HGF R, also known as Met (from N-methyl-N’-nitro-N-nitrosoguanidine induced), is a glycosylated receptor tyrosine kinase that plays a central role in epithelial morphogenesis and cancer development. HGF R is synthesized as a single chain precursor which undergoes cotranslational proteolytic cleavage. This generates a mature HGF R that is a disulfide-linked dimer composed of a 50 kDa extracellular alpha chain and a 145 kDa transmembrane beta chain (1, 2). The extracellular domain (ECD) contains a seven bladed beta -propeller sema domain, a cysteine-rich PSI/MRS, and four Ig-like E-set domains, while the cytoplasmic region includes the tyrosine kinase domain (3, 4). An alternately spliced form of mouse HGF R lacks a cytoplasmic juxtamembrane region important for regulation of signal transduction (5, 6). The sema domain, which is formed by both the alpha and beta chains of HGF R, mediates both ligand binding and receptor dimerization (3, 7). Ligand-induced tyrosine phosphorylation in the cytoplasmic region activates the kinase domain and provides docking sites for multiple SH2-containing molecules (8, 9). HGF stimulation induces HGF R downregulation via internalization and proteasome-dependent degradation (10). In the absence of ligand, HGF R forms non-covalent complexes with a variety of membrane proteins including CD44v6, CD151, EGF R, Fas, integrin alpha 6/ beta 4, plexins B1, 2, 3, and MSP R/Ron (11-18). Ligation of one complex component triggers activation of the other, followed by cooperative signaling effects (11-18). Formation of some of these heteromeric complexes is a requirement for epithelial cell morphogenesis and tumor cell invasion (11, 15, 16). Paracrine induction of epithelial cell scattering and branching tubulogenesis results from the stimulation of HGF R on undifferentiated epithelium by HGF released from neighboring mesenchymal cells (19). Genetic polymorphisms, chromosomal translocation,
over-expression, and additional splicing and proteolytic cleavage of HGF R have been described in a wide range of cancers (1). Within the ECD, mouse HGF R shares 87%, 87%, and 94% amino acid sequence identity with canine, human, and rat HGF R, respectively.

Key Product Details
Species Reactivity
Validated:
Mouse
Cited:
Human, Mouse, Rat, Transgenic Mouse
Applications
Validated:
Immunohistochemistry, Western Blot, Blockade of Receptor-ligand Interaction, Immunocytochemistry
Cited:
Immunohistochemistry, Immunohistochemistry-Paraffin, Immunohistochemistry-Frozen, Western Blot, Neutralization, Flow Cytometry, Immunocytochemistry, Immunoprecipitation, ELISA Development, ELISA Development (Capture)
Label
Unconjugated
Antibody Source
Polyclonal Goat IgG
Loading...
Product Specifications
Immunogen
S. frugiperda insect ovarian cell line Sf 21-derived recombinant mouse HGF R/c-MET
Glu25-Asn929
Accession # P16056
Glu25-Asn929
Accession # P16056
Specificity
Detects mouse HGF R/c-MET in direct ELISAs and Western blots. In direct ELISAs, approximately 10% cross-reactivity with recombinant human (rh) HGF R and less than 1% cross-reactivity with rhMSP R is observed.
Clonality
Polyclonal
Host
Goat
Isotype
IgG
Endotoxin Level
<0.10 EU per 1 μg of the antibody by the LAL method.
Scientific Data Images for Mouse HGFR/c-MET Antibody
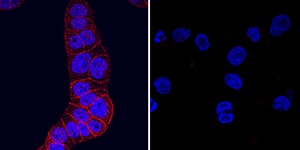
HGF R/c‑MET in HT‑29 and U937 Human Cell Lines.
HGF R/c-MET was detected in immersion fixed HT-29 human colon adenocarcinoma cell line (positive control, left panel) and U937 human histiocytic lymphoma cell line (negative control, right panel) using Goat Anti-Mouse HGF R/c-MET Antigen Affinity-purified Polyclonal Antibody (Catalog # AF527) at 5 µg/mL for 3 hours at room temperature. Cells were stained using the NorthernLights™ 557-conjugated Anti-Goat IgG Secondary Antibody (red; Catalog # NL001) and counterstained with DAPI (blue). Specific staining was localized to plasma membrane. View our protocol for Fluorescent ICC Staining of Cells on Coverslips.HGF R/c‑MET in Mouse Embryo.
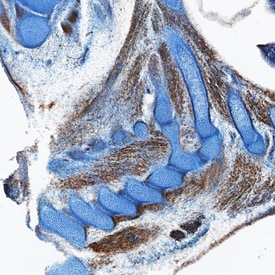
HGF R/c-MET was detected in immersion fixed frozen sections of mouse embryo (15 d.p.c.) using Goat Anti-Mouse HGF R/c-MET Antigen Affinity-purified Polyclonal Antibody (Catalog # AF527) at 15 µg/mL overnight at 4 °C. Tissue was stained using the Anti-Goat HRP-DAB Cell & Tissue Staining Kit (brown; Catalog # CTS008) and counterstained with hematoxylin (blue). Specific staining was localized to cytoplasm in muscle cells. View our protocol for Chromogenic IHC Staining of Frozen Tissue Sections.Detection of Mouse HGFR/c-MET by Immunohistochemistry
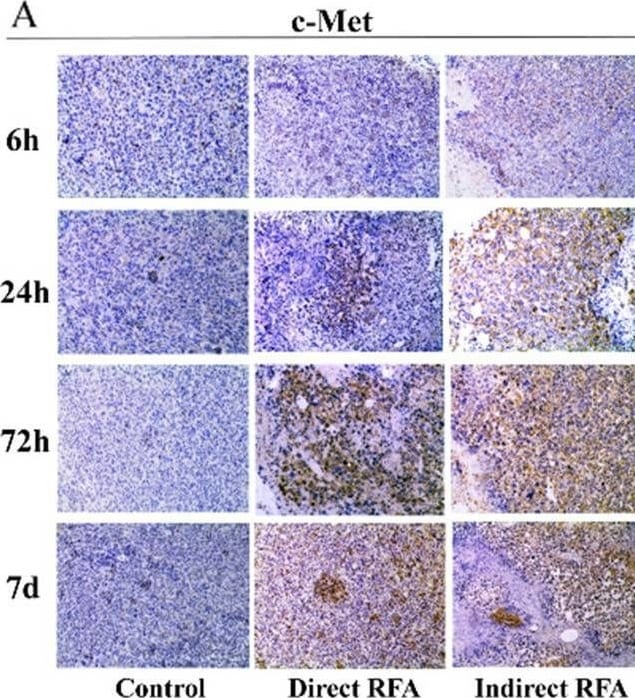
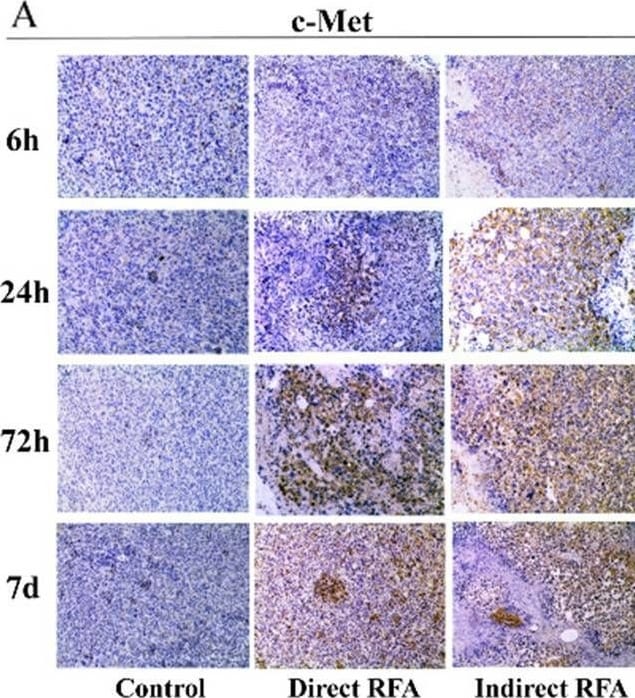
Assessment of pathologic findings obtained at 6 h, 24 h, 72 h and 7 days after treatment in different groups and the semi-quantitative analysis of c-Met (A-B), IL-6 (C-D) and HIF-alpha (E-F). The expression of c-Met increased after RFA and reached the peak at 72 h (> 50% positive rate) in both direct RFA and indirect RFA groups. The expression of IL-6 increased after RFA in the indirect RFA group and it was significantly higher than the direct RFA group from 6 h to 72 h after treatment. The expression of HIF-alpha was higher in the indirect RFA group than the direct RFA group from 6 h to 24 h after RFA. The bar graph showed the percentage of positive cells among the direct RFA group (blue), indirect RFA group (red) and control group (grey) at different time points after RFA. **P < 0.001. The representative microphotographs were under 200 × magnification (A, C, E) Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/35710408), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse HGFR/c-MET by Immunohistochemistry
Assessment of pathologic findings obtained at 6 h, 24 h, 72 h and 7 days after treatment in different groups and the semi-quantitative analysis of c-Met (A-B), IL-6 (C-D) and HIF-alpha (E-F). The expression of c-Met increased after RFA and reached the peak at 72 h (> 50% positive rate) in both direct RFA and indirect RFA groups. The expression of IL-6 increased after RFA in the indirect RFA group and it was significantly higher than the direct RFA group from 6 h to 72 h after treatment. The expression of HIF-alpha was higher in the indirect RFA group than the direct RFA group from 6 h to 24 h after RFA. The bar graph showed the percentage of positive cells among the direct RFA group (blue), indirect RFA group (red) and control group (grey) at different time points after RFA. **P < 0.001. The representative microphotographs were under 200 × magnification (A, C, E) Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/35710408), licensed under a CC-BY license. Not internally tested by R&D Systems.Applications for Mouse HGFR/c-MET Antibody
Application
Recommended Usage
Blockade of Receptor-ligand Interaction
In a functional ELISA, 0.3-1 µg/mL of this antibody will block 50% of the binding of 5 ng/mL of Recombinant Human HGF (Catalog # 256-GF) to immobilized Recombinant Mouse HGF R/c-MET Fc Chimera (Catalog # 527‑ME) coated at 1 µg/mL (100 µL/well). At 20 μg/mL, this antibody will block >90% of the binding.
Immunocytochemistry
5-15 µg/mL
Sample: Immersion fixed HT-29 human colon adenocarcinoma cell line
Sample: Immersion fixed HT-29 human colon adenocarcinoma cell line
Immunohistochemistry
5-15 µg/mL
Sample: Immersion fixed frozen sections of mouse embryo (15 d.p.c.)
Sample: Immersion fixed frozen sections of mouse embryo (15 d.p.c.)
Western Blot
0.1 µg/mL
Sample: Recombinant Mouse HGF R/c-MET Fc Chimera (Catalog # 527-ME)
Sample: Recombinant Mouse HGF R/c-MET Fc Chimera (Catalog # 527-ME)
Reviewed Applications
Read 5 reviews rated 4.4 using AF527 in the following applications:
Formulation, Preparation, and Storage
Purification
Antigen Affinity-purified
Reconstitution
Reconstitute at 0.2 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: HGFR/c-MET
References
- Birchmeier, C. et al. (2003) Nat. Rev. Mol. Cell Biol. 4:915.
- Corso, S. et al. (2005) Trends Mol. Med. 11:284.
- Gherardi, E. et al. (2003) Proc. Natl. Acad. Sci. USA 100:12039.
- Chan, A.M. et al. (1988) Oncogene 2:593.
- Lee, C.-C. and K.M. Yamada (1994) J. Biol. Chem. 269:19457.
- Lee, C.-C., et al. (1995) J. Biol. Chem. 270:507.
- Kong-Beltran, M. et al. (2004) Cancer Cell 6:75.
- Naldini, L. et al. (1991) Mol. Cell. Biol. 11:1793.
- Ponzetto, C. et al. (1994) Cell 77:261.
- Jeffers, M. et al. (1997) Mol. Cell. Biol. 17:799.
- Orian-Rousseau, V. et al. (2002) Genes Dev. 16:3074.
- Klosek, S.K. et al. (2005) Biochem. Biophys. Res. Commun. 336:408.
- Jo, M. et al. (2000) J. Biol. Chem. 275:8806.
- Wang, X. et al. (2002) Mol. Cell 9:411.
- Trusolino, L. et al. (2001) Cell 107:643.
- Giordano, S. et al. (2002) Nat. Cell Biol. 4:720.
- Conrotto, P. et al. (2004) Oncogene 23:5131.
- Follenzi, A. et al. (2000) Oncogene 19:3041.
- Sonnenberg, E. et al. (1993) J. Cell Biol. 123:223.
Long Name
Hepatocyte Growth Factor Receptor
Alternate Names
c-MET, cMET, HGF R, MET
Gene Symbol
MET
UniProt
Additional HGFR/c-MET Products
Product Documents for Mouse HGFR/c-MET Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Mouse HGFR/c-MET Antibody
For research use only
Citations for Mouse HGFR/c-MET Antibody
Customer Reviews for Mouse HGFR/c-MET Antibody (5)
4.4 out of 5
5 Customer Ratings
Have you used Mouse HGFR/c-MET Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
Showing
1
-
5 of
5 reviews
Showing All
Filter By:
-
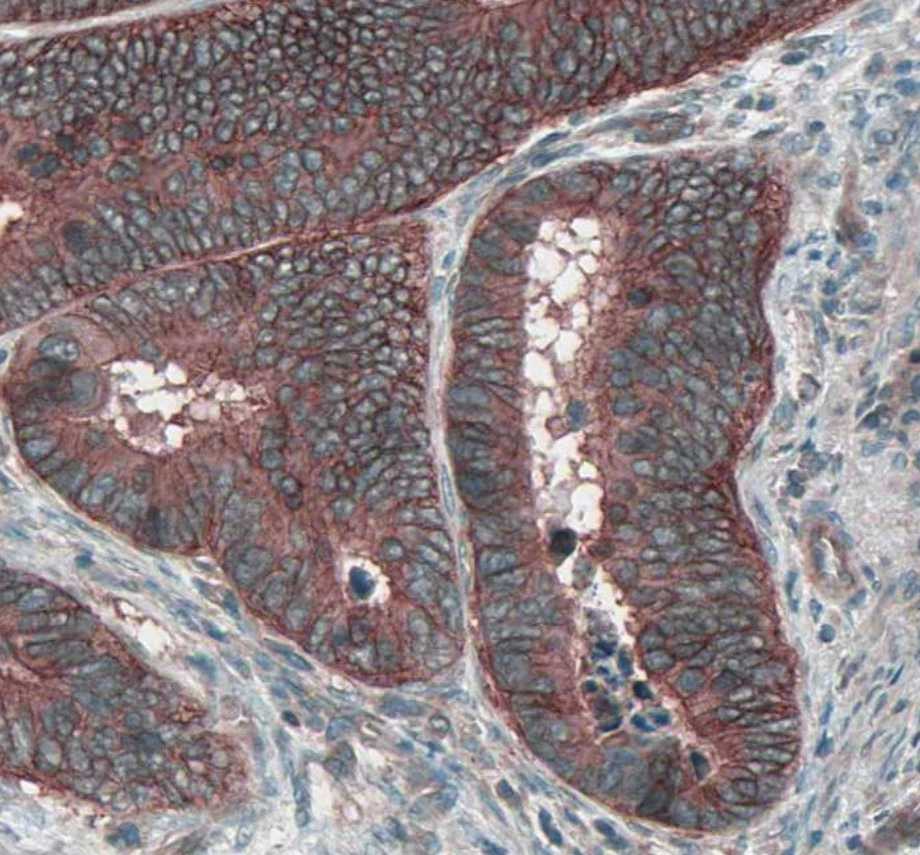
Application: ImmunohistochemistrySample Tested: Colon cancer tissueSpecies: MouseVerified Customer | Posted 01/18/2022
-
Application: Immunohistochemistry-FrozenSample Tested: See PMID 23074208Species: MouseVerified Customer | Posted 01/08/2015
-
Application: Western BlotSample Tested: See PMID 23509284Species: MouseVerified Customer | Posted 01/08/2015
-
Application: Immunohistochemistry-ParaffinSample Tested: See PMID 24218051Species: MouseVerified Customer | Posted 01/08/2015
-
Application: Immunohistochemistry-FrozenSample Tested: See PMID 24279352Species: MouseVerified Customer | Posted 01/08/2015
There are no reviews that match your criteria.
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- ICC Cell Smear Protocol for Suspension Cells
- ICC Immunocytochemistry Protocol Videos
- ICC for Adherent Cells
- IHC Sample Preparation (Frozen sections vs Paraffin)
- Immunocytochemistry (ICC) Protocol
- Immunocytochemistry Troubleshooting
- Immunofluorescence of Organoids Embedded in Cultrex Basement Membrane Extract
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Fluorescent ICC Staining of Cell Smears - Graphic
- Protocol for the Fluorescent ICC Staining of Cultured Cells on Coverslips - Graphic
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent ICC Staining of Cells on Coverslips
- Protocol for the Preparation and Fluorescent ICC Staining of Non-adherent Cells
- Protocol for the Preparation and Fluorescent ICC Staining of Stem Cells on Coverslips
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- Protocol for the Preparation of a Cell Smear for Non-adherent Cell ICC - Graphic
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Immunohistochemistry
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars