Human HNF‑3 beta /FoxA2 Antibody
R&D Systems | Catalog # AF2400

Key Product Details
Validated by
Biological Validation
Species Reactivity
Validated:
Human
Cited:
Human, Mouse, Rat, Chimpanzee, Primate - Macaca mulatta (Rhesus Macaque), Transgenic Rat, Xenograft
Applications
Validated:
Immunohistochemistry, Western Blot, Immunocytochemistry, Chromatin Immunoprecipitation (ChIP)
Cited:
Immunohistochemistry, Immunohistochemistry-Paraffin, Immunohistochemistry-Frozen, Western Blot, Flow Cytometry, Immunocytochemistry, Immunoprecipitation, Chromatin Immunoprecipitation (ChIP), Chromatin Immunoprecipitation Sequencing, CyTof
Label
Unconjugated
Antibody Source
Polyclonal Goat IgG
Loading...
Product Specifications
Immunogen
E. coli-derived recombinant human HNF‑3 beta /FoxA2
Met242-Ser457
Accession # Q9Y261
Met242-Ser457
Accession # Q9Y261
Specificity
Detects human HNF‑3 beta /FoxA2 in direct ELISAs and Western blots.
Clonality
Polyclonal
Host
Goat
Isotype
IgG
Scientific Data Images for Human HNF‑3 beta /FoxA2 Antibody
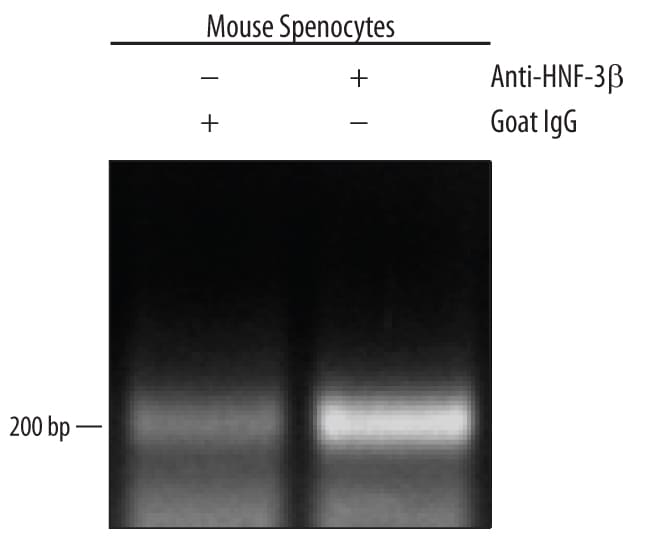
Detection of HNF‑3 beta /FoxA2-regulated Genes by Chromatin Immunoprecipitation.
Mouse splenocytes were fixed using formaldehyde, resuspended in lysis buffer, and sonicated to shear chromatin. HNF-3 beta /FoxA2/DNA complexes were immunoprecipitated using 5 µg Goat Anti-Human HNF-3 beta /FoxA2 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF2400) or control antibody (AB-108-C) for 15 minutes in an ultrasonic bath, followed by Biotinylated Anti-Goat IgG Secondary Antibody (BAF109). Immunocomplexes were captured using 50 µL of MagCellect Streptavidin Ferrofluid (MAG999) and DNA was purified using chelating resin solution. TheE-RABPpromoter was detected by standard PCR.HNF‑3 beta /FoxA2 in Endoderm Differentiated BG01V Human Stem Cells.
HNF-3 beta /FoxA2 was detected in immersion fixed endoderm differentiated BG01V human embryonic stem cells using 10 µg/mL Goat Anti-Human HNF-3 beta /FoxA2 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF2400) for 3 hours at room temperature. Cells were stained with the NorthernLights™ 557-conjugated Anti-Goat IgG Secondary Antibody (red; NL001) and counterstained with DAPI (blue). View our protocol for Fluorescent ICC Staining of Cells on Coverslips.HNF‑3 beta /FoxA2 in Human Liver.
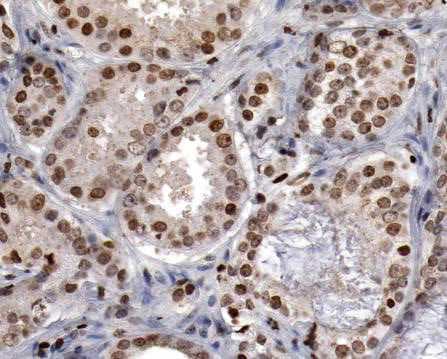
HNF-3 beta /FoxA2 was detected in immersion fixed paraffin-embedded sections of human liver using Goat Anti-Human HNF-3 beta /FoxA2 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF2400) at 3 µg/mL for 1 hour at room temperature followed by incubation with the Anti-Goat IgG VisUCyte™ HRP Polymer Antibody (VC004). Before incubation with the primary antibody, tissue was subjected to heat-induced epitope retrieval using Antigen Retrieval Reagent-Basic (CTS013). Tissue was stained using DAB (brown) and counterstained with hematoxylin (blue). Specific staining was localized to cytoplasm and nuclei. View our protocol for IHC Staining with VisUCyte HRP Polymer Detection Reagents.Detection of HNF‑3 beta /FoxA2 in HepG2 Cells (Positive) and THP‑1 Cells (Negative).
HNF‑3 beta /FoxA2 was detected in immersion fixed HepG2 Human Hepatocellular Carcinoma Cells (Positive) and absent in THP‑1 Human Acute Monocytic Leukemia Cells (Negative) using Goat Anti-Human HNF‑3 beta /FoxA2 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF2400) at 5 µg/mL for 3 hours at room temperature. Cells were stained using the NorthernLights™ 557-conjugated Anti-Goat IgG Secondary Antibody (red; Catalog # NL001) and counterstained with DAPI (blue). Specific staining was localized to cell nuclei. View our protocol for Fluorescent ICC Staining of Cells on Coverslips.Detection of Human HNF‑3 beta /FoxA2 by Western Blot.
Western blot shows lysates of HepG2 human hepatocellular carcinoma cells. PVDF membrane was probed with 1 µg/mL of Goat Anti-Human HNF‑3 beta /FoxA2 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF2400) followed by HRP-conjugated Anti-Goat IgG Secondary Antibody (Catalog # HAF017). A specific band was detected for HNF‑3 beta /FoxA2 at approximately 50 kDa (as indicated). This experiment was conducted under reducing conditions and using Western Blot Buffer Group 1.Detection of Human HNF-3 beta/FoxA2 by Immunocytochemistry/Immunofluorescence
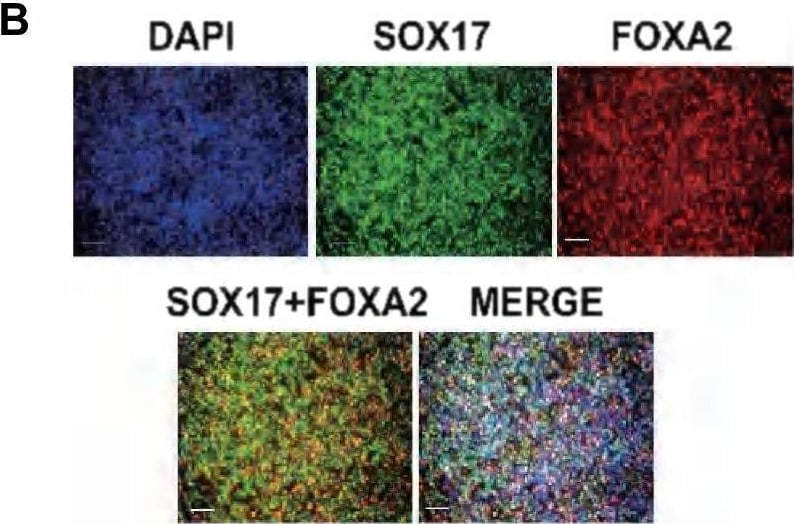
Differentiation of hESCs into definitive endoderm cells by Activin A with CHIR99021.a The relative primitive streak (BRACHYRUY, MESP1, and MIXL1) gene expression of the day 1 differentiated cells by adding Activin A (100 ng/mL) with CHIR99021 (2–5 µM) or Wnt3a (25–100 ng/mL) treatments were determined by real-time quantitative PCR (qPCR). None, no WNT signaling pathway activators were used on day 1 for PS differentiation (Activin A only). b, c After 1 day, the medium was changed to Activin A (100 ng/mL) and 1 × CTS-B27 to induce DE differentiation. The definitive endoderm (FOXA2 and SOX17) and mesoderm (MESP2 and HAND1) relative gene expression levels were determined by qPCR. d Immunofluorescence analysis of the expression of SOX17 and FoxA2 for Activin A with 2 µM CHIR99021-induced differentiated cells on day 3. e, f The expression of SOX17 and FoxA2 for Activin A with 2 µM CHIR99021-induced differentiated cells was determined by flow cytometry on day 3. Isotype control antibodies were used as controls. At a specific gene expression, datum points lacking common letters differ, p < 0.05. Data are represented as the mean ± SD. Scale bar, 100 µm Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/31601782), licensed under a CC-BY license. Not internally tested by R&D Systems.Immunofluorescent Staining of Adult Stem Cell-derived Liver Organoids.
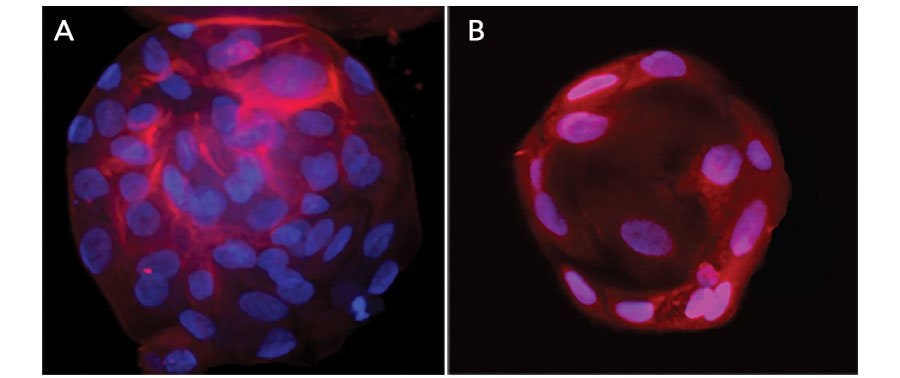
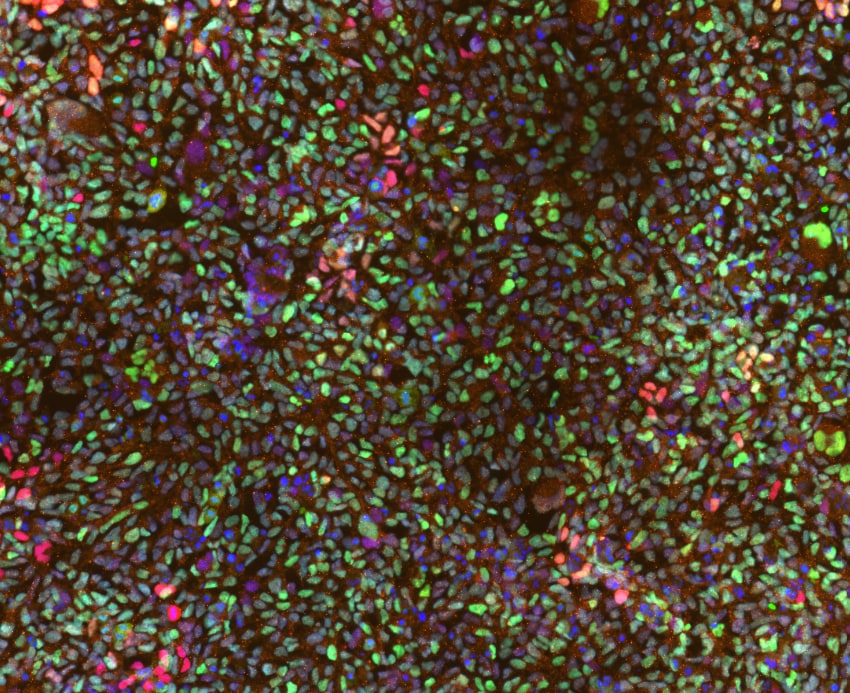
Adult stem cell-derived liver organoids were generated following the steps detailed in the human liver organoid culture protocol. Differentiated human liver organoids were stained using a (A) Mouse Anti-Human Serum Albumin Monoclonal Antibody (Catalog # MAB1455; red) and a (B) Goat Anti-Human HNF-3beta Antigen Affinity-purified Polyclonal Antibody (Catalog # AF2400; red) and counterstained with DAPI (Catalog # 5748; blue).Detection of HNF-3 beta /FoxA2 by Flow Cytometry
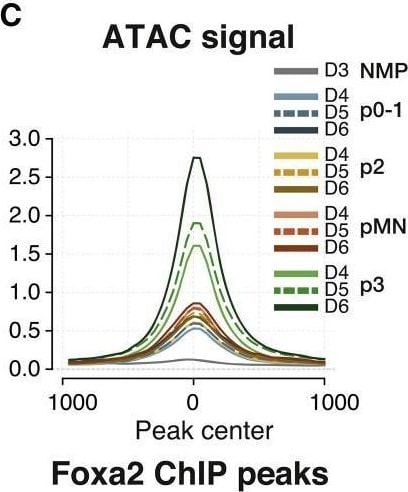
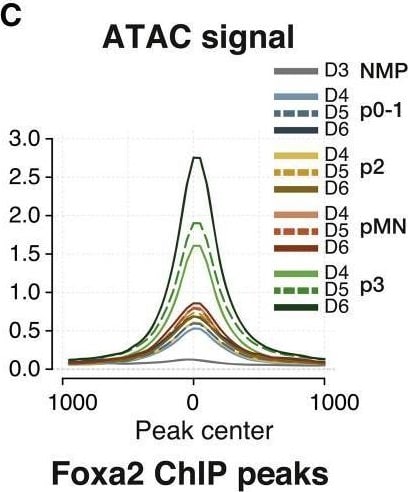
FOXA2 drives the p3-specific chromatin accessibility program(A) Footprinting score for the FOX motif is highest in p3 samples.(B) Foxa2 expression in p3 NPs suggests it is the most likely candidate to drive the footprinting signal.(C) Average ATAC-seq accessibility at FOXA2 ChIP-seq peaks13 in the indicated samples shows these regions are highly accessible in p3 NPs.(D) Normalized FOXA2 ChIP-seq coverage showing accessibility in p3-specific elements from the groups of ATAC-seq elements in the indicated clusters from Figure 3B.(E) Genetic lineage tracing indicates that cells that expressed Foxa2 at E8.5 (tamoxifen administration) have contributed to the p3 progenitor and V3 neuronal cell types by E11.5 (red arrows).(F) Quantifications of p3 and V3 cells expressing tdTomato in embryos collected after tamoxifen administration at the indicated times. Biological replicates: n = 2 for E7.5, n = 4 for E8.5, n = 3 for E9.5.(G) Foxa2−/− ES cells fail to generate p3 NPs when exposed to 500 nM SAG.(H) Representative flow cytometry plots of the quantifications in (G) showing a marked reduction in p3 NPs from Foxa2−/− ES cells compared with wild type. Cells are gated for SOX2+ live neural progenitors.(I) Representative immunohistochemistry staining for SOX2, OLIG2 and NKX2,2 showing reduced number of cells expressing NKX2.2 in Foxa2−/− mutant cells at day 6 of differentiation treated with 500 nM SAG.Scale bars, 50 μm.See also Figure S4. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/36516856), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of HNF-3 beta /FoxA2 by Immunocytochemistry/ Immunofluorescence
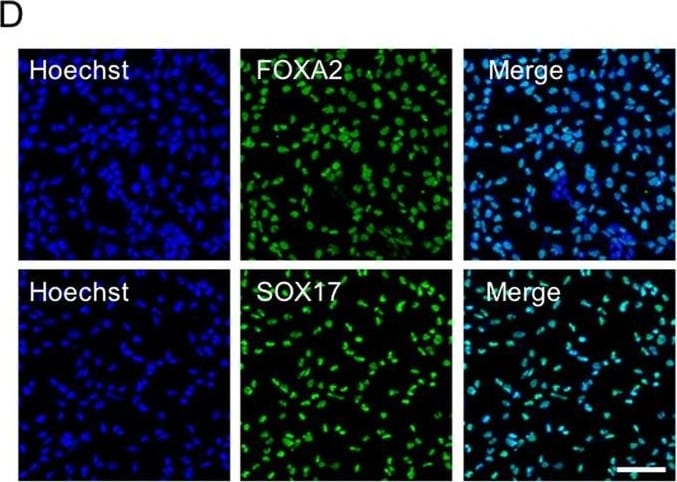
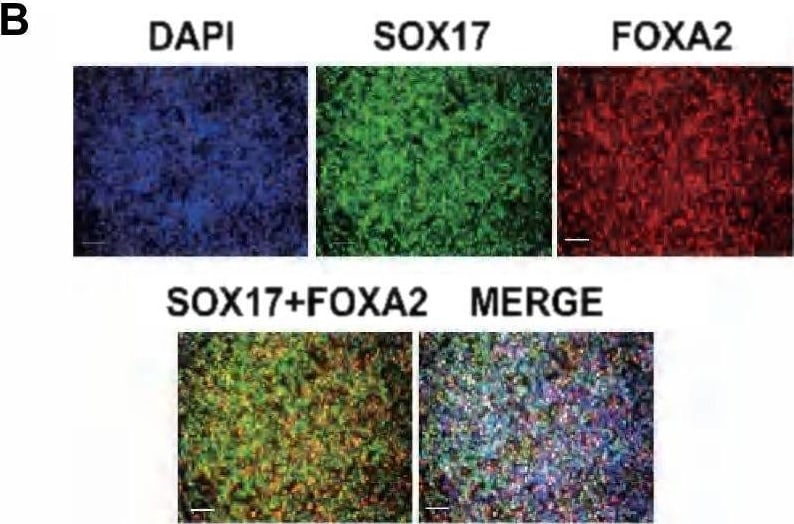
Expression profiles and functional enrichments of marker genes across time points identified during hESC differentiation. A. UMAP visualization of the expression of well-defined marker genes of different stages in hESC differentiation. B. The representative immunofluorescence staining for definitive endoderm cells with antibodies against SOX17 and FOXA2. DAPI serves as a nucleus indicator. The individual color channels were merged to assess the colocalization of SOX17 an FOXA2 expression in the nuclei. Scale bars, 200 μm. C. Dot plot showing the average and percentage expression of well-defined marker genes in different stages or cell types. Genes are colored according to their mean expression level. Diameter denotes fractional expression. SC-alpha, SC-beta, and SC-EC represent stem-cell-derived alpha, beta, and enterochromaffin cells, respectively. D. Heatmap showing the expression of the top ten marker genes of each time point. lncRNA genes are indicated by arrows. E. Enriched GO terms of marker genes of each time point. F. UMAP visualization of the expression of genes related to endoderm formation. G. Violin plots showing the expression of lncRNA genes that listed in the top 10 marker genes of each time point Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/36907881), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of HNF-3 beta /FoxA2 by Immunocytochemistry/ Immunofluorescence
Expression profiles and functional enrichments of marker genes across time points identified during hESC differentiation. A. UMAP visualization of the expression of well-defined marker genes of different stages in hESC differentiation. B. The representative immunofluorescence staining for definitive endoderm cells with antibodies against SOX17 and FOXA2. DAPI serves as a nucleus indicator. The individual color channels were merged to assess the colocalization of SOX17 an FOXA2 expression in the nuclei. Scale bars, 200 μm. C. Dot plot showing the average and percentage expression of well-defined marker genes in different stages or cell types. Genes are colored according to their mean expression level. Diameter denotes fractional expression. SC-alpha, SC-beta, and SC-EC represent stem-cell-derived alpha, beta, and enterochromaffin cells, respectively. D. Heatmap showing the expression of the top ten marker genes of each time point. lncRNA genes are indicated by arrows. E. Enriched GO terms of marker genes of each time point. F. UMAP visualization of the expression of genes related to endoderm formation. G. Violin plots showing the expression of lncRNA genes that listed in the top 10 marker genes of each time point Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/36907881), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of HNF-3 beta /FoxA2 by Flow Cytometry
FOXA2 drives the p3-specific chromatin accessibility program(A) Footprinting score for the FOX motif is highest in p3 samples.(B) Foxa2 expression in p3 NPs suggests it is the most likely candidate to drive the footprinting signal.(C) Average ATAC-seq accessibility at FOXA2 ChIP-seq peaks13 in the indicated samples shows these regions are highly accessible in p3 NPs.(D) Normalized FOXA2 ChIP-seq coverage showing accessibility in p3-specific elements from the groups of ATAC-seq elements in the indicated clusters from Figure 3B.(E) Genetic lineage tracing indicates that cells that expressed Foxa2 at E8.5 (tamoxifen administration) have contributed to the p3 progenitor and V3 neuronal cell types by E11.5 (red arrows).(F) Quantifications of p3 and V3 cells expressing tdTomato in embryos collected after tamoxifen administration at the indicated times. Biological replicates: n = 2 for E7.5, n = 4 for E8.5, n = 3 for E9.5.(G) Foxa2−/− ES cells fail to generate p3 NPs when exposed to 500 nM SAG.(H) Representative flow cytometry plots of the quantifications in (G) showing a marked reduction in p3 NPs from Foxa2−/− ES cells compared with wild type. Cells are gated for SOX2+ live neural progenitors.(I) Representative immunohistochemistry staining for SOX2, OLIG2 and NKX2,2 showing reduced number of cells expressing NKX2.2 in Foxa2−/− mutant cells at day 6 of differentiation treated with 500 nM SAG.Scale bars, 50 μm.See also Figure S4. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/36516856), licensed under a CC-BY license. Not internally tested by R&D Systems.Applications for Human HNF‑3 beta /FoxA2 Antibody
Application
Recommended Usage
Chromatin Immunoprecipitation (ChIP)
5 µg/5 x 106 cells
Sample: Mouse splenocyte chromatin, E-RABP promoter detected by standard PCR.
Sample: Mouse splenocyte chromatin, E-RABP promoter detected by standard PCR.
Immunocytochemistry
5-15 µg/mL
Sample: Immersion fixed endoderm differentiated BG01V human embryonic stem cells and immersion fixed HepG2 Human Hepatocellular Carcinoma Cells (Positive) and absent in THP‑1 Human Acute Monocytic Leukemia Cells (Negative)
Sample: Immersion fixed endoderm differentiated BG01V human embryonic stem cells and immersion fixed HepG2 Human Hepatocellular Carcinoma Cells (Positive) and absent in THP‑1 Human Acute Monocytic Leukemia Cells (Negative)
Immunohistochemistry
3-15 µg/mL
Sample: Immersion fixed paraffin-embedded sections of human liver
Sample: Immersion fixed paraffin-embedded sections of human liver
Western Blot
1 µg/mL
Sample: HepG2 human hepatocellular carcinoma cell line
Sample: HepG2 human hepatocellular carcinoma cell line
Reviewed Applications
Read 3 reviews rated 4.7 using AF2400 in the following applications:
Formulation, Preparation, and Storage
Purification
Antigen Affinity-purified
Reconstitution
Reconstitute at 0.2 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: HNF-3 beta/FoxA2
Long Name
Hepatocyte Nuclear Factor-3, beta
Alternate Names
FoxA2, HNF3 beta
Entrez Gene IDs
3170 (Human)
Gene Symbol
FOXA2
UniProt
Additional HNF-3 beta/FoxA2 Products
Product Documents for Human HNF‑3 beta /FoxA2 Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human HNF‑3 beta /FoxA2 Antibody
For research use only
Citations for Human HNF‑3 beta /FoxA2 Antibody
Customer Reviews for Human HNF‑3 beta /FoxA2 Antibody (3)
4.7 out of 5
3 Customer Ratings
Have you used Human HNF‑3 beta /FoxA2 Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
Showing
1
-
3 of
3 reviews
Showing All
Filter By:
-
Application: ImmunohistochemistrySample Tested: Midbrain organoid derived from iPSCSpecies: Midbrain organoid derived from iPSCVerified Customer | Posted 01/21/2026FOXA2-488 OTX2-568 DAPIWorks well on our tissue. Dilution used: 1:100
-
Application: ImmunohistochemistrySample Tested: Prostate cancer tissueSpecies: HumanVerified Customer | Posted 09/10/2021
-
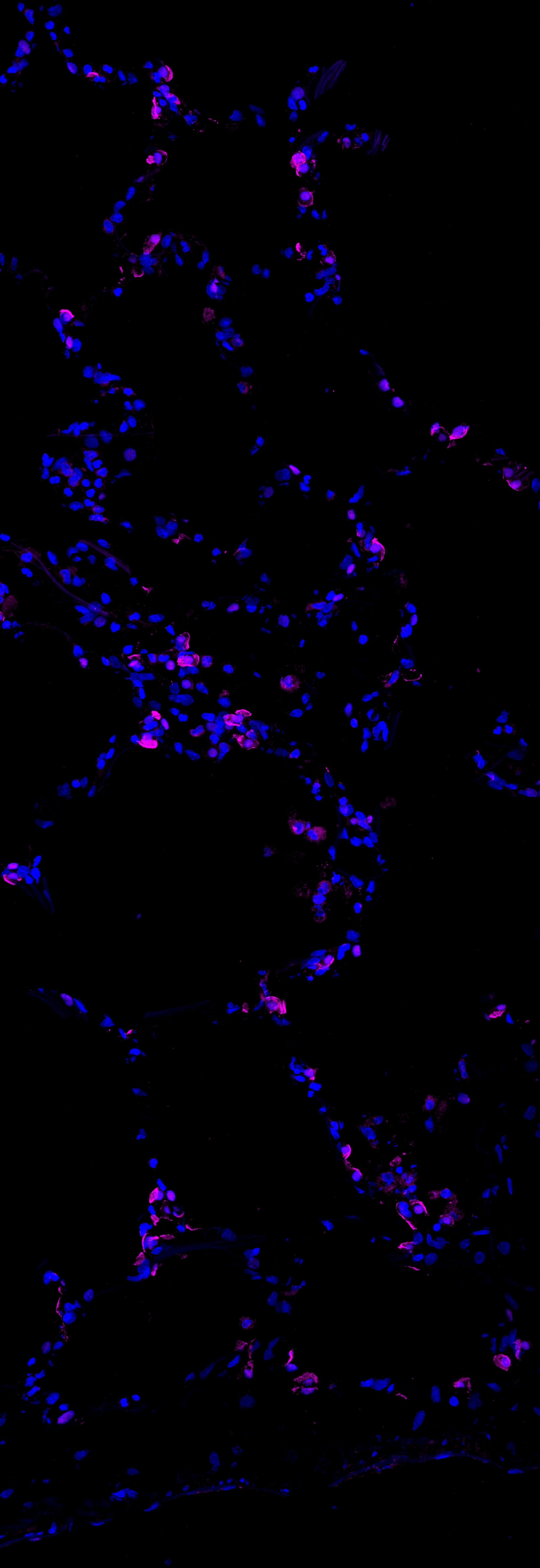
Application: Immunocytochemistry/ImmunofluorescenceSample Tested: alveoliSpecies: HumanVerified Customer | Posted 02/15/2019dapi- BLUE magenta - foxa2
There are no reviews that match your criteria.
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- ChIP Protocol Video
- Chromatin Immunoprecipitation (ChIP) Protocol
- Chromatin Immunoprecipitation Protocol
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- ICC Cell Smear Protocol for Suspension Cells
- ICC Immunocytochemistry Protocol Videos
- ICC for Adherent Cells
- IHC Sample Preparation (Frozen sections vs Paraffin)
- Immunocytochemistry (ICC) Protocol
- Immunocytochemistry Troubleshooting
- Immunofluorescence of Organoids Embedded in Cultrex Basement Membrane Extract
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Fluorescent ICC Staining of Cell Smears - Graphic
- Protocol for the Fluorescent ICC Staining of Cultured Cells on Coverslips - Graphic
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent ICC Staining of Cells on Coverslips
- Protocol for the Preparation and Fluorescent ICC Staining of Non-adherent Cells
- Protocol for the Preparation and Fluorescent ICC Staining of Stem Cells on Coverslips
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- Protocol for the Preparation of a Cell Smear for Non-adherent Cell ICC - Graphic
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Immunohistochemistry
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...
Associated Pathways