal colons of mice receiving different bacteria. Sectioning protocol was designed to preserve mucus and luminal content. Sections were stained with fluorescently labeled antibodies: anti-E-cadherin (green), anti-Muc2 (red), and anti-LPS (blue). The first column shows bright-field images of the sections. The last column shows an overlay of all stains. The white dotted lines represent the boundary of the epithelium and mucus layers. The leftmost parts represent the epithelium, the center parts represent the mucus layers and the rightmost parts represent the lumen (scale bar = 100 μm). Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/31811125), licensed under a CC-BY license. Not internally tested by R&D Systems.Applications for Human/Mouse E‑Cadherin Antibody
Application
Recommended Usage
CyTOF-ready
Ready to be labeled using established conjugation methods. No BSA or other carrier proteins that could interfere with conjugation.
Dual RNAscope ISH-IHC Compatible
0.3-15 µg/mL
Sample: Immersion fixed paraffin-embedded sections of human stomach
Sample: Immersion fixed paraffin-embedded sections of human stomach
Flow Cytometry
0.25 µg/106 cells
Sample: MCF‑7 human breast cancer cell line
Sample: MCF‑7 human breast cancer cell line
Immunocytochemistry
5-15 µg/mL
Sample: Immersion fixed BG01V human embryonic stem cells, immersion fixed human epidermoid carcinoma cells, and immersion fixed A549 human lung carcinoma cell line untreated or treated with 10 ng/ML TGF-beta (Catalog # 240-B) for 48 hours
Sample: Immersion fixed BG01V human embryonic stem cells, immersion fixed human epidermoid carcinoma cells, and immersion fixed A549 human lung carcinoma cell line untreated or treated with 10 ng/ML TGF-beta (Catalog # 240-B) for 48 hours
Immunohistochemistry
0.3-15 µg/mL
Sample: Immersion fixed paraffin-embedded sections of human stomach
Sample: Immersion fixed paraffin-embedded sections of human stomach
Simple Western
5 µg/mL
Sample: Exosome Standards (HT-29) (Catalog # NBP3-11685), 4T1 mouse breast cancer cell line, P19 mouse embryonal carcinoma cell line, A431 human epithelial carcinoma cell line, and MCF‑7 human breast cancer cell line
Sample: Exosome Standards (HT-29) (Catalog # NBP3-11685), 4T1 mouse breast cancer cell line, P19 mouse embryonal carcinoma cell line, A431 human epithelial carcinoma cell line, and MCF‑7 human breast cancer cell line
Western Blot
0.5 µg/mL
Sample: A431 human epithelial carcinoma cell line, A549 human lung carcinoma cell line, HepG2 human hepatocellular carcinoma cell line, P19 mouse embryonal carcinoma cell line, and 4T1 mouse breast cancer cell line
Sample: A431 human epithelial carcinoma cell line, A549 human lung carcinoma cell line, HepG2 human hepatocellular carcinoma cell line, P19 mouse embryonal carcinoma cell line, and 4T1 mouse breast cancer cell line
Reviewed Applications
Read 6 reviews rated 4 using AF648 in the following applications:
Flow Cytometry Panel Builder
Bio-Techne Knows Flow Cytometry
Save time and reduce costly mistakes by quickly finding compatible reagents using the Panel Builder Tool.
Advanced Features
- Spectra Viewer - Custom analysis of spectra from multiple fluorochromes
- Spillover Popups - Visualize the spectra of individual fluorochromes
- Antigen Density Selector - Match fluorochrome brightness with antigen density
Formulation, Preparation, and Storage
Purification
Antigen Affinity-purified
Reconstitution
Reconstitute at 0.2 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. See Certificate of Analysis for details.
*Small pack size (-SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
*Small pack size (-SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: E-Cadherin
References
- Bussemakers, M.J.G. et al. (1993) Mol. Biol. Reports 17:123.
- Overduin, M. et al. (1995) Science 267:386.
- Takeichi, M. (1991) Science 251:1451.
Alternate Names
Arc-1, CAD1, Cadherin-1, CD324, CDH1, Cell-CAM120/80, ECAD, ECadherin, L-CAM, Uvomorulin
Gene Symbol
CDH1
UniProt
Additional E-Cadherin Products
Product Documents for Human/Mouse E‑Cadherin Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human/Mouse E‑Cadherin Antibody
For research use only
Citations for Human/Mouse E‑Cadherin Antibody
Customer Reviews for Human/Mouse E‑Cadherin Antibody (6)
4 out of 5
6 Customer Ratings
Have you used Human/Mouse E‑Cadherin Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
Showing
1
-
5 of
6 reviews
Showing All
Filter By:
-
Application: ImmunohistochemistrySample Tested: human colorectal cancer tissueSpecies: HumanVerified Customer | Posted 10/24/2023IHC staining for CDH1 was performed on human colon cancer tissue
-
Application: ImmunohistochemistrySample Tested: embryonic brain/ choroid plexusSpecies: MouseVerified Customer | Posted 01/22/2019The antibody did not give any specific staining on mouse paraffin sections of choroid plexus, Epitope retrieval: Sodium citrateBio-Techne ResponseThank you for reviewing our product. We are sorry to hear that this antibody did not perform as expected. We have been in touch with the customer to resolve this issue according to our Product Guarantee and to the customer’s satisfaction.
-
Application: ImmunohistochemistrySample Tested: Kidney cancer tissueSpecies: HumanVerified Customer | Posted 12/10/2017
-
Application: ImmunohistochemistrySample Tested: skin tumorSpecies: HumanVerified Customer | Posted 08/29/2016Antigen retrieval: 10mM Citrate pH6 Antibody dilution: 1:400 Secondary antibody: Alexa 647 donkey anti-goat secondary antibody
-
Application: ImmunohistochemistrySample Tested: Stomach tissueSpecies: MouseVerified Customer | Posted 08/22/2016Antigen retrieval: 10mM Citrate buffer pH6. Antibody dilution: 1:400 Secondary antibody: CY3 conjugated donkey anti-goat secondary antibody @ 1:300
-
Application: ImmunohistochemistrySample Tested: HindgutSpecies: HumanVerified Customer | Posted 07/19/2016
There are no reviews that match your criteria.
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- 7-Amino Actinomycin D (7-AAD) Cell Viability Flow Cytometry Protocol
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Extracellular Membrane Flow Cytometry Protocol
- Flow Cytometry Protocol for Cell Surface Markers
- Flow Cytometry Protocol for Staining Membrane Associated Proteins
- Flow Cytometry Staining Protocols
- Flow Cytometry Troubleshooting Guide
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- ICC Cell Smear Protocol for Suspension Cells
- ICC Immunocytochemistry Protocol Videos
- ICC for Adherent Cells
- IHC Sample Preparation (Frozen sections vs Paraffin)
- ISH-IHC Protocol for Chromogenic Detection on Formalin Fixed Paraffin Embedded (FFPE) Tissue
- Immunocytochemistry (ICC) Protocol
- Immunocytochemistry Troubleshooting
- Immunofluorescence of Organoids Embedded in Cultrex Basement Membrane Extract
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Intracellular Flow Cytometry Protocol Using Alcohol (Methanol)
- Intracellular Flow Cytometry Protocol Using Detergents
- Intracellular Nuclear Staining Flow Cytometry Protocol Using Detergents
- Intracellular Staining Flow Cytometry Protocol Using Alcohol Permeabilization
- Intracellular Staining Flow Cytometry Protocol Using Detergents to Permeabilize Cells
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Propidium Iodide Cell Viability Flow Cytometry Protocol
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Liperfluo
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Characterization of Human Th22 Cells
- Protocol for the Characterization of Human Th9 Cells
- Protocol for the Fluorescent ICC Staining of Cell Smears - Graphic
- Protocol for the Fluorescent ICC Staining of Cultured Cells on Coverslips - Graphic
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent ICC Staining of Cells on Coverslips
- Protocol for the Preparation and Fluorescent ICC Staining of Non-adherent Cells
- Protocol for the Preparation and Fluorescent ICC Staining of Stem Cells on Coverslips
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- Protocol for the Preparation of a Cell Smear for Non-adherent Cell ICC - Graphic
- Protocol: Annexin V and PI Staining by Flow Cytometry
- Protocol: Annexin V and PI Staining for Apoptosis by Flow Cytometry
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Fluorokine Flow Cytometry Kits
- Troubleshooting Guide: Immunohistochemistry
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...
Associated Pathways