Interleukin 2 was initially identified as a T cell growth factor that is produced by T cells following activation by mitogens or antigens (1). IL-2 has since been found to also stimulate the growth and differentiation of B cells, natural killer (NK) cells, lymphocyte activated killer (LAK) cells, monocytes/macrophages and oligodendrocytes (2).
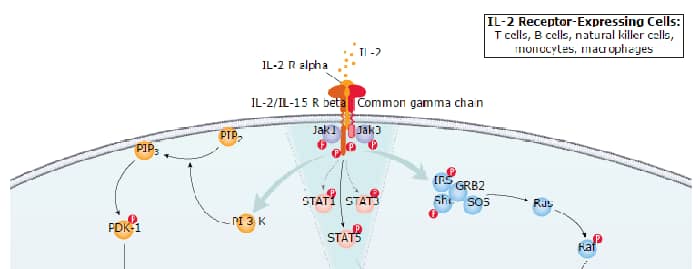
The biological activity of IL-2 is mediated by the binding of IL-2 to cell surface receptor complexes. The functional high-affinity receptor that mediate IL-2 signals is composed of three polypeptide chains, the IL-2 receptor alpha, beta and gamma subunits (3). IL-2 also signals via the intermediate affinity receptor complex of the beta and gamma subunits (4). In T cells, the beta and gamma subunits are shared with the IL-15 receptor complex (5). The gamma subunit of the IL-2 receptor complex has also been shown to be a subunit of the receptor complexes of IL-4, IL-7, IL-9 and IL-21 (6).
At the amino acid sequence level, equine IL-2 shares 72%, 70%, 56% and 54% sequence similarities with human, porcine, rat and mouse IL-2, respectively. It has been reported that equine IL-2 augmented proliferation in equine peripheral blood mononuclear cells, but has no effect on mouse CTLL-2 cells (7).