The tumor suppressor gene PTEN (phosphatase and tensin homolog deleted on chromosome 10), also known as MMAC1 (mutated in multiple advanced cancers 1), encodes a phosphatase that contains the catalytic signature motif (HCxxGxxRS/T) found in all members of the protein tyrosine phosphatase family. In vitro, the recombinant PTEN has both lipid phosphatase and protein phosphatase activities (1, 2). Interestingly, accumulating evidence has shown that the tumor suppressor activity of PTEN relies on its ability to dephosphorylate phosphatidylinositol (3,4,5)-triphosphate specifically at position 3 of the inositol ring (3). This activity reduces the levels of phosphatidylinositol (3,4,5)-triphosphate which is specifically produced from phosphatidylinositol (4,5)-diphosphate by PI 3-kinase upon activation by a variety of stimuli. Therefore, PTEN antagonizes PI 3-kinase-induced downstream signaling events and cellular processes including cell growth, apoptosis and cell motility. In vivo, the importance of PTEN catalytic activity in its tumor suppressor functions is underscored by the fact that the majority of PTEN missense mutations detected in tumor specimens target the phosphatase domain and cause a loss in PTEN phosphatase activity (4).
Human/Mouse PTEN Antibody
R&D Systems | Catalog # MAB847

Key Product Details
Validated by
Species Reactivity
Validated:
Cited:
Applications
Validated:
Cited:
Label
Antibody Source
Product Specifications
Immunogen
Thr2-Val403
Accession # P60484
Specificity
Clonality
Host
Isotype
Scientific Data Images for Human/Mouse PTEN Antibody
Detection of Mouse PTEN by Western Blot.
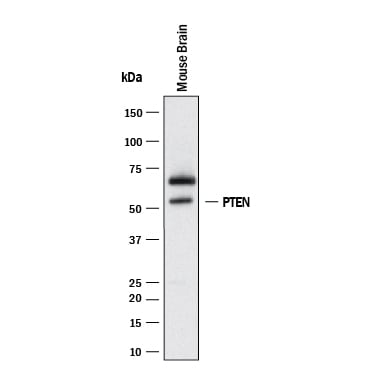
Western blot shows lysates of mouse brain tissue. PVDF membrane was probed with 2 µg/mL of Mouse Anti-Human/Mouse PTEN Monoclonal Antibody (Catalog # MAB847) followed by HRP-conjugated Anti-Mouse IgG Secondary Antibody (HAF018). A specific band was detected for PTEN at approximately 54 kDa (as indicated). This experiment was conducted under reducing conditions and using Western Blot Buffer Group 1.**For Western blot, we recommend the use of Rabbit Anti-Human/Mouse/Rat PTEN Affinitiy-purified Polyclonal Ab (AF847).PTEN in Human Liver.
PTEN was detected in immersion fixed paraffin-embedded sections of human liver array using Mouse Anti-Human/Mouse PTEN Monoclonal Antibody (Catalog # MAB847) at 25 µg/mL overnight at 4 °C. Tissue was stained using the Anti-Mouse HRP-DAB Cell & Tissue Staining Kit (brown; Catalog # CTS002) and counterstained with hematoxylin (blue). Lower panel shows a lack of labeling if primary antibodies are omitted and tissue is stained only with secondary antibody followed by incubation with detection reagents. View our protocol for Chromogenic IHC Staining of Paraffin-embedded Tissue Sections.Detection of PTEN in Human U937 cell line by Flow Cytometry.
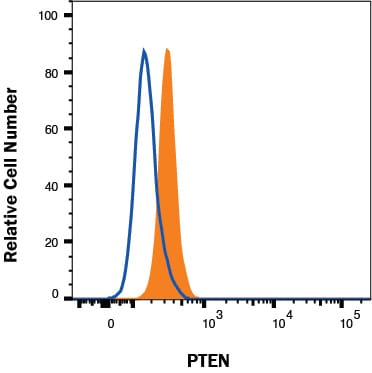
Human U937 histiocytic lymphoma cell line was stained with Mouse Anti-Human/Mouse PTEN Monoclonal Antibody (Catalog # MAB847, filled histogram) or isotype control antibody (MAB002, open histogram), followed by Allophycocyanin-conjugated Anti-Mouse IgG F(ab')2Secondary Antibody (F0101B). To facilitate intracellular staining, cells were fixed with Flow Cytometry Fixation Buffer (FC004) and permeabilized with Flow Cytometry Permeabilization/Wash Buffer I (FC005). Staining was performed using our protocol for Staining Intracellular Molecules.Detection of PTEN in Human PBMC lymphocytes by Flow Cytometry.
Human peripheral blood mononuclear cell lymphocytes were stained with Mouse Anti-Human/Mouse PTEN Monoclonal Antibody (Catalog # MAB847, filled histogram) or isotype control antibody (Catalog # MAB002, open histogram), followed by Phycoerythrin-conjugated Anti-Mouse IgG F(ab')2Secondary Antibody (Catalog # F0102B). To facilitate intracellular staining, cells were fixed with paraformaldehyde and permeabilized with saponin.Western Blot Shows Human PTEN Specificity by Using Knockout Cell Line.
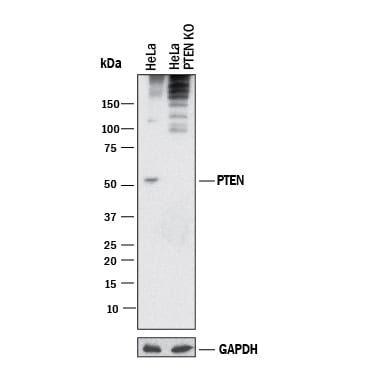
Western blot shows lysates of HeLa human cervical epithelial carcinoma parental cell line and PTEN knockout HeLa cell line (KO). PVDF membrane was probed with 2 µg/mL of Mouse Anti-Human/Mouse PTEN Monoclonal Antibody (Catalog # MAB847) followed by HRP-conjugated Anti-Mouse IgG Secondary Antibody (HAF018). A specific band was detected for PTEN at approximately 55 kDa (as indicated) in the parental HeLa cell line, but is not detectable in knockout HeLa cell line. GAPDH (MAB5718) is shown as a loading control. This experiment was conducted under reducing conditions and using Western Blot Buffer Group 1.Detection of Rat PTEN by Western Blot
Western blot analysis of apoptotic-related proteins after furin siRNA transfection.Gramulosa cells were transfected with furin siRNA or control siRNA for 48 h before being subjected to protein extraction and Western blot with the indicated antibodies. Shown at the left were representative Western blot images. Three such experiments were quantified by measuring the intensity of apoptotic-related proteins relative to the alpha -tubulin (loading) control. The anti-apoptotic proteins XIAP and p-Akt were decreased by appropriate 3-folds (**, P<0.01) and 2-folds (**, P<0.01). On the contrary, the levels of pro-apoptotic, cleaved caspase-3 and PTEN were increased appropriate 4-folds (**, P<0.01) and 4.5-folds (**, P<0.01). Image collected and cropped by CiteAb from the following publication (https://dx.plos.org/10.1371/journal.pone.0050479), licensed under a CC-BY license. Not internally tested by R&D Systems.Applications for Human/Mouse PTEN Antibody
CyTOF-ready
Immunohistochemistry
Sample: Immersion fixed paraffin-embedded sections of human liver
Intracellular Staining by Flow Cytometry
Sample: Human peripheral blood lymphocytes and U937 cell line fixed with paraformaldehyde and permeabilized with saponin
Knockout Validated
Western Blot
**For Western blot, we recommend Rabbit Anti-Human/Mouse/Rat PTEN Affinity-purified Polyclonal Ab (Catalog # AF847)
Flow Cytometry Panel Builder
Bio-Techne Knows Flow Cytometry
Save time and reduce costly mistakes by quickly finding compatible reagents using the Panel Builder Tool.
Advanced Features
- Spectra Viewer - Custom analysis of spectra from multiple fluorochromes
- Spillover Popups - Visualize the spectra of individual fluorochromes
- Antigen Density Selector - Match fluorochrome brightness with antigen density
Formulation, Preparation, and Storage
Purification
Reconstitution
Reconstitute at 0.5 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Formulation
Shipping
Stability & Storage
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: PTEN
References
- Maehama, T. and J. Dixon (1998) J. Biol. Chem. 273:13375.
- Das, S. et al. (2003) Proc. Natl. Acad. Sci. USA 100:7491.
- Myers, M. et al. (1998) Proc. Natl. Acad. Sci. USA 95:13513.
- Waite, K. and C. Eng (2002) Am. J. Hum. Genet. 70:829.
Long Name
Alternate Names
Gene Symbol
UniProt
Additional PTEN Products
Product Documents for Human/Mouse PTEN Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human/Mouse PTEN Antibody
This product is covered by the following U.S. patent: USSN # 10/299,003.
For research use only
Related Research Areas
Citations for Human/Mouse PTEN Antibody
Customer Reviews for Human/Mouse PTEN Antibody
There are currently no reviews for this product. Be the first to review Human/Mouse PTEN Antibody and earn rewards!
Have you used Human/Mouse PTEN Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- 7-Amino Actinomycin D (7-AAD) Cell Viability Flow Cytometry Protocol
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Extracellular Membrane Flow Cytometry Protocol
- Flow Cytometry Protocol for Cell Surface Markers
- Flow Cytometry Protocol for Staining Membrane Associated Proteins
- Flow Cytometry Staining Protocols
- Flow Cytometry Troubleshooting Guide
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- IHC Sample Preparation (Frozen sections vs Paraffin)
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Intracellular Flow Cytometry Protocol Using Alcohol (Methanol)
- Intracellular Flow Cytometry Protocol Using Detergents
- Intracellular Nuclear Staining Flow Cytometry Protocol Using Detergents
- Intracellular Staining Flow Cytometry Protocol Using Alcohol Permeabilization
- Intracellular Staining Flow Cytometry Protocol Using Detergents to Permeabilize Cells
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Propidium Iodide Cell Viability Flow Cytometry Protocol
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Liperfluo
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Characterization of Human Th22 Cells
- Protocol for the Characterization of Human Th9 Cells
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- Protocol: Annexin V and PI Staining by Flow Cytometry
- Protocol: Annexin V and PI Staining for Apoptosis by Flow Cytometry
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Fluorokine Flow Cytometry Kits
- Troubleshooting Guide: Immunohistochemistry
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Associated Pathways
 Akt Signaling Pathway
Akt Signaling Pathway
 mTOR Signaling Pathway
mTOR Signaling Pathway