13 results for "p38 Inhibitors" in Products
p38 Inhibitors
p38 kinases are a group of mitogen-activated protein kinases (MAPKs). Like JNK/c-Jun, the p38 kinases are responsive to stress stimuli such as osmotic shock and cytokine stimulation; they are also classified as stress-activated protein kinases (SAPKs). p38 also responds to insulin and growth factor stimulation and is often activated during normal immune and inflammatory responses. The active form of p38 phosphorylates specific cellular targets and also regulates gene expression via activation ...
Selective inhibitor of p38 MAPK
| Alternate Names: | Adezmapimod |
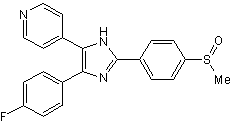
| Chemical Name: | 4-[5-(4-Fluorophenyl)-2-[4-(methylsulfonyl)phenyl]-1H-imidazol-4-yl]pyridine |
| Purity: | ≥98% |
Potent, selective inhibitor of p38 MAPK
| Chemical Name: | 4-[4-(4-Fluorophenyl)-5-(4-pyridinyl)-1H-imidazol-2-yl]phenol |
| Purity: | ≥99% |
High affinity and selective p38 kinase inhibitor
| Alternate Names: | Doramapimod |
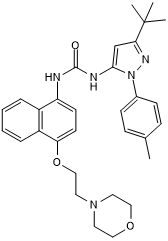
| Chemical Name: | N-[3-(1,1-Dimethylethyl)-1-(4-methylphenyl)-1H-pyrazol-5-yl]-N'-[4-[2-(4-morpholinyl)ethoxy]-1-naphthalenyl]urea |
| Purity: | ≥98% |
Selective inhibitor of p38 MAPK; water-soluble
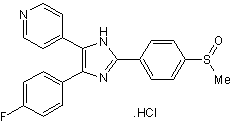
| Chemical Name: | 4-[5-(4-Fluorophenyl)-2-[4-(methylsulphonyl)phenyl]-1H-imidazol-4-yl]pyridine hydrochloride |
| Purity: | ≥98% |
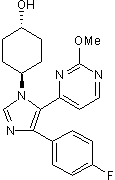
Potent, selective p38 MAPK inhibitor; orally active
| Chemical Name: | trans-4-[4-(4-Fluorophenyl)-5-(2-methoxy-4-pyrimidinyl)-1H-imidazol-1-yl]cyclohexanol |
| Purity: | ≥98% |
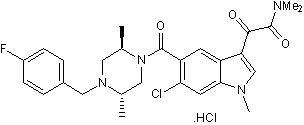
Selective p38 MAPK inhibitor
| Chemical Name: | 6-Chloro-5-[[(2R,5S)-4-[(4-fluorophenyl)methyl]-2,5-dimethyl-1-piperazinyl]carbonyl]-N,N,1-trimethyl-α-oxo-1H-Indole-3-acetamide hydrochloride |
| Purity: | ≥98% |
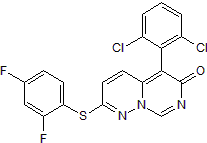
Potent and selective p38α inhibitor
| Alternate Names: | VX745,VX-745,Neflamapimod |
| Chemical Name: | 5-(2,6-Dichlorophenyl)-2-[2,4-difluorophenyl)thio]-6H-pyrimido[1,6-b]pyridazin-6-one |
| Purity: | ≥98% |
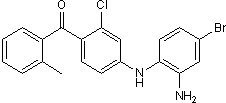
Selective inhibitor of p38α and p38β2
| Chemical Name: | (2-Methylphenyl)-[4-[(2-amino-4-bromophenyl)amino]-2-chlorophenyl]methanone |
| Purity: | ≥98% |
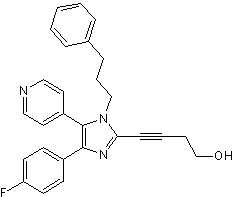
Potent, selective p38α and p38β inhibitor
| Chemical Name: | 4-[4-(4-Fluorophenyl)-1-(3-phenylpropyl)-5-(4-pyridinyl)-1H-imidazol-2-yl]-3-butyn-1-ol |
| Purity: | ≥98% |
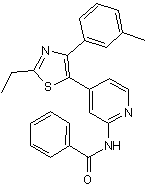
Potent p38 MAPK inhibitor; anti-inflammatory
| Chemical Name: | N-[4-[2-Ethyl-4-(3-methylphenyl)-5-thiazolyl]-2-pyridinyl]benzamide |
| Purity: | ≥98% |
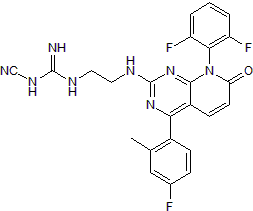
p38 MAPK inhibitor
| Chemical Name: | N-Cyano-N'-[2-[[8-(2,6-difluorophenyl)-4-(4-fluoro-2-methylphenyl)-7,8-dihydro-7-oxopyrido[2,3-d]pyrimidin-2-yl]amino]ethyl]guanidine |
| Purity: | ≥98% |
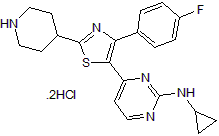
p38 MAPK inhibitor; anti-inflammatory
| Chemical Name: | N-Cyclopropyl-4-[4-(4-fluorophenyl)-2-(4-piperidinyl)-5-thiazolyl]-2-pyrimidinamine dihydrochloride |
| Purity: | ≥98% |
Potent and selective p38α/β inhibitor
| Chemical Name: | (-)-3-[3-Bromo-4-[(2,4-difluorophenyl)methoxy]-6-methyl-2-oxo-1(2H)-pyridinyl]-N,4-dimethylbenzamide |
| Purity: | ≥98% |