The type I class A macrophage scavenger receptor (SR-AI; also MSR-AI) is a 70-80 kDa protein that belongs to an ever expanding family of transmembrane molecules collectively referred to as scavenger receptors (1-3). Receptors of this family contain characteristic extracellular domains and bind to a series of generally unrelated, but negatively-charged/polyanionic ligands (1, 3). Human SR-AI is a type II transmembrane glycoprotein that is 451 amino acids (aa) in length. It contains a 50 aa cytoplasmic tail, a 26 aa transmembrane segment and a 375 aa extracellular region (4, 5). The extracellular region contains four definitive domains, with a membrane proximal spacer of 33 aa, an alpha -helical coiled-coil domain of 163 aa, a collagen-like domain of 69 aa, and a cysteine-rich C-terminus of 110 aa (4, 6). The cysteine-rich domain (CRD) forms three intrachain disulfide bonds (7). The functional form of the molecule is a 220-230 kDa membrane-associated trimer that, in human, apparently has two disulfide bonded chains and a third noncovalently associated subunit (8, 9). Human extracellular region is 73% and 72% aa identical to bovine and mouse SR-AI extracellular region, respectively. The human gene for SR-A gives rise to three isoforms; the I isoform of 451 aa, the II isoform of 358 aa, and the III isoform of 388 aa (4, 5, 10). All are equal through the first 344 aa which includes the cytoplasmic tail through the collagenous domain. Isoform II (SR-AII) shows a severe truncation of the CRD, but is expressed on the cell surface. Isoform III (SR-AIII) has a modest truncation of the CRD, and cannot be expressed on the cell surface. Their functions are unknown. However, relative to SR-AI, SR-AII is known to show differential sensitivity to LPS and receptor binding to gram-negative bacteria (9, 11), while SR-AIII is known to be a dominant-negative isoform (10). SR-AIII may achieve this by either heterotrimerizing with SR-AI, or simply eliminating the production of SR-AI mRNA.
Human SR‑AI/MSR Antibody
R&D Systems | Catalog # AF2708

Key Product Details
Validated by
Biological Validation
Species Reactivity
Validated:
Human
Cited:
Human
Applications
Validated:
Immunohistochemistry, Western Blot, Blockade of Receptor-ligand Interaction, Immunocytochemistry
Cited:
Western Blot, Immunoprecipitation
Label
Unconjugated
Antibody Source
Polyclonal Goat IgG
Loading...
Product Specifications
Immunogen
Mouse myeloma cell line NS0-derived recombinant human SR-AI/MSR type A isoform 1
Lys77-Leu451
Accession # P21757
Lys77-Leu451
Accession # P21757
Specificity
Detects human SR-AI/MSR in direct ELISAs and Western blots. In direct ELISAs, approximately 25% cross-reactivity with recombinant mouse SR-AI is observed.
Clonality
Polyclonal
Host
Goat
Isotype
IgG
Endotoxin Level
<0.10 EU per 1 μg of the antibody by the LAL method.
Scientific Data Images for Human SR‑AI/MSR Antibody
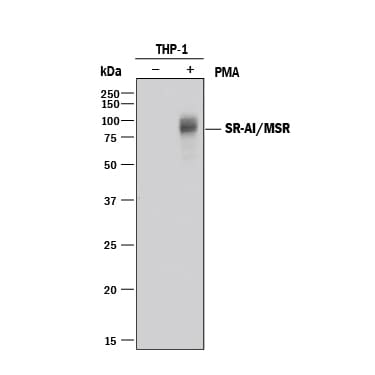
Detection of Human SR‑AI/MSR by Western Blot.
Western blot shows lysates of THP-1 human acute monocytic leukemia cell line untreated (-) or treated (+) with PMA. PVDF membrane was probed with 0.2 µg/mL of Goat Anti-Human SR-AI/MSR Antigen Affinity-purified Polyclonal Antibody (Catalog # AF2708) followed by HRP-conjugated Anti-Goat IgG Secondary Antibody (Catalog # HAF017). A specific band was detected for SR-AI/MSR at approximately 80 kDa (as indicated). This experiment was conducted under reducing conditions and using Immunoblot Buffer Group 1.SR‑AI/MSR in THP-1 Human Cell Line.
SR-AI/MSR was detected in immersion fixed THP-1 human acute monocytic leukemia cell line untreated (right panel) and treated with PMA (left panel) using Goat Anti-Human SR-AI/MSR Antigen Affinity-purified Polyclonal Antibody (Catalog # AF2708) at 1.7 µg/mL for 3 hours at room temperature. Cells were stained using the NorthernLights™ 557-conjugated Anti-Goat IgG Secondary Antibody (red; Catalog # NL001) and counterstained with DAPI (blue). Specific staining was localized to cytoplasm. View our protocol for Fluorescent ICC Staining of Non-adherent Cells.SR-AI/MSR in Human Lung Tissue.
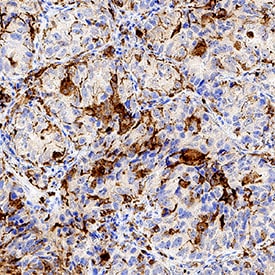
SR-AI/MSR was detected in immersion fixed paraffin-embedded sections of human lung tissue using Goat Anti-Human SR-AI/MSR Antigen Affinity-purified Polyclonal Antibody (Catalog # AF2708) at 1 µg/mL for 1 hour at room temperature followed by incubation with the Anti-Goat IgG VisUCyte™ HRP Polymer Antibody (Catalog # VC004). Tissue was stained using DAB (brown) and counterstained with hematoxylin (blue). Specific staining was localized to macrophages. View our protocol for IHC Staining with VisUCyte HRP Polymer Detection Reagents.Detection of Human SR-AI/MSR by Immunohistochemistry
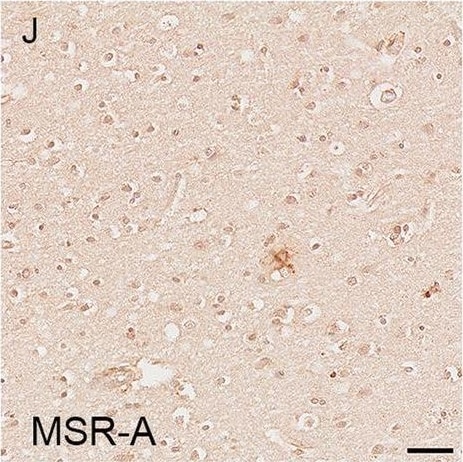
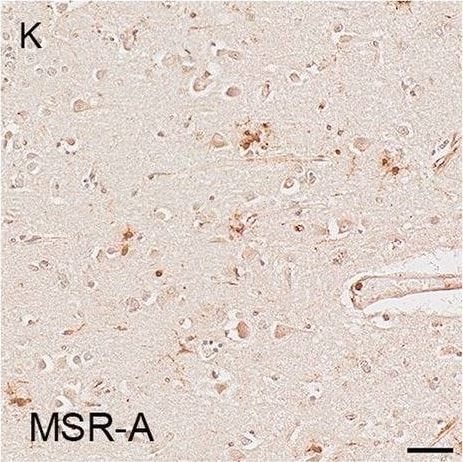
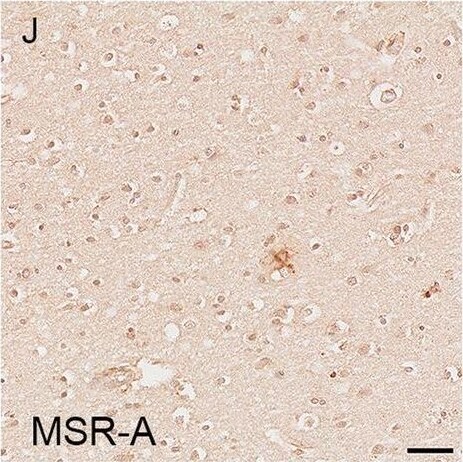
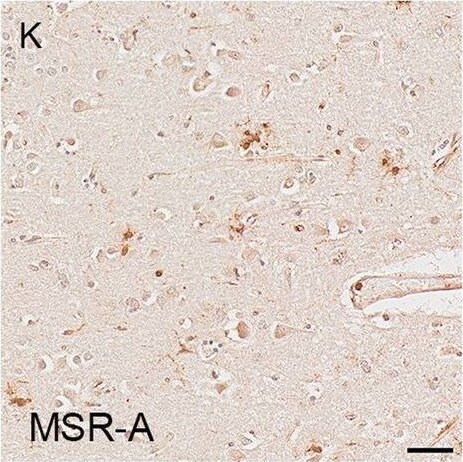
Illustrations and quantification in the temporal lobe (TL) of the immunolabelling expressed as protein load (%) for the microglial markers TSPO (A–C), Iba1 (D–F), HLA–DR (G–I) and MSR-A (J–L). A significant increase with Braak stage was seen for TSPO load (P < 0.0001), while no difference between Braak stages was detected for the other microglial markers. Counterstaining: Haematoxylin. Scale bars = 50μm Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/37580767), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human SR-AI/MSR by Immunohistochemistry
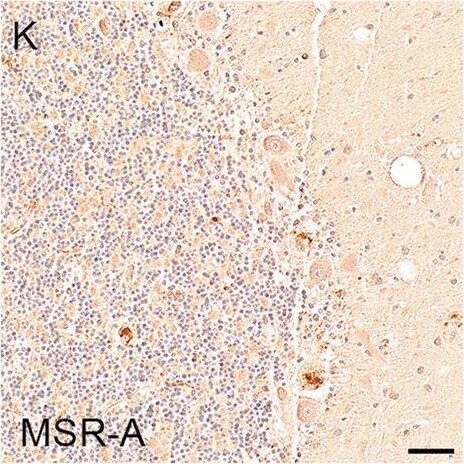
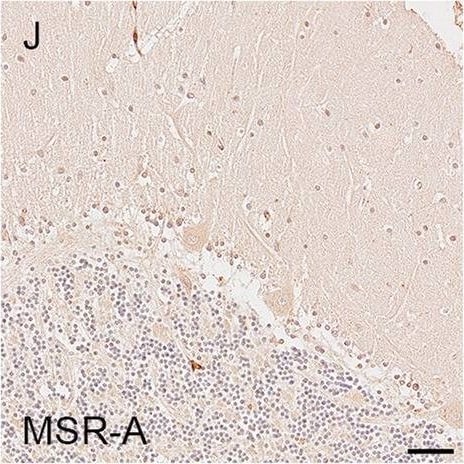
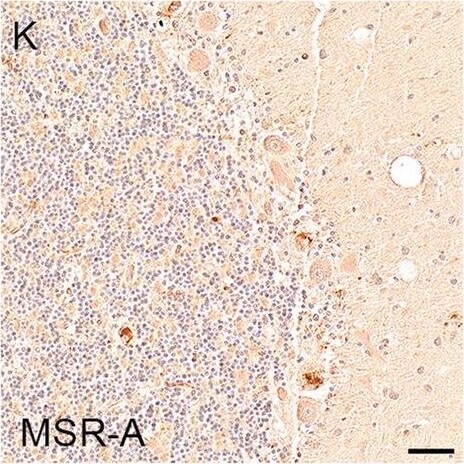
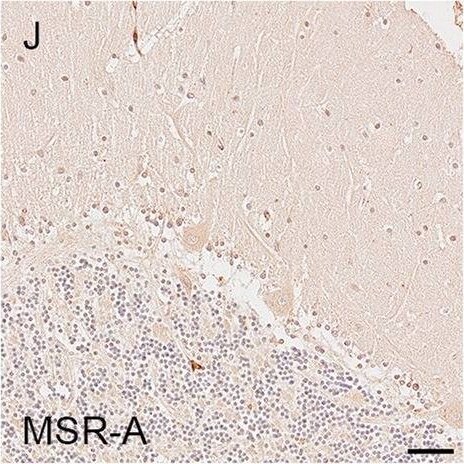
Illustrations and quantification in the cerebellar cortex (Cb) of immunostaining expressed as protein load (%) for the microglial markers TSPO (A–C), Iba1 (D–F), HLA–DR (G–I) and MSR-A (J–L). A significant increase with Braak stage was seen for Iba1 load (P = 0.012). Counterstaining: Haematoxylin. Scale bars = 50μm Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/37580767), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human SR-AI/MSR by Immunohistochemistry
Illustrations and quantification in the temporal lobe (TL) of the immunolabelling expressed as protein load (%) for the microglial markers TSPO (A–C), Iba1 (D–F), HLA–DR (G–I) and MSR-A (J–L). A significant increase with Braak stage was seen for TSPO load (P < 0.0001), while no difference between Braak stages was detected for the other microglial markers. Counterstaining: Haematoxylin. Scale bars = 50μm Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/37580767), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human SR-AI/MSR by Immunohistochemistry
Illustrations and quantification in the temporal lobe (TL) of the immunolabelling expressed as protein load (%) for the microglial markers TSPO (A–C), Iba1 (D–F), HLA–DR (G–I) and MSR-A (J–L). A significant increase with Braak stage was seen for TSPO load (P < 0.0001), while no difference between Braak stages was detected for the other microglial markers. Counterstaining: Haematoxylin. Scale bars = 50μm Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/37580767), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human SR-AI/MSR by Immunohistochemistry
Illustrations and quantification in the cerebellar cortex (Cb) of immunostaining expressed as protein load (%) for the microglial markers TSPO (A–C), Iba1 (D–F), HLA–DR (G–I) and MSR-A (J–L). A significant increase with Braak stage was seen for Iba1 load (P = 0.012). Counterstaining: Haematoxylin. Scale bars = 50μm Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/37580767), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human SR-AI/MSR by Immunohistochemistry
Illustrations and quantification in the cerebellar cortex (Cb) of immunostaining expressed as protein load (%) for the microglial markers TSPO (A–C), Iba1 (D–F), HLA–DR (G–I) and MSR-A (J–L). A significant increase with Braak stage was seen for Iba1 load (P = 0.012). Counterstaining: Haematoxylin. Scale bars = 50μm Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/37580767), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human SR-AI/MSR by Immunohistochemistry
Illustrations and quantification in the cerebellar cortex (Cb) of immunostaining expressed as protein load (%) for the microglial markers TSPO (A–C), Iba1 (D–F), HLA–DR (G–I) and MSR-A (J–L). A significant increase with Braak stage was seen for Iba1 load (P = 0.012). Counterstaining: Haematoxylin. Scale bars = 50μm Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/37580767), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human SR-AI/MSR by Immunohistochemistry
Illustrations and quantification in the temporal lobe (TL) of the immunolabelling expressed as protein load (%) for the microglial markers TSPO (A–C), Iba1 (D–F), HLA–DR (G–I) and MSR-A (J–L). A significant increase with Braak stage was seen for TSPO load (P < 0.0001), while no difference between Braak stages was detected for the other microglial markers. Counterstaining: Haematoxylin. Scale bars = 50μm Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/37580767), licensed under a CC-BY license. Not internally tested by R&D Systems.Applications for Human SR‑AI/MSR Antibody
Application
Recommended Usage
Blockade of Receptor-ligand Interaction
In a functional ELISA, 0.5-2 µg/mL of this antibody will block 50% of the binding of 400 ng/mL of biotinylated AGE-BSA to immobilized Recombinant Human SR-AI/MSR (Catalog # 2708-MS) coated at 5 µg/mL (100 µL/well). At 20 μg/mL, this antibody will block >90% of the binding.
Immunocytochemistry
1-15 µg/mL
Sample: Immersion fixed THP‑1 human acute monocytic leukemia cell line
Sample: Immersion fixed THP‑1 human acute monocytic leukemia cell line
Immunohistochemistry
1-15 µg/mL
Sample: Immersion fixed paraffin-embedded sections of human lung tissue
Sample: Immersion fixed paraffin-embedded sections of human lung tissue
Western Blot
0.2 µg/mL
Sample: THP‑1 human acute monocytic leukemia cell line treated with PMA
Sample: THP‑1 human acute monocytic leukemia cell line treated with PMA
Formulation, Preparation, and Storage
Purification
Antigen Affinity-purified
Reconstitution
Reconstitute at 0.2 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: SR-AI/MSR
References
- Platt, N. and S. Gordon (2001) J. Clin. Invest. 108:649.
- Linton, M.F. and S. Fazio (2001) Curr. Opin. Lipidol. 12:489.
- Platt, N. and S. Gordon (1998) Chem. Biol. 5:R193.
- Matsumoto, A. et al. (1990) Proc. Natl. Acad. Sci. USA 87:9133.
- Emi, M. et al. (1993) J. Biol. Chem. 268:2120.
- Naito, M. et al. (1992) Am. J. Pathol. 141:591.
- Resnick, D. et al. (1996) J. Biol. Chem. 271:26924.
- Ashkenas, J. et al. (1993) J. Lipid Res. 34:983.
- Penman, M. et al. (1991) J. Biol. Chem. 266:23985.
- Gough, P.J. et al. (1998) J. Lipid Res. 39:531.
- Peiser, L. et al. (2000) Inf. Immun. 68:1953.
Long Name
Macrophage Scavenger Receptor Types I and II
Alternate Names
CD204, MSR1, SCARA1, SRAI
Gene Symbol
MSR1
UniProt
Additional SR-AI/MSR Products
Product Documents for Human SR‑AI/MSR Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human SR‑AI/MSR Antibody
For research use only
Related Research Areas
Citations for Human SR‑AI/MSR Antibody
Customer Reviews for Human SR‑AI/MSR Antibody
There are currently no reviews for this product. Be the first to review Human SR‑AI/MSR Antibody and earn rewards!
Have you used Human SR‑AI/MSR Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- ICC Cell Smear Protocol for Suspension Cells
- ICC Immunocytochemistry Protocol Videos
- ICC for Adherent Cells
- IHC Sample Preparation (Frozen sections vs Paraffin)
- Immunocytochemistry (ICC) Protocol
- Immunocytochemistry Troubleshooting
- Immunofluorescence of Organoids Embedded in Cultrex Basement Membrane Extract
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Fluorescent ICC Staining of Cell Smears - Graphic
- Protocol for the Fluorescent ICC Staining of Cultured Cells on Coverslips - Graphic
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent ICC Staining of Cells on Coverslips
- Protocol for the Preparation and Fluorescent ICC Staining of Non-adherent Cells
- Protocol for the Preparation and Fluorescent ICC Staining of Stem Cells on Coverslips
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- Protocol for the Preparation of a Cell Smear for Non-adherent Cell ICC - Graphic
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Immunohistochemistry
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...
Associated Pathways