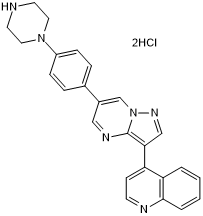
LDN 193189 dihydrochloride
Tocris Bioscience | Catalog # 6053

Key Product Details
Description
Product Description
LDN 193189 dihydrochloride is a potent and selective ALK2 and ALK3 inhibitor (IC50 values are 5 and 30 nM, respectively); inhibits BMP4-mediated Smad1/5/8 activation. Exhibits >200-fold selectivity for BMP signaling over TGF-β signaling. Also exhibits selectivity over AMPK, PDGFR and MAPK signaling. Promotes neural induction of hPSCs in combination with SB 431542 (Cat.No. 1614). Also induces differentiation of hPSCs into nociceptive sensory neurons in combination with SB 431542 (Cat.No. 1614), SU 5402 (Cat.No.3300), CHIR 99021 (Cat.No. 4423) and DAPT (Cat.No. 2634). Used in protocols to generate pancreatic beta cells, or midbrain dopaminergic neurons from hPSCs (see below).LDN 193189 synthesized to cGMP guidelines and LDN 193189 in solution also available.
For more information about how LDN 193189 dihydrochloride may be used, see our protocols: Generation of β cells from hPSCs, Generating Midbrain Dopaminergic Neurons from hPSCs, Accelerated Induction of Cortical Neurons from hiPSCs.
Scientific Data Images for LDN 193189 dihydrochloride
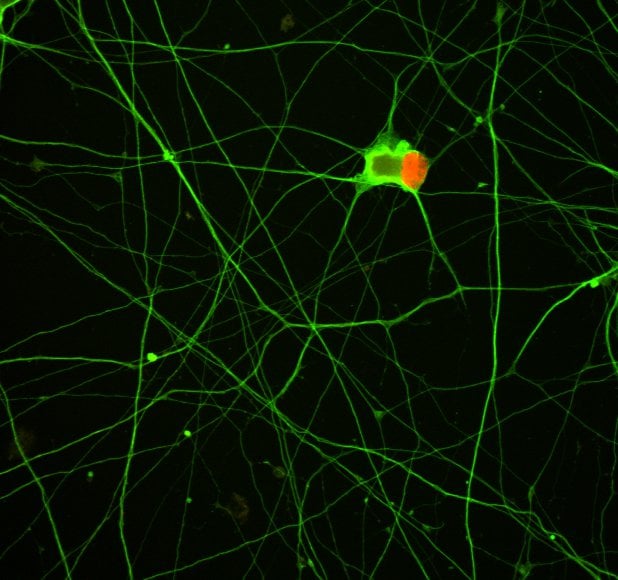
Application of LDN 193189 in motor neurons differentiated from iPSCs.
Image shows IBJ6 human fibroblast induced pluripotent stem cells which have been differentiated into motor neurons. This was performed using a combination of LDN 193189 (100 nM) and SB 431542 (Catalog # 1614, Tocris) followed by Retinoic acid (Catalog # 0695, Tocris) and Purmorphamine (Catalog # 4551, Tocris) culture on Laminin for 18 days. Commitment to the motor neuron fate was assessed using the motor neuron marker anti-Human Islet-1 (red, R&D Systems, Catalog # AF1837) and the general neuronal marker, beta III tubulin (green, R&D Systems, Catalog # MAB1195). For visualization, the neurons were stained using Northernlights™ 557-conjugated Donkey anti-Goat Secondary Antibody (R&D Systems, Catalog # NL001) and Northernlights™ 493-conjugated Donkey anti-Mouse Secondary Antibody (R&D Systems, Catalog # NL009 NL009) and counterstained with DAPI (blue, Catalog # 5748, Tocris).Licensing Information
Sold for research purposes under exclusive agreement from The Brigham and Women's Hospital Inc. US patents 8,507,501 and 9,045,484
Product Specifications for LDN 193189 dihydrochloride
Molecular Weight
Formula
Storage
Purity
Chemical Name
CAS Number
PubChem ID
InChI Key
SMILES
The technical data provided above is for guidance only. For batch specific data refer to the Certificate of Analysis.
Solubility
| Solvent | Max Conc. mg/mL | Max Conc. mM | |
|---|---|---|---|
| Solubility | |||
| water | 23.97 | 50 | |
| DMSO | 4.79 | 10 |
Preparing Stock Solutions for LDN 193189 dihydrochloride
The following data is based on the product molecular weight 479.40.
Batch specific molecular weights may vary from batch to batch due to the degree of hydration, which all affect the solvent volumes required to prepare stock solutions.
| Concentration / Solvent Volume / Mass | 1 mg | 5 mg | 10 mg |
|---|---|---|---|
| 0.5 mM | 4.17 mL | 20.86 mL | 41.72 mL |
| 2.5 mM | 0.83 mL | 4.17 mL | 8.34 mL |
| 5 mM | 0.42 mL | 2.09 mL | 4.17 mL |
| 25 mM | 0.08 mL | 0.42 mL | 0.83 mL |
Calculators
Background References
References are publications that support the biological activity of the product. See our Citations tab to view 131 publications citing the usage of this product.
- Wimmer Human blood vessel organoids as a model of diabetic vasculopathy. Nature 2019 PMID: 30651639
- Kadoshima Self-organization of axial polarity, inside-out layer pattern, and species-specific progenitor dynamics in human ES cell-derived neocortex. Proc.Natl.Acad.Sci.USA. 2013 PMID: 24277810
- Dye In vitro generation of human pluripotent stem cell derived lung organoids. Elife 2015 PMID: 25803487
- Lancaster Generation of Cerebral Organoids from Human Pluripotent Stem Cells Nat. Protoc. 2015 PMID: 25188634
- Li Chemical approaches to stem cell biology and therapeutics. Cell Stem Cell 2013 PMID: 24012368
- Cuny Structure-activity relationship study of bone morphogenetic protein (BMP) signaling inhibitors. Bioorg.Med.Chem.Lett. 2008 PMID: 18621530
- Chambers Combined small-molecule inhibition accelerates developmental timing and converts human pluripotent stem cells into nociceptors. Nat.Biotechnol. 2012 PMID: 22750882
- Yu BMP type I receptor inhibition reduces heterotopic ossification. Nat.Med. 2008 PMID: 19029982
Product Documents for LDN 193189 dihydrochloride
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Product Specific Notices for LDN 193189 dihydrochloride
For research use only
Related Research Areas
Citations for LDN 193189 dihydrochloride
Customer Reviews for LDN 193189 dihydrochloride (3)
Have you used LDN 193189 dihydrochloride?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
-
Species: HumanAssay Type: In VitroCell Line/Tissue: TC28a2Verified Customer | Posted 12/11/2020Be wary of storage as activity will degrade over time.To perform inhibition of BMP-like receptors during various interrogations. Performed blanket inhibition of BMP-like receptor kinase activity with 50nM (working) and selective inhibition of ALK3/BMPR1A with 5nM (working) leaving ALK6/BMPR1B active. Non-toxic at concentrations used.
-
Species: HumanCell Line/Tissue: Induced pluripotent stem cellsVerified Customer | Posted 12/11/2020I used the product for 10 days to differentiate ipscs into a neuroepitheliumBio-Techne ResponseThank you for reviewing our product. We are sorry to hear that this compound did not perform as expected. We have been in touch with the customer to resolve this issue according to our Product Guarantee and to the customer’s satisfaction.
-
Species: HumanAssay Type: In VitroCell Line/Tissue: Breast CancerVerified Customer | Posted 12/09/2019I used it as a BMP pathway inhibitor in Breast cancer Cells. I used the concentration of 100nM and it works really well.
There are no reviews that match your criteria.