CD44 is a ubiquitously expressed protein that is the major receptor for hyaluronan and exerts control over cell growth and migration (1-3). Human CD44 has a 20 amino acid (aa) signal sequence, an extracellular domain (ECD) with a 100 aa hyaluronan-binding disulfide-stabilized link region and a 325-530 aa stem region, a 21 aa transmembrane domain, and a 72 aa cytoplasmic domain. Within the stem, ten variably spliced exons (v1-10, exons 6-15) produce multiple protein isoforms (1‑3). The standard or hematopoietic form, CD44s, does not include the variable segments (1‑3). Cancer aggressiveness and T cell activation have been correlated with expression of specific isoforms (1, 3). With variable N- and O-glycosylation and splicing within the stalk, CD44 can range from 80-200 kDa (1). Within the N‑terminal invariant portion of the ECD (aa 21-220), human CD44 shares 76%, 76%, 86%, 83%, and 79% identity with corresponding mouse, rat, equine, canine, and bovine CD44, respectively. The many reported functions of CD44 fall within three categories (1). First, CD44 binds hyaluronan and other ligands within the extracellular matrix and can function as a “platform” for growth factors and metalloproteinases. Second, CD44 can function as a co-receptor that modifies activity of receptors including MET and the ERBB family of tyrosine kinases. Third, the CD44 intracellular domain links the plasma membrane to the actin cytoskeleton via the ERM proteins, ezrin, radixin and moesin. CD44 can be synthesized in a soluble form (4) or may be cleaved at multiple sites by either membrane-type matrix metalloproteinases, or ADAM proteases to produce soluble ectodomains (5, 6). The cellular portion may then undergo gamma secretase-dependent intramembrane cleavage to form an A beta -like transmembrane portion and a cytoplasmic signaling portion that affects gene expression (7, 8). These cleavage events are thought to promote metastasis by enhancing tumor cell motility and growth (1, 5).
Human CD44s Pan Specific Antibody
R&D Systems | Catalog # BBA10

Key Product Details
Validated by
Knockout/Knockdown
Species Reactivity
Validated:
Human
Cited:
Human, Mouse, Rat, Naked mole-rat, Transgenic Mouse
Applications
Validated:
Knockout Validated, Immunohistochemistry, Western Blot, Flow Cytometry, Simple Western, Immunoprecipitation, CyTOF-ready
Cited:
Immunohistochemistry, Western Blot, Neutralization, Flow Cytometry, Immunocytochemistry, Simple Western, Immunoprecipitation, Cell Culture, Surface Plasmon Resonance
Label
Unconjugated
Antibody Source
Monoclonal Mouse IgG2A Clone # 2C5
Loading...
Product Specifications
Immunogen
Recombinant human CD44v3-10 (includes the invariant N-terminal exons and CD44v3-10 exons)
Specificity
Detects human CD44s on a panel of CD44 transfected COS cells by flow cytometry (Fox, S.B. et al. (1994) Cancer Res.
Clonality
Monoclonal
Host
Mouse
Isotype
IgG2A
Scientific Data Images for Human CD44s Pan Specific Antibody
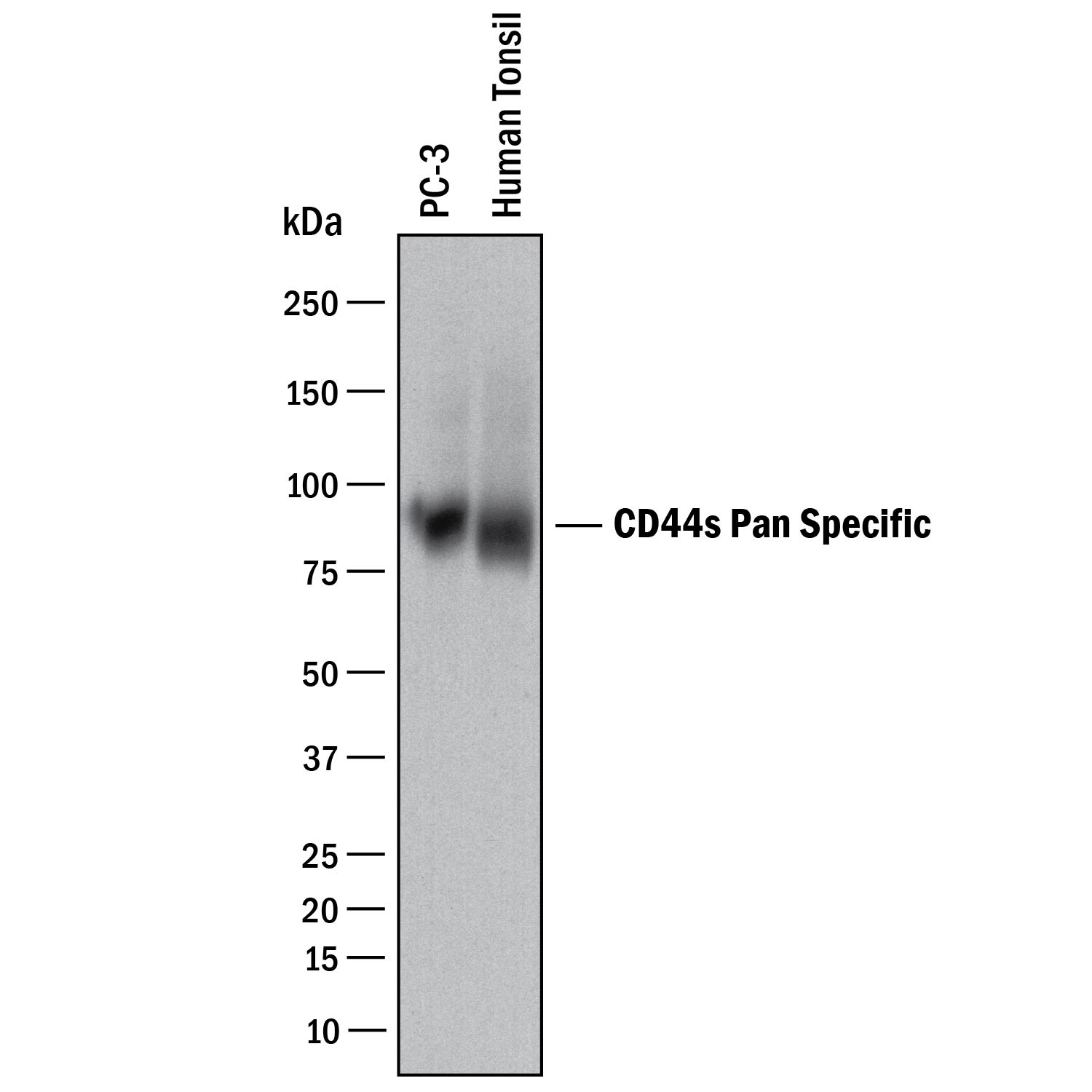
Detection of Human CD44 by Western Blot.
Western blot shows lysates of PC-3 human prostate cancer cell line and human tonsil tissue. PVDF membrane was probed with 2 µg/mL of Mouse Anti-Human CD44 s Pan Specific Monoclonal Antibody (Catalog # BBA10) followed by HRP-conjugated Anti-Mouse IgG Secondary Antibody (HAF018). A specific band was detected for CD44 at approximately 90 kDa (as indicated). This experiment was conducted under reducing conditions and using Immunoblot Buffer Group 1.CD44 in Human Lymphoma.
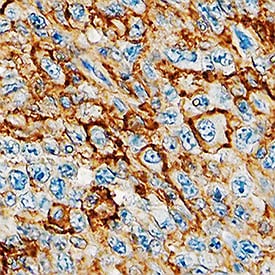
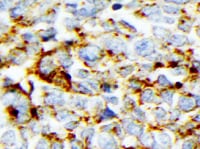
CD44 was detected in immersion fixed paraffin-embedded sections of human lymphoma using Mouse Anti-Human CD44 s Pan Specific Monoclonal Antibody (Catalog # BBA10) at 15 µg/mL overnight at 4 °C. Tissue was stained using the Anti-Mouse HRP-DAB Cell & Tissue Staining Kit (brown; CTS002) and counterstained with hematoxylin (blue). Specific staining was localized to plasma membrane. View our protocol for Chromogenic IHC Staining of Paraffin-embedded Tissue Sections.Detection of Human CD44 by Simple WesternTM.
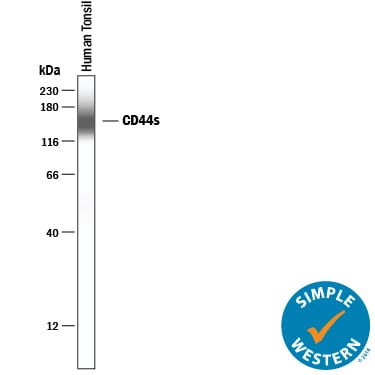
Simple Western lane view shows lysates of human tonsil tissue, loaded at 0.2 mg/mL. A specific band was detected for CD44 at approximately 156 kDa (as indicated) using 20 µg/mL of Mouse Anti-Human CD44 s Pan Specific Monoclonal Antibody (Catalog # BBA10). This experiment was conducted under reducing conditions and using the 12-230 kDa separation system.Western Blot Shows CD44 Specificity Using Knockout Cell Line.
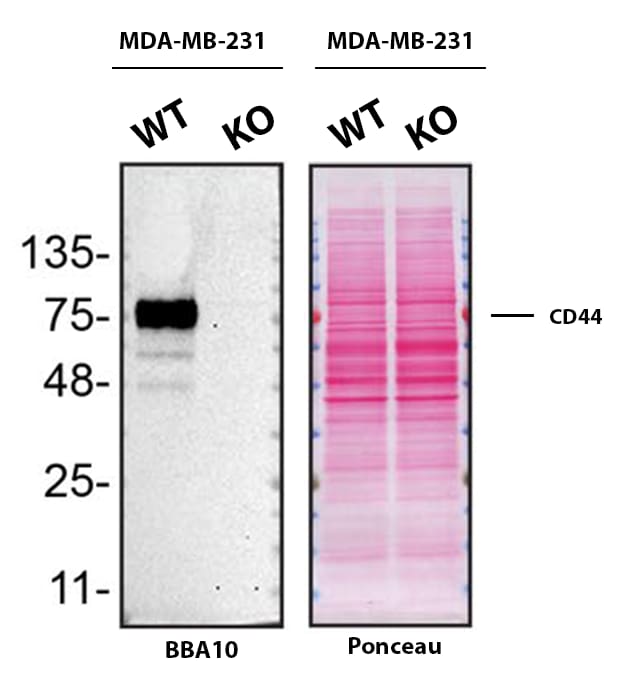
Western blot shows lysates of MDA‑MB‑231 human breast cancer parental cell line and CD44 knockout MDA-MB-231 cell line (KO). Nitrocellulose membrane was probed with Mouse Anti-Human CD44 s Pan Specific Monoclonal Antibody (Catalog # BBA10) followed by HRP-conjugated secondary antibody. A specific band was detected for CD44 at approximately 81.5 kDa (as indicated) in the parental MDA-MB-231 cell line, but is not detectable in knockout MDA-MB-231 cell line. Primary antibody concentration used: 2.5 µg/mL. The Ponceau stained transfer of the blot is shown. This experiment was conducted under reducing conditions. Image, protocol, and testing courtesy of YCharOS Inc. See ycharos.com for additional details.Detection of CD44 by Immunoprecipitation.
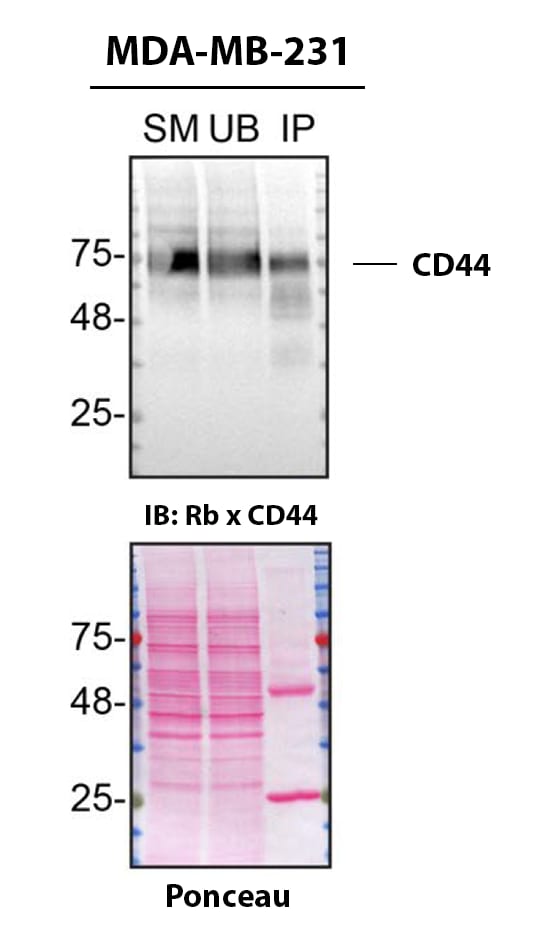
MDA‑MB‑231 human breast cancer cell line lysates were prepared and immunoprecipitation was performed using 2.0 μg of Mouse Anti-Human CD44 s Pan Specific Monoclonal Antibody (Catalog # BBA10) pre-coupled to Dynabeads Protein G. Immunoprecipitated CD44 was detected in Western Blot with a rabbit CD44 antibody used at 1/3000. The Ponceau stained transfer of the blot is shown. SM=4% starting material; UB=4% unbound fraction; IP=immunoprecipitate; HC=antibody heavy chain. Image, protocol and testing courtesy of YCharOS Inc. (ycharos.com).CD44 Specificity is Shown by Immunocytochemistry in Knockout Cell Line.
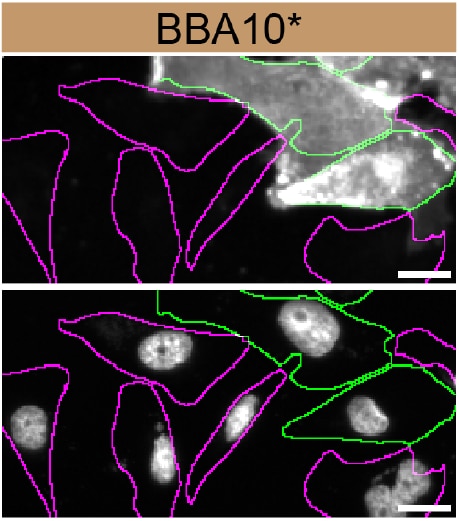
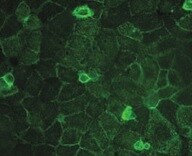
MDA‑MB‑231 human breast cancer parental cell line WT and CD44 MDA-MB-231 KO cells were labelled with a green or a far-red fluorescent dye, respectively. Cells were stained with Mouse Anti-Human CD44 s Pan Specific Monoclonal Antibody (Catalog # BBA10) followed by incubation with an Alexa-fluor 555 conjugated secondary antibody (upper panel). DAPI-only counterstained cells shown on a lower panel. Acquisition of the blue (nucleus-DAPI), green (identification of WT cells), red (antibody staining) and far-red (identification of KO cells) channels was performed. Representative images of the blue and red (grayscale) channels are shown. WT and KO cells are outlined with green and magenta dashed line, respectively. Primary antibody concentration used: 2 μg/mL. Image, protocol and testing courtesy of YCharOS Inc. (ycharos.com).Detection of CD44 by Flow Cytometry.
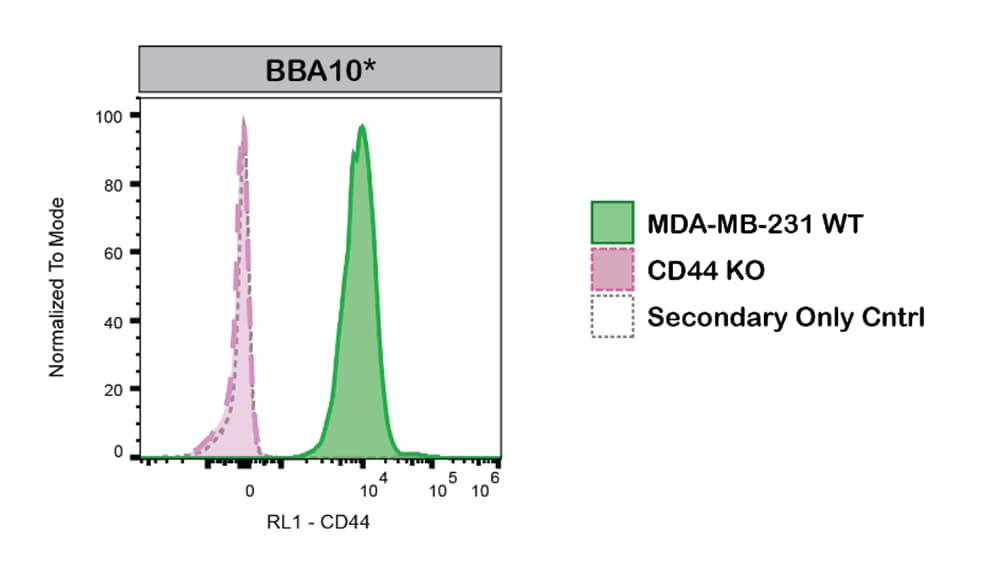
MDA‑MB‑231 human breast cancer parental cell line WT and CD44 KO cells were labelled with a green or violet, fluorescent dye, respectively. WT and KO cells were mixed in a 1:1 ratio, fixed in 4% PFA and permeabilized in 0.1% saponin. 400,000 cells were stained with Mouse Anti-Human CD44 s Pan Specific Monoclonal Antibody (Catalog # BBA10) and a secondary antibody. Antibody staining was quantified, and representative images showing the staining intensity in the KO population (pink histogram, dashed line) compared to the WT cells (green histogram, solid line) are presented. Histograms with dotted lines represent secondary antibody-only controls in both WT and KO cells. Primary antibody concentration: 1 μg/mL. Image, protocol and testing courtesy of YCharOS Inc. (ycharos.com).Detection of Human CD44 by Immunocytochemistry/Immunofluorescence
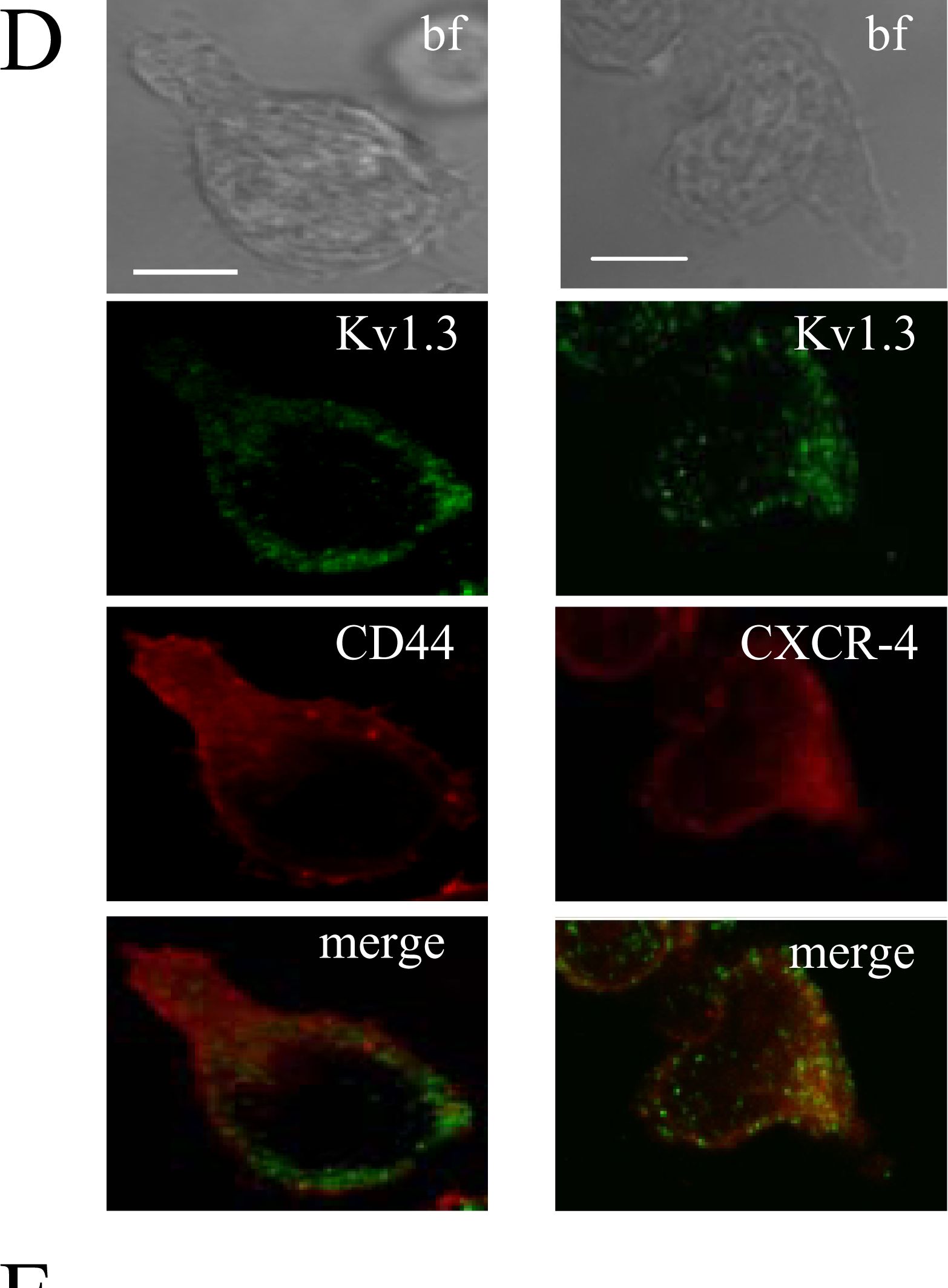
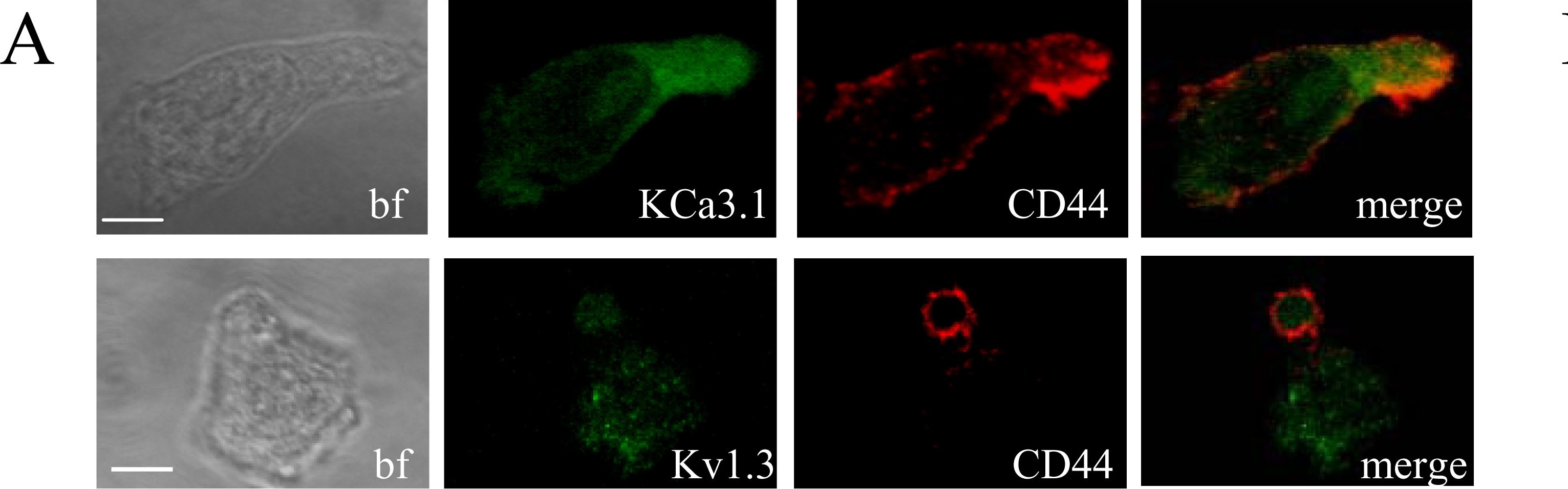
Differential localization of Kv1.3 and KCa3.1 in migrating T cells.A. Distribution of Kv1.3 and KCa3.1 at the uropod. T cells were transiently transfected with either YFP-KCa3.1 or GFP-Kv1.3 (green) and stained with anti-CD44 antibody (uropod; red) without permeabilization. Yellow areas in the merge images indicate colocalization. Scale bar = 5 µm. B. Distribution of Kv1.3 and KCa3.1 at the leading-edge. T cells, transfected with either YFP-KCa3.1 or GFP-Kv1.3 (green) and stained with anti-CXCR-4 antibody (leading-edge; red) without permeabilization, were analyzed by confocal microscopy. Colocalization between the two proteins is indicated by yellow areas in the merge images. Scale bar = 5 µm. C. Correlation coefficients for KCa3.1 and Kv1.3 localization in the uropod (U) and leading-edge (L). The data are the average of n = 15 cells for KCa3.1 at the U and n = 8 at the L, and n = 16 for Kv1.3 at the U and n = 11 at the U from 2 healthy individuals. Statistical significance was established by one way ANOVA. D. Localization of native Kv1.3 in the leading-edge. T cells from one healthy individual were fixed and stained with extracellular anti-Kv1.3 antibody (green) together with antibodies either against CD44 (red; left) or CXCR-4 (red, right). Yellow colors in the merge images indicate strong correlation. Scale bar = 5 µm. E. Average Correlation coefficients of native Kv1.3 with the leading-edge (L) (n = 9) and the uropod (U) markers (n = 11). Image collected and cropped by CiteAb from the following publication (https://dx.plos.org/10.1371/journal.pone.0043859), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human CD44 by Immunocytochemistry/Immunofluorescence
Differential localization of Kv1.3 and KCa3.1 in migrating T cells.A. Distribution of Kv1.3 and KCa3.1 at the uropod. T cells were transiently transfected with either YFP-KCa3.1 or GFP-Kv1.3 (green) and stained with anti-CD44 antibody (uropod; red) without permeabilization. Yellow areas in the merge images indicate colocalization. Scale bar = 5 µm. B. Distribution of Kv1.3 and KCa3.1 at the leading-edge. T cells, transfected with either YFP-KCa3.1 or GFP-Kv1.3 (green) and stained with anti-CXCR-4 antibody (leading-edge; red) without permeabilization, were analyzed by confocal microscopy. Colocalization between the two proteins is indicated by yellow areas in the merge images. Scale bar = 5 µm. C. Correlation coefficients for KCa3.1 and Kv1.3 localization in the uropod (U) and leading-edge (L). The data are the average of n = 15 cells for KCa3.1 at the U and n = 8 at the L, and n = 16 for Kv1.3 at the U and n = 11 at the U from 2 healthy individuals. Statistical significance was established by one way ANOVA. D. Localization of native Kv1.3 in the leading-edge. T cells from one healthy individual were fixed and stained with extracellular anti-Kv1.3 antibody (green) together with antibodies either against CD44 (red; left) or CXCR-4 (red, right). Yellow colors in the merge images indicate strong correlation. Scale bar = 5 µm. E. Average Correlation coefficients of native Kv1.3 with the leading-edge (L) (n = 9) and the uropod (U) markers (n = 11). Image collected and cropped by CiteAb from the following publication (https://dx.plos.org/10.1371/journal.pone.0043859), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of CD44s Pan Specific by Western Blot
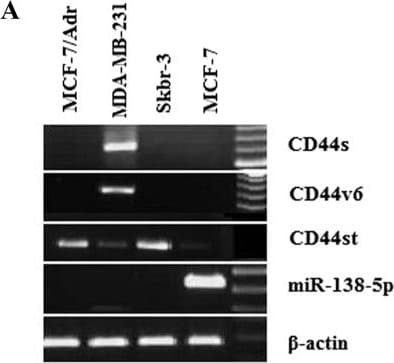
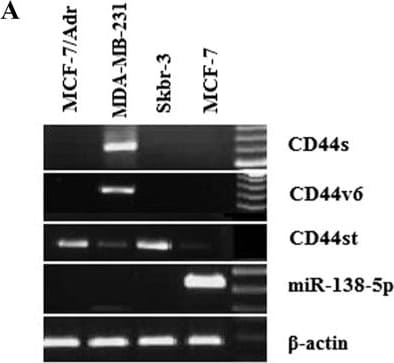
A-B Expression of CD44st, CD44s, CD44v6 mRNA and miR-138-5p in breast cancer cell lines. A Semi-quantitative PCR agarose electrophoresis plots of different cell lines B Quantitative PCR results of different cell lines Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/36964570), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of CD44s Pan Specific by Western Blot
A-B Expression of CD44st, CD44s, CD44v6 mRNA and miR-138-5p in breast cancer cell lines. A Semi-quantitative PCR agarose electrophoresis plots of different cell lines B Quantitative PCR results of different cell lines Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/36964570), licensed under a CC-BY license. Not internally tested by R&D Systems.Applications for Human CD44s Pan Specific Antibody
Application
Recommended Usage
CyTOF-ready
Ready to be labeled using established conjugation methods. No BSA or other carrier proteins that could interfere with conjugation.
Flow Cytometry
2.5 µg/106 cells
Sample: Human whole blood monocytes & MDA-MB-231 human breast cancer cell line lysates
Sample: Human whole blood monocytes & MDA-MB-231 human breast cancer cell line lysates
Immunohistochemistry
8-25 µg/mL
Sample: Immersion fixed paraffin-embedded sections of human lymphoma
Sample: Immersion fixed paraffin-embedded sections of human lymphoma
Immunoprecipitation
Fox, S.B. et al. (1994) Cancer Res. 54:4539.
Knockout Validated
CD44 is specifically detected in MDA-MB-231 parental cell line but is not detectable in knockout MDA-MB-231 cell line.
Simple Western
20 µg/mL
Sample: Human tonsil tissue
Sample: Human tonsil tissue
Western Blot
2 µg/mL
Sample: PC‑3 human prostate cancer cell line and human tonsil tissue
Sample: PC‑3 human prostate cancer cell line and human tonsil tissue
Reviewed Applications
Read 14 reviews rated 4.6 using BBA10 in the following applications:
Flow Cytometry Panel Builder
Bio-Techne Knows Flow Cytometry
Save time and reduce costly mistakes by quickly finding compatible reagents using the Panel Builder Tool.
Advanced Features
- Spectra Viewer - Custom analysis of spectra from multiple fluorochromes
- Spillover Popups - Visualize the spectra of individual fluorochromes
- Antigen Density Selector - Match fluorochrome brightness with antigen density
Formulation, Preparation, and Storage
Purification
Protein A or G purified from ascites
Reconstitution
Sterile PBS to a final concentration of 0.5 mg/mL.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose.
Shipping
The product is shipped at ambient temperature. Upon receipt, store it immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: CD44
References
- Ponta, H. et al. (2003) Nat. Rev. Mol. Cell Biol. 4:33.
- Screaton, G.R. et al. (1992) Proc. Natl. Acad. Sci. USA 89:12160.
- Lynch, K.W. (2004) Nat. Rev. Immunol. 4:931.
- Yu, Q. and B.P. Toole (1996) J. Biol. Chem. 271:20603.
- Nagano, O. and H. Saya (2004) Cancer Sci. 95:930.
- Nakamura, H. et al. (2004) Cancer Res. 64:876.
- Murakami, D. et al. (2003) Oncogene 22:1511.
- Lammich, S. et al. (2002) J. Biol. Chem. 277:44754.
Alternate Names
CD44, ECMR-III, HCAM, HCELL, LHR, MDU2, MDU3, MIC4, MUTCH-I, Pgp1
Gene Symbol
CD44
Additional CD44 Products
Product Documents for Human CD44s Pan Specific Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human CD44s Pan Specific Antibody
For research use only
Related Research Areas
Citations for Human CD44s Pan Specific Antibody
Customer Reviews for Human CD44s Pan Specific Antibody (14)
4.6 out of 5
14 Customer Ratings
Have you used Human CD44s Pan Specific Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
Showing
1
-
5 of
14 reviews
Showing All
Filter By:
-
Application: ImmunohistochemistrySample Tested: Neuroendocrine tumorSpecies: HumanVerified Customer | Posted 04/19/2022
-
Application: Immunocytochemistry/ImmunofluorescenceSample Tested: Prostate cellsSpecies: HumanVerified Customer | Posted 08/15/2021
-
Application: ELISASample Tested: Serum and PlasmaSpecies: HumanVerified Customer | Posted 06/12/2019
-
Application: ELISASample Tested: Serum and PlasmaSpecies: HumanVerified Customer | Posted 01/30/2019MAB7045 was used as the capture antibody along with BBA10 as the detection antibody. Recombinant human CD44 Fc Chimera (3660-CD) was used as the calibrator material. Human serum and plasma samples were diluted 1:5 and all were quantifiable. Parallelism looked good.
-
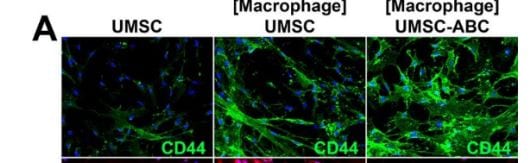
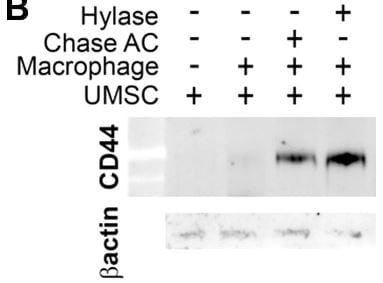
Application: IHC for UMUCSample Tested: Umbilical vein endothelial cellsSpecies: HumanVerified Customer | Posted 01/19/2019IHC for UMUC with one control 2 condition
-
Sample Tested: HUVEC human umbilical vein endothelial cellsSpecies: HumanVerified Customer | Posted 01/18/2019Chondroitiase treated increase CD44 when co-culture with macrophage
-
Application: ELISASample Tested: EDTA PlasmaSpecies: HumanVerified Customer | Posted 11/08/2018
-
Application: Western BlotSample Tested: A549 human lung carcinoma cell lineSpecies: HumanVerified Customer | Posted 06/28/2017
-
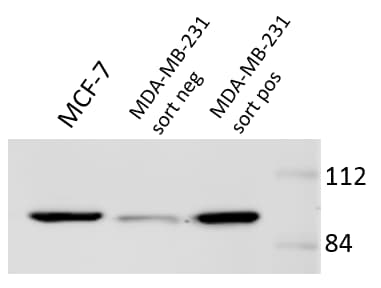
Application: Western BlotSample Tested: Cell LysatesSpecies: HumanVerified Customer | Posted 12/04/2016Protein expression confirms sorting results. Antibody is sensitive and reliable across multiple cell lines.
-
Application: Western BlotSample Tested: See PMID: 21364036Species: HumanVerified Customer | Posted 01/16/2015
-
Application: Western BlotSample Tested: See PMID: 21730156Species: HumanVerified Customer | Posted 01/16/2015
-
Application: Western BlotSample Tested: See PMID: 23736004Species: HumanVerified Customer | Posted 01/16/2015
-
Application: Western BlotSample Tested: See PMID: 23457460Species: HumanVerified Customer | Posted 01/16/2015
-
Application: Western BlotSample Tested: See PMID: 24512624Species: HumanVerified Customer | Posted 01/16/2015
There are no reviews that match your criteria.
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- 7-Amino Actinomycin D (7-AAD) Cell Viability Flow Cytometry Protocol
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Extracellular Membrane Flow Cytometry Protocol
- Flow Cytometry Protocol for Cell Surface Markers
- Flow Cytometry Protocol for Staining Membrane Associated Proteins
- Flow Cytometry Staining Protocols
- Flow Cytometry Troubleshooting Guide
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- IHC Sample Preparation (Frozen sections vs Paraffin)
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Immunoprecipitation Protocol
- Intracellular Flow Cytometry Protocol Using Alcohol (Methanol)
- Intracellular Flow Cytometry Protocol Using Detergents
- Intracellular Nuclear Staining Flow Cytometry Protocol Using Detergents
- Intracellular Staining Flow Cytometry Protocol Using Alcohol Permeabilization
- Intracellular Staining Flow Cytometry Protocol Using Detergents to Permeabilize Cells
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Propidium Iodide Cell Viability Flow Cytometry Protocol
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Liperfluo
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Characterization of Human Th22 Cells
- Protocol for the Characterization of Human Th9 Cells
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- Protocol: Annexin V and PI Staining by Flow Cytometry
- Protocol: Annexin V and PI Staining for Apoptosis by Flow Cytometry
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Fluorokine Flow Cytometry Kits
- Troubleshooting Guide: Immunohistochemistry
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...
Associated Pathways
 Hematopoietic Stem Cell Differentiation Pathways & Lineage-specific Markers
Hematopoietic Stem Cell Differentiation Pathways & Lineage-specific Markers
 Innate Lymphoid Cell Differentiation Pathways
Innate Lymphoid Cell Differentiation Pathways
 Mesenchymal Stem Cell Differentiation Pathways & Lineage-specific Markers
Mesenchymal Stem Cell Differentiation Pathways & Lineage-specific Markers
 Neural Stem Cell Differentiation Pathways & Lineage-specific Markers
Neural Stem Cell Differentiation Pathways & Lineage-specific Markers