Human/Mouse Wnt‑11 Antibody
R&D Systems | Catalog # AF2647

Key Product Details
Validated by
Biological Validation
Species Reactivity
Validated:
Human, Mouse
Cited:
Human
Applications
Validated:
Immunohistochemistry, Western Blot, Immunocytochemistry
Cited:
Immunohistochemistry-Paraffin
Label
Unconjugated
Antibody Source
Polyclonal Goat IgG
Loading...
Product Specifications
Immunogen
E. coli-derived recombinant mouse Wnt‑11 peptide
Leu39-Ala79 and Lys225-Arg297
Accession # Q059Y4
Leu39-Ala79 and Lys225-Arg297
Accession # Q059Y4
Specificity
Detects mouse Wnt-11 in direct ELISAs and Western blots.
Clonality
Polyclonal
Host
Goat
Isotype
IgG
Scientific Data Images for Human/Mouse Wnt‑11 Antibody
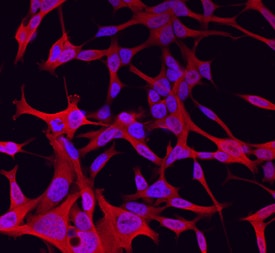
Wnt‑11 in LNCaP Human Cell Line.
Wnt-11 was detected in immersion fixed LNCaP human prostate cancer cell line using Goat Anti-Mouse Wnt-11 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF2647) at 10 µg/mL for 3 hours at room temperature. Cells were stained using the NorthernLights™ 557-conjugated Anti-Goat IgG Secondary Antibody (red; Catalog # NL001) and counterstained with DAPI (blue). Specific staining was localized to cytoplasm. View our protocol for Fluorescent ICC Staining of Cells on Coverslips.Detection of Mouse Wnt-11 by Western Blot
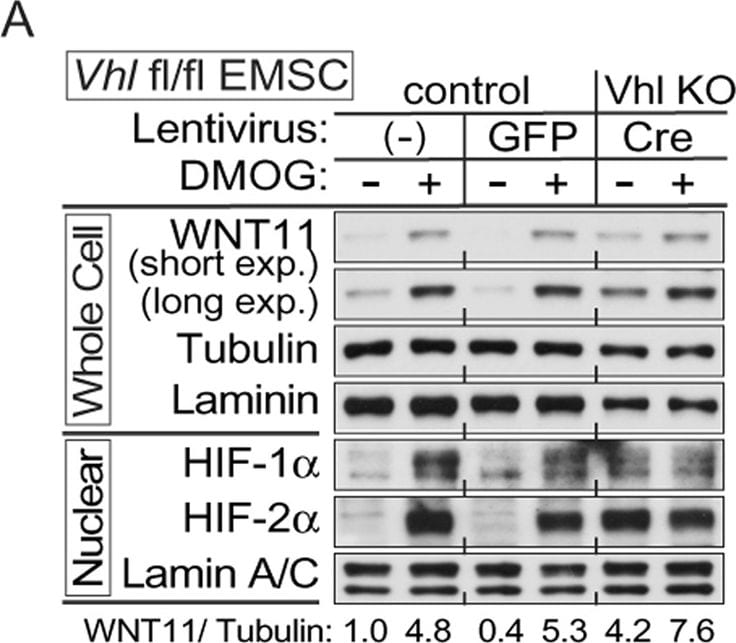
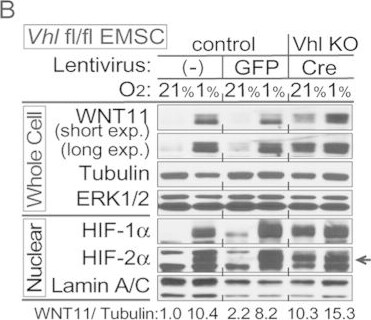
Hypoxia induces expression of WNT11 through VHL.(A,B) Higher basal levels of WNT11 protein in Vhl-deleted cells (lenti-Cre infected Vhlf/f). EMSCs isolated from Vhlf/f mouse were infected with lentivirus carrying either GFP gene (for control) or Cre recombinase (for knockout). Non-infected cells were also used as a control. Immunoblot analysis of control or Vhl KO EMSCs treated with 0.1 mM DMOG (A), and EMSCs exposed to air (21% O2) or hypoxia (1% O2) for 24 hrs (B). Laminin, alpha -tubulin, and lamin A/C were used as loading controls, WNT11 normalized to alpha -Tubulin was shown. (C,D) Inactivation of the Vhl gene results in increased Wnt11 mRNA. Wnt11 and Vegf mRNA levels in liver (C) or duodenum (D) were measured by qPCR in Liver-VhlcKO or duodenum-VhlcKO and control mice (n = 5 per group). Values normalized to Tbp mRNA are expressed relative to tissues from control mice. For panels (C,D), values are mean ± s.e.m. *p < 0.05, **p < 0.01. Image collected and cropped by CiteAb from the following publication (https://www.nature.com/articles/srep21520), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human Wnt-11 by Western Blot
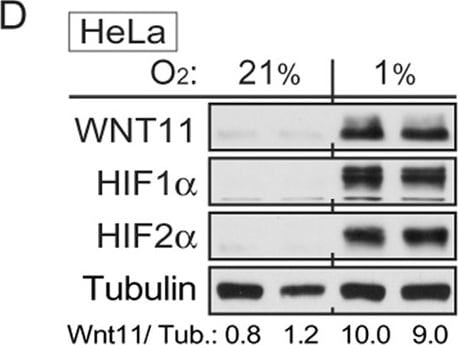
WNT11 is induced by hypoxia or hypoxic mimetics in different cell types.(A) Increased Wnt11 mRNA in EMSC adipocytes (Day 12) after hypoxia-mimetic treatments. EMSC adipocytes were treated with CoCl2 (0.1 mM), DFO (0.1 mM) or DMOG (0.1 mM) for 24 hrs. Values were normalized to Tbp mRNA and are expressed relative to control (n = 3). (B,C) Increased Wnt11 mRNA by hypoxia in EMSC preadipocytes and adipocytes (Day 0–12 after differentiation) (B), and C2C12 myoblast and myocyte (Day 0 and 8 after differentiation) (C). Wnt11 mRNA was assessed by quantitative PCR in cells exposed to air (21% O2) or hypoxia (1% O2) for 24 hrs. (n = 4). Values were normalized to Tbp mRNA and are expressed relative to 21% O2 samples (left panel). (D) Immunoblot analyses of HeLa cells under normal air or hypoxia for 24 hrs. (E,F) Induction of Wnt11 by increasing concentrations of DMOG in MDA-MB-231 cells (E) and 4T1 cells (F). (G) EMSCs treated with 0.1 mM DMOG for the indicated times. Wnt11 and Vegf mRNA expression was measured by qPCR and normalized to Tbp mRNA (n = 4). (H) WNT11 protein levels after DMOG treatment normalized to alpha -Tubulin (upper panel; n = 4). Representative immunoblots of EMSCs treated with 0.1 mM DMOG for the indicated times (Lower panel). (I) Protein expression in MDA-MB-231 cells treated with 0.1 mM DMOG. (J) Induction of Wnt11 promoter activity by hypoxia or hypoxia mimetics. pGL3-Wnt11 promoter plasmid was transfected into C2C12 cells. Cells were incubated with DMOG (left panel, n = 4) or under 21% O2 or 1% O2 (right panel, n = 8) for 24 hrs. For panels (A–C,G,H,J), values are mean ± s.e.m. *p < 0.05, **p < 0.01. For panels of immunoblotting, laminin, alpha -tubulin, and ERK were used as loading controls, WNT11 normalized to alpha -Tubulin was shown. Image collected and cropped by CiteAb from the following publication (https://www.nature.com/articles/srep21520), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse Wnt-11 by Western Blot
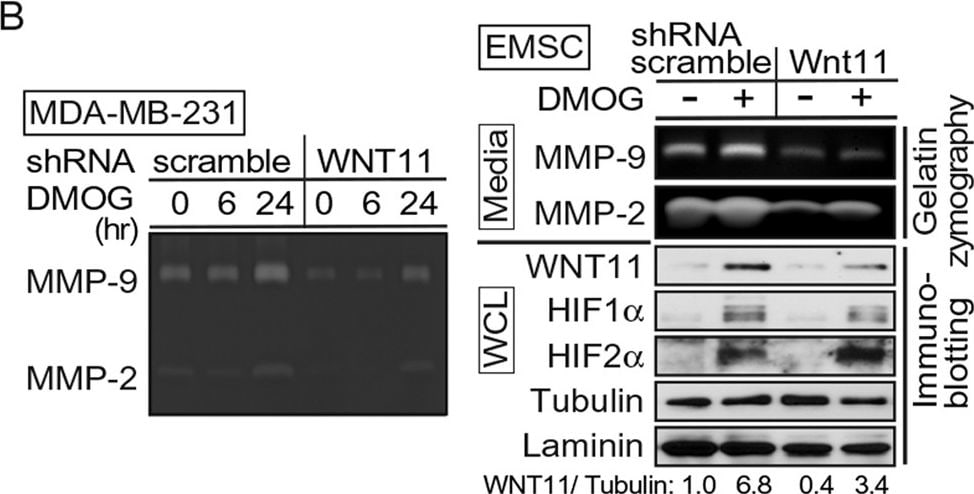
WNT11 regulates MMPs activities.(A–C) (Top panels): Serum-free medium was conditioned for 24 hrs by the indicated cells, concentrated 20-fold and assayed by gelatin zymography. Gelatinolytic activity is indicated by clear zones against a dark background of stained substrate. (Bottom): Whole cell extracts were immunoblotted with indicated antibodies. (A) Overexpression of Wnt11 in EMSC or BT473 cells enhances activity of MMP-9 and MMP-2. (B) Impaired activity of MMP-9 and MMP-2 in MDA-MB-231 cells (left) or EMSCs (right) stably expressing Wnt11 shRNAs and treated with DMOG. (C) WNT11 is required for MMP-9 and MMP-2 activity in MDA-MB-231 cells (left) or EMSCs (right) under normoxic and hypoxic culture conditions. (D) WNT11 regulates MMP2 protein in media. (Top): conditioned media from indicated cells and treatments. (Bottom): whole cell lysates were immunoblotted with indicated antibodies. (E) Recombinant WNT11 induces both MMP-2 protein and MMP-2 activity in media. (Top panels): Gelatin zymography and immunoblot of serum-free medium conditioned for the indicated times after recombinant WNT11 (r-WNT11) treatment. (Bottom): Whole cell lysates were immunoblotted with indicated antibodies. (F) MMP-2 inhibitor attenuated induced migration by WNT11. MDA-MB-231 cells infected with lentiviruses for stable expression of Wnt11 or GFP (n = 4) were incubated with either vehicle or 1 μM of ARP100. Media in the lower compartment had same concentration of DMSO or inhibitor. Values are mean ± s.e.m. *p < 0.05, **p < 0.01. For panels (A–D), HIF-1 alpha and HIF-2 alpha were shown as a marker of hypoxia, WNT11 normalized to alpha -Tubulin was shown. Image collected and cropped by CiteAb from the following publication (https://www.nature.com/articles/srep21520), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human Wnt-11 by Western Blot
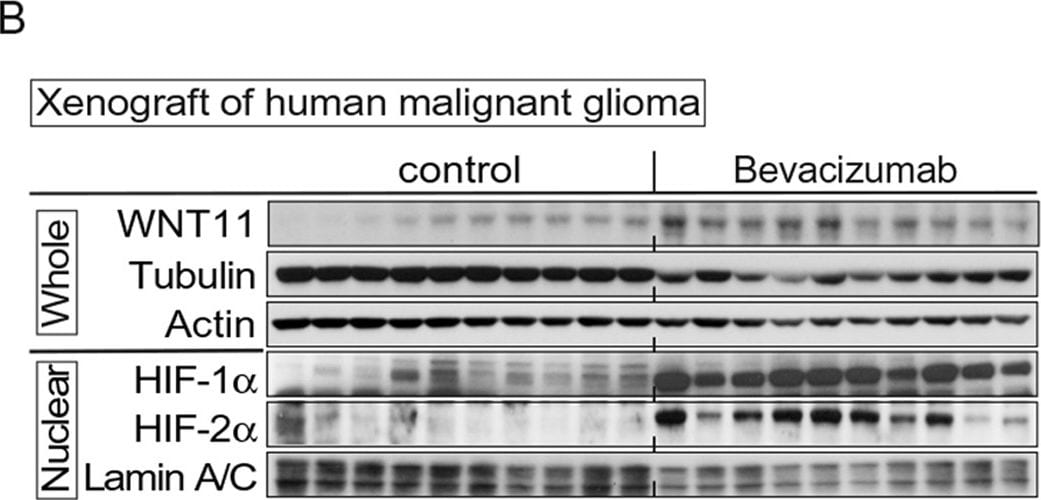
Induced WNT11 expression with tumor hypoxia and WNT11 regulates tumor growth.Antiangiogenic therapy induces WNT11 expression in the orthotopic malignant glioma model. Athymic mice implanted with U87 delta EGFR cells were administered either bevacizumab (6 mg/kg) or vehicle three times per week for 4 weeks. (A) Increased Wnt11 mRNA in xenografts from mice treated with bevacizumab. Values were normalized to HPRT mRNA and are expressed relative to control (n = 10 per group). Values are mean ± s.e.m. *p < 0.05, **p < 0.01. (B) Bevacizumab increased expression of HIF-1 alpha and HIF-2 alpha and WNT11. First 10 lanes are control tumors, and the last 10 lanes are tumors from bevacizumab-treated animals. Lysates from whole tissue and nuclei are indicated. alpha -Tubulin, actin and lamin A/C are loading controls. Image collected and cropped by CiteAb from the following publication (https://www.nature.com/articles/srep21520), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse Wnt-11 by Western Blot
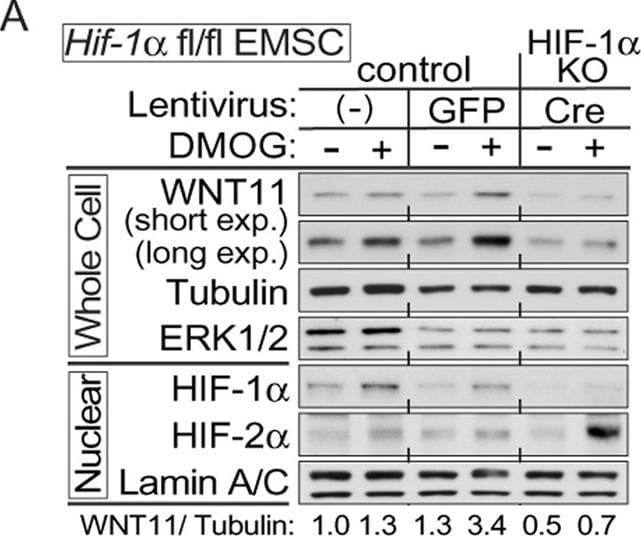
HIF-1 alpha is the predominant transcriptional regulator of WNT11 expression during hypoxia.(A,C–E) EMSCs isolated from the indicated mouse genotypes were infected with lentivirus expressing GFP or Cre recombinase. Non-infected cells and GFP infected cells served as controls. Immunoblot analyses of EMSCs derived from the indicated genotypes treated with 0.1 mM DMOG for 24 hrs. (A) Attenuated WNT11 expression in Hif-1 alpha KO EMSCs (lenti-Cre infected Hif-1af/f). (B) HIF-1 alpha regulates WNT11 expression during hypoxia. Impaired WNT11 expression in MDA-MB-231 cells stably expressing HIF-1 alpha shRNAs with hypoxia. Cells were exposed to air (21% O2) or hypoxia (1% O2) for 24 hrs (left panel). Overexpression of HIF-1 alpha in MDA-MB-231 cells enhances WNT11 expression (right panel). (C) Markedly increased WNT11 levels in Hif-1 alpha overexpressing EMSCs (lenti-Cre infected Hif-1 alpha -Tgfl-Stop). (D) Elevated HIF-1 alpha and WNT11 protein after DMOG treatment in HIF-2 alpha KO cells (lenti-Cre infectied Hif-2 alpha fl/fl). (E) Little effect of HIF-2 alpha overexpression on WNT11 protein expression in EMSC (lenti-Cre infectied Hif-2 alpha -Tgfl-Stop). (F) HIF-1 alpha binds to WNT11 and VEGF promoters as assessed by ChIP analyses (n = 3 each condition). (G) WNT11 expression was suppressed in cells with a beta -catenin deficiency. EMSCs stably expressing shRNAs against beta -catenin or scrambled control were treated with 0.1 mM DMOG for 24 hrs and analyzed by immunoblotting. (H) Co-transfection with expression vectors for beta -catenin, Hif-1 alpha and ARNT stimulates further induction of Wnt11 promoter activity. Constructs encoding Hif-1 alpha, Hif-2 alpha, ARNT, beta -catenin and Wnt11 promoter-luciferase reporter, were transiently transfected into HEK293T cells. Cells were harvested and luciferase activities were measured 48 hrs after transfection (n = 3). For panels (A–E,G), laminin, alpha -tubulin, ERK1/2 and lamin A/C were used as loading controls, WNT11 normalized to alpha -Tubulin was shown under the bots. For panels (F,H), values are mean ± s.e.m. *p < 0.05, **p < 0.01 Image collected and cropped by CiteAb from the following publication (https://www.nature.com/articles/srep21520), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse Wnt-11 by Western Blot
Hypoxia induces expression of WNT11 through VHL.(A,B) Higher basal levels of WNT11 protein in Vhl-deleted cells (lenti-Cre infected Vhlf/f). EMSCs isolated from Vhlf/f mouse were infected with lentivirus carrying either GFP gene (for control) or Cre recombinase (for knockout). Non-infected cells were also used as a control. Immunoblot analysis of control or Vhl KO EMSCs treated with 0.1 mM DMOG (A), and EMSCs exposed to air (21% O2) or hypoxia (1% O2) for 24 hrs (B). Laminin, alpha -tubulin, and lamin A/C were used as loading controls, WNT11 normalized to alpha -Tubulin was shown. (C,D) Inactivation of the Vhl gene results in increased Wnt11 mRNA. Wnt11 and Vegf mRNA levels in liver (C) or duodenum (D) were measured by qPCR in Liver-VhlcKO or duodenum-VhlcKO and control mice (n = 5 per group). Values normalized to Tbp mRNA are expressed relative to tissues from control mice. For panels (C,D), values are mean ± s.e.m. *p < 0.05, **p < 0.01. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/26861754), licensed under a CC-BY license. Not internally tested by R&D Systems.Applications for Human/Mouse Wnt‑11 Antibody
Application
Recommended Usage
Immunocytochemistry
5-15 µg/mL
Sample: Immersion fixed LNCaP human prostate cancer cell line
Sample: Immersion fixed LNCaP human prostate cancer cell line
Immunohistochemistry
5-15 µg/mL
Sample: Immersion fixed frozen sections of mouse embryo (E13-15)
Sample: Immersion fixed frozen sections of mouse embryo (E13-15)
Western Blot
0.1 µg/mL
Sample: Recombinant Mouse Wnt‑11
Sample: Recombinant Mouse Wnt‑11
Formulation, Preparation, and Storage
Purification
Antigen Affinity-purified
Reconstitution
Reconstitute at 0.2 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: Wnt-11
Long Name
Wingless-type MMTV Integration Site Family, Member 11
Alternate Names
Wnt11
Gene Symbol
WNT11
UniProt
Additional Wnt-11 Products
Product Documents for Human/Mouse Wnt‑11 Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human/Mouse Wnt‑11 Antibody
For research use only
Related Research Areas
Citations for Human/Mouse Wnt‑11 Antibody
Customer Reviews for Human/Mouse Wnt‑11 Antibody
There are currently no reviews for this product. Be the first to review Human/Mouse Wnt‑11 Antibody and earn rewards!
Have you used Human/Mouse Wnt‑11 Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- ICC Cell Smear Protocol for Suspension Cells
- ICC Immunocytochemistry Protocol Videos
- ICC for Adherent Cells
- IHC Sample Preparation (Frozen sections vs Paraffin)
- Immunocytochemistry (ICC) Protocol
- Immunocytochemistry Troubleshooting
- Immunofluorescence of Organoids Embedded in Cultrex Basement Membrane Extract
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Fluorescent ICC Staining of Cell Smears - Graphic
- Protocol for the Fluorescent ICC Staining of Cultured Cells on Coverslips - Graphic
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent ICC Staining of Cells on Coverslips
- Protocol for the Preparation and Fluorescent ICC Staining of Non-adherent Cells
- Protocol for the Preparation and Fluorescent ICC Staining of Stem Cells on Coverslips
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- Protocol for the Preparation of a Cell Smear for Non-adherent Cell ICC - Graphic
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Immunohistochemistry
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...
Associated Pathways
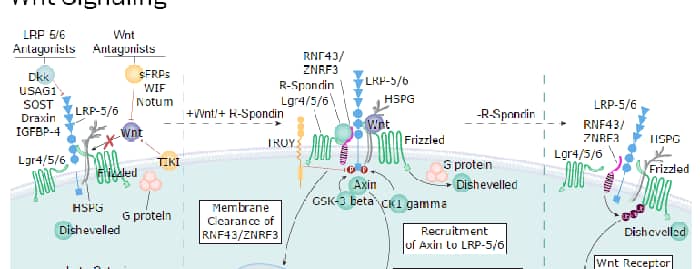
 Wnt Signaling Pathways: beta-Catenin-dependent Wnt Signaling
Wnt Signaling Pathways: beta-Catenin-dependent Wnt Signaling