Signal Transducer and Activator of Transcription (STAT) proteins are transcription factors activated in response to cytokine, growth factor, or hormone receptor signaling. Janus kinases (JAKs) phosphorylate STAT proteins and induce dimerization. Homo- or heterodimers translocate to the nucleus where they bind to DNA and activate transcription.
Human Phospho-STAT3 (Y705) Antibody
R&D Systems | Catalog # AF4607

Key Product Details
Validated by
Biological Validation
Species Reactivity
Validated:
Human
Cited:
Human, Mouse
Applications
Validated:
Western Blot, Flow Cytometry, Immunocytochemistry, Simple Western, CyTOF-ready
Cited:
Western Blot, Flow Cytometry
Label
Unconjugated
Antibody Source
Polyclonal Rabbit IgG
Loading...
Product Specifications
Immunogen
Phosphopeptide containing human STAT3 Y705 site
Specificity
Detects human STAT3 when phosphorylated at Y705.
Clonality
Polyclonal
Host
Rabbit
Isotype
IgG
Scientific Data Images for Human Phospho-STAT3 (Y705) Antibody
Detection of Human Phospho-STAT3 (Y705) by Western Blot.
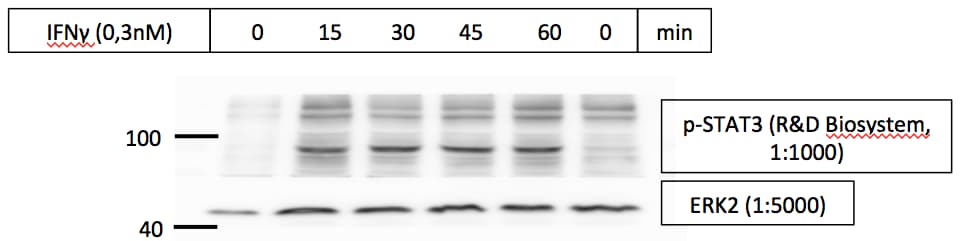
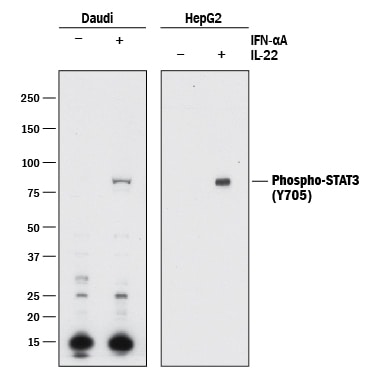
Western blot shows lysates of Daudi human Burkitt's lymphoma cell line and HepG2 human hepatocellular carcinoma cell line untreated (-) or treated (+) with 500 U/mL Recombinant Human IFN-aA (Catalog # 11100-1) for 20 minutes or 50 µg/mL Recombinant Human IL-22 (Catalog # 782-IL) for 15 minutes. PVDF membrane was probed with 0.5 µg/mL of Rabbit Anti-Human Phospho-STAT3 (Y705) Antigen Affinity-purified Polyclonal Antibody (Catalog # AF4607) followed by HRP-conjugated Anti-Rabbit IgG Secondary Antibody (Catalog # HAF008). A specific band was detected for STAT3 at approximately 95 kDa (as indicated). This experiment was conducted under reducing conditions and using Immunoblot Buffer Group 1.Detection of Phospho-STAT3 (Y705) in IFN-alpha-treated Human Daudi Cell Line by Flow Cytometry.
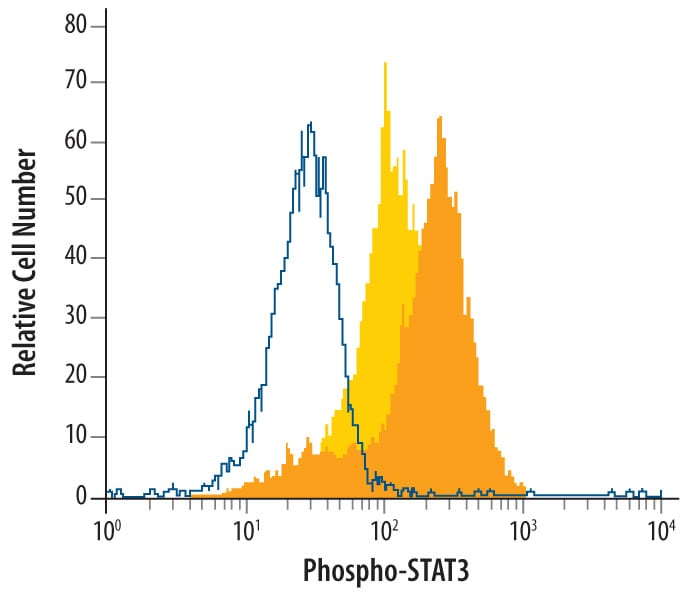
Daudi human Burkitt's lymphoma cell line was unstimulated (light orange filled histogram) or treated with 500 U/mL rhIFN-alpha for 20 minutes (dark orange filled histogram) was stained with Human Phospho-STAT3 (Y705) Antigen Affinity-purified Polyclonal Antibody (Catalog # AF4607) or control antibody (Catalog # AB-105-C, open histogram), followed by Phycoerythrin-conjugated Anti-Rabbit IgG Secondary Antibody (Catalog # F0110). To facilitate intracellular staining, cells were fixed with paraformaldehyde and permeabilized with methanol.STAT3 in Daudi Human Burkitt's Lymphoma Cells.
Phospho-STAT3 was detected in immersion fixed IFN-alpha treated Daudi human Burkitt's lymphoma cell line using 10 µg/mL Human Phospho-STAT3 (Y705) Antigen Affinity-purified Polyclonal Antibody (Catalog # AF4607) for 3 hours at room temperature. Cells were stained with the NorthernLights™ 557-conjugated Anti-Rabbit IgG Secondary Antibody (red; Catalog # NL004) and counterstained with DAPI (blue). View our protocol for Fluorescent ICC Staining of Cells on Coverslips.Detection of Human STAT3 by Simple WesternTM.
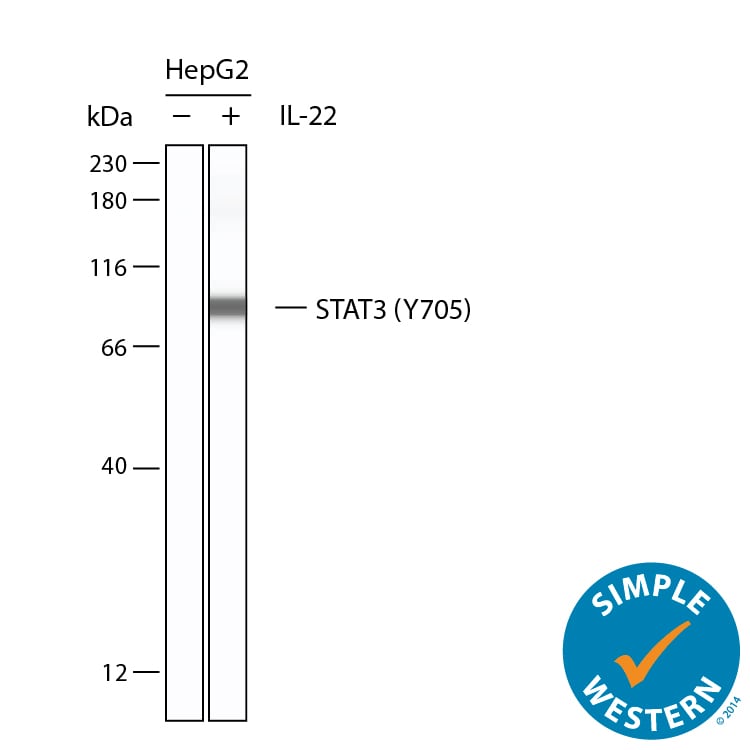
Simple Western shows lysates of Daudi human Burkitt's lymphoma cell line untreated (-) or treated (+) with 50 µg/mL Recombinant Human IL‑22 (Catalog # 782-IL) for 15 minutes, loaded at 0.2 mg/ml. A specific band was detected for STAT3 at approximately 91 kDa (as indicated) using 10 µg/mL of Rabbit Anti-Human Phospho-STAT3 (Y705) Antigen Affinity-purified Polyclonal Antibody (Catalog # AF4607). This experiment was conducted under reducing conditions and using the 12-230kDa separation system.Detection of Human STAT3 by Western Blot
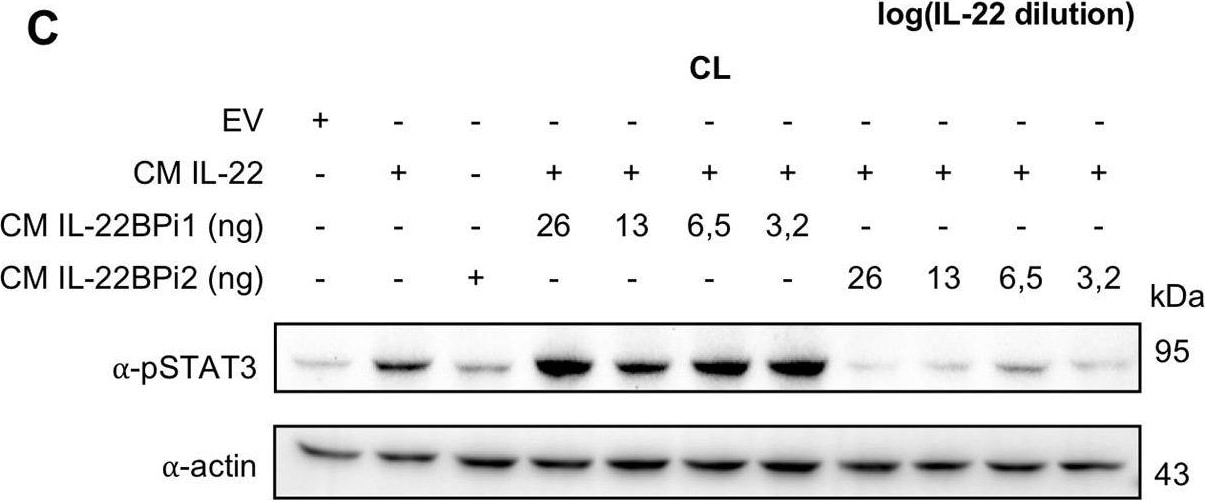
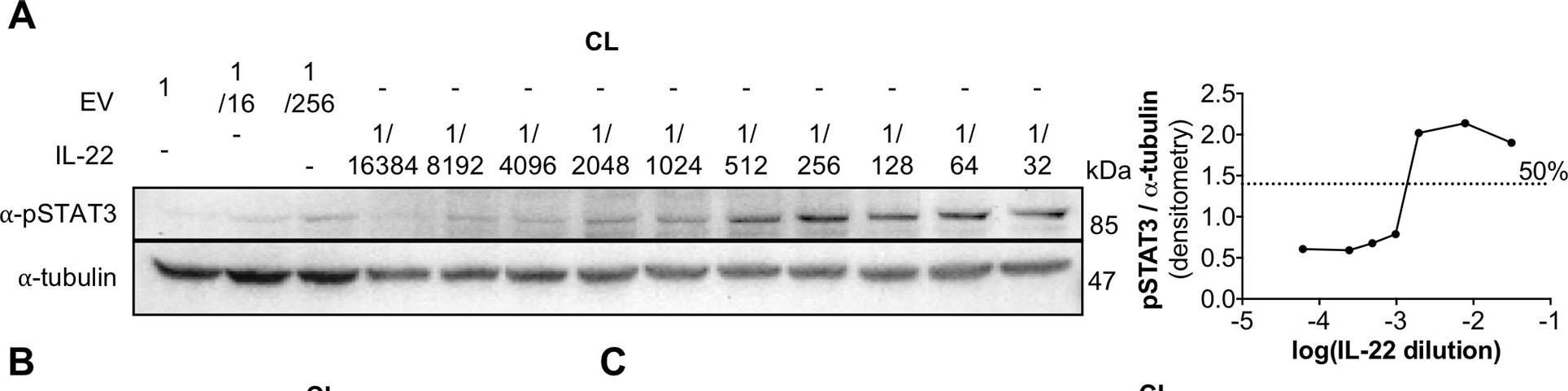
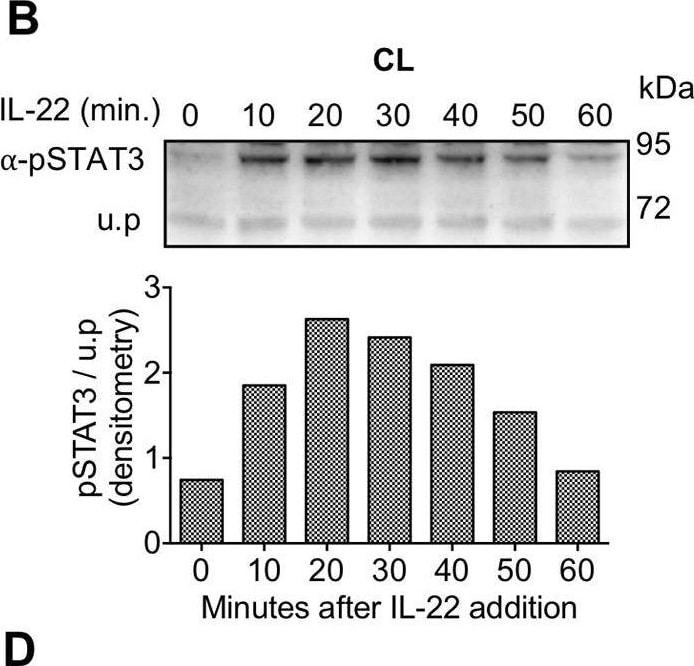
IL-22BPi1 does not interact with IL-22 or IL-22BPi2. (A) A549 cells were exposed for 30 minutes to dilutions of IL-22-containing culture medium (CM) previously produced by IL22-transfected HEK293 cells. A549 cell lysates (CL) were immunoblotted for pSTAT3 and tubulin as loading control. The relative densitometry of pSTAT3 normalized to that of tubulin is also represented. (B) A549 cells were treated with the optimum IL-22 dilution from A (1/512) for different periods of time, lysed and immunoblotted for pSTAT3. An unspecific protein band (u.p.) was used as loading control. (C) IL-22BP concentration in conditioned medium (CM) of transfected HEK293 cells was measured by ELISA, and the indicated amounts in nanograms (ng) of IL-22BPi1 or IL-22BPi2 were pre-incubated for 1 h at 37°C with the selected IL-22 concentration from (A). A549 were exposed to the pre-incubated combinations for 20 min. An excess of IL-22BPi2 was used as phosphorylation blocking control (lane 3). Cells were lysed and immunoblotted for pSTAT3 and actin as loading control. (D) HeLa cells were co-transfected with the indicated expression plasmids, 24 h later cells were lysed and the CM was subjected to acetone precipitation (AP), and proteins were immunoblotted for FLAG. Intracellular IL-22BPi1 is indicated with dark purple arrows, intracellular and secreted IL-22BPi2 is indicated with green arrows, and co-expressed IL-22 and IL-17 are also indicated. (E) Conditioned media (CM) from 3 independent experiments, in which expression vectors for the three IL-22BP isoforms were individually transfected into HEK293 cells together with either IL-17 or IL-22 expression vectors or an empty vector control (EV), were analyzed 24 h after transfection by ELISA for IL-22BP (mean ± SEM; n = 3; **p < 0.01 by unpaired t-test). (F) IL-22BPi1 does not interact with IL-22BPi2. IL-22BPi1-MF expression plasmid containing Myc and FLAG tags was co-transfected with an inducible pTRE3G-based vector expressing IL-22BPi2 with only a FLAG tag. After 24 h, cells were induced for IL-22BPi2 production by adding Tet-Express activator to the medium for a further 24 h. Cells were lysed and immunoprecipitated with anti-Myc agarose, the flow-through fractions were then further subjected to FLAG immunoprecipitation. CL and eluted fractions were immunoblotted for FLAG. Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/30619294), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human STAT3 by Western Blot
IL-22BPi1 does not interact with IL-22 or IL-22BPi2. (A) A549 cells were exposed for 30 minutes to dilutions of IL-22-containing culture medium (CM) previously produced by IL22-transfected HEK293 cells. A549 cell lysates (CL) were immunoblotted for pSTAT3 and tubulin as loading control. The relative densitometry of pSTAT3 normalized to that of tubulin is also represented. (B) A549 cells were treated with the optimum IL-22 dilution from A (1/512) for different periods of time, lysed and immunoblotted for pSTAT3. An unspecific protein band (u.p.) was used as loading control. (C) IL-22BP concentration in conditioned medium (CM) of transfected HEK293 cells was measured by ELISA, and the indicated amounts in nanograms (ng) of IL-22BPi1 or IL-22BPi2 were pre-incubated for 1 h at 37°C with the selected IL-22 concentration from (A). A549 were exposed to the pre-incubated combinations for 20 min. An excess of IL-22BPi2 was used as phosphorylation blocking control (lane 3). Cells were lysed and immunoblotted for pSTAT3 and actin as loading control. (D) HeLa cells were co-transfected with the indicated expression plasmids, 24 h later cells were lysed and the CM was subjected to acetone precipitation (AP), and proteins were immunoblotted for FLAG. Intracellular IL-22BPi1 is indicated with dark purple arrows, intracellular and secreted IL-22BPi2 is indicated with green arrows, and co-expressed IL-22 and IL-17 are also indicated. (E) Conditioned media (CM) from 3 independent experiments, in which expression vectors for the three IL-22BP isoforms were individually transfected into HEK293 cells together with either IL-17 or IL-22 expression vectors or an empty vector control (EV), were analyzed 24 h after transfection by ELISA for IL-22BP (mean ± SEM; n = 3; **p < 0.01 by unpaired t-test). (F) IL-22BPi1 does not interact with IL-22BPi2. IL-22BPi1-MF expression plasmid containing Myc and FLAG tags was co-transfected with an inducible pTRE3G-based vector expressing IL-22BPi2 with only a FLAG tag. After 24 h, cells were induced for IL-22BPi2 production by adding Tet-Express activator to the medium for a further 24 h. Cells were lysed and immunoprecipitated with anti-Myc agarose, the flow-through fractions were then further subjected to FLAG immunoprecipitation. CL and eluted fractions were immunoblotted for FLAG. Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/30619294), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human STAT3 by Western Blot
IL-22BPi1 does not interact with IL-22 or IL-22BPi2. (A) A549 cells were exposed for 30 minutes to dilutions of IL-22-containing culture medium (CM) previously produced by IL22-transfected HEK293 cells. A549 cell lysates (CL) were immunoblotted for pSTAT3 and tubulin as loading control. The relative densitometry of pSTAT3 normalized to that of tubulin is also represented. (B) A549 cells were treated with the optimum IL-22 dilution from A (1/512) for different periods of time, lysed and immunoblotted for pSTAT3. An unspecific protein band (u.p.) was used as loading control. (C) IL-22BP concentration in conditioned medium (CM) of transfected HEK293 cells was measured by ELISA, and the indicated amounts in nanograms (ng) of IL-22BPi1 or IL-22BPi2 were pre-incubated for 1 h at 37°C with the selected IL-22 concentration from (A). A549 were exposed to the pre-incubated combinations for 20 min. An excess of IL-22BPi2 was used as phosphorylation blocking control (lane 3). Cells were lysed and immunoblotted for pSTAT3 and actin as loading control. (D) HeLa cells were co-transfected with the indicated expression plasmids, 24 h later cells were lysed and the CM was subjected to acetone precipitation (AP), and proteins were immunoblotted for FLAG. Intracellular IL-22BPi1 is indicated with dark purple arrows, intracellular and secreted IL-22BPi2 is indicated with green arrows, and co-expressed IL-22 and IL-17 are also indicated. (E) Conditioned media (CM) from 3 independent experiments, in which expression vectors for the three IL-22BP isoforms were individually transfected into HEK293 cells together with either IL-17 or IL-22 expression vectors or an empty vector control (EV), were analyzed 24 h after transfection by ELISA for IL-22BP (mean ± SEM; n = 3; **p < 0.01 by unpaired t-test). (F) IL-22BPi1 does not interact with IL-22BPi2. IL-22BPi1-MF expression plasmid containing Myc and FLAG tags was co-transfected with an inducible pTRE3G-based vector expressing IL-22BPi2 with only a FLAG tag. After 24 h, cells were induced for IL-22BPi2 production by adding Tet-Express activator to the medium for a further 24 h. Cells were lysed and immunoprecipitated with anti-Myc agarose, the flow-through fractions were then further subjected to FLAG immunoprecipitation. CL and eluted fractions were immunoblotted for FLAG. Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/30619294), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human Human Phospho-STAT3 (Y705) Antibody by Western Blot
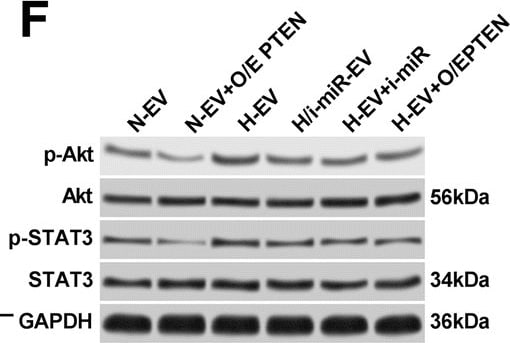
N-EV and H-EV treatment promote macrophage M2 polarization by delivering miR-21-5p that targets PTEN. a, western blot analysis of PTEN protein expression level in induced macrophages. H/i-miR-EV, monocytes were induced with the presence of EV secreted by miR-21-5p-inhibited, hypoxia pre-challenged MSCs; H-EV + i-miR, monocytes were transfected with miR-21-5p inhibitor-expressing vector before induction with the presence of H-EV. Macrophages induced without MSC-EV were used as negative control (NC). b, c, flow cytometry determining the percentage of CD163+CD206+ cells among total CD68+ cells after induction. N-EV + O/E PTEN or H-EV + O/E PTEN, monocytes were transfected with PTEN overexpressing vector before N-EV or H-EV treatment, respectively. d–f, western blot detecting Akt and STAT3 protein expression as well as their activating phosphorylation (p-Ser473 for Akt and p-tyr705 for STAT3) in macrophages after induction. g–i, ELISA evaluating IL-10, TGF-beta and VEGF-alpha in macrophage culture medium after induction. Macrophages induced with the presence of N-EV were used as negative control in b–i. Tukey’s test was used for statistical analysis. *, p < 0.05; **, p < 0.01; ***, p < 0.001; ****, p < 0.0001 Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/30736829), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Phospho-STAT3 (Y705) by Flow Cytometry
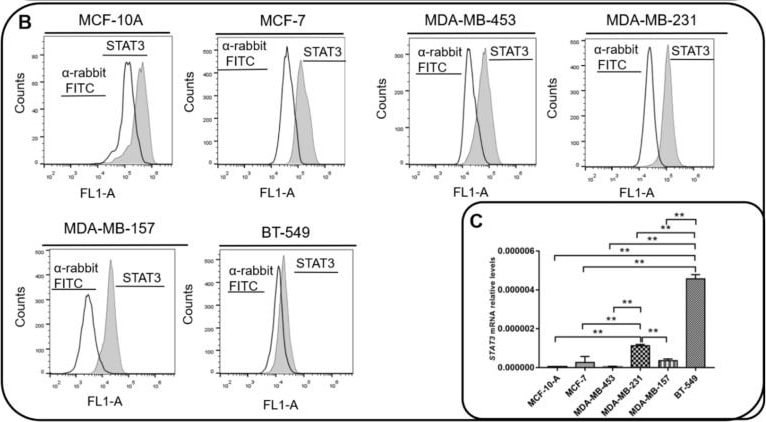
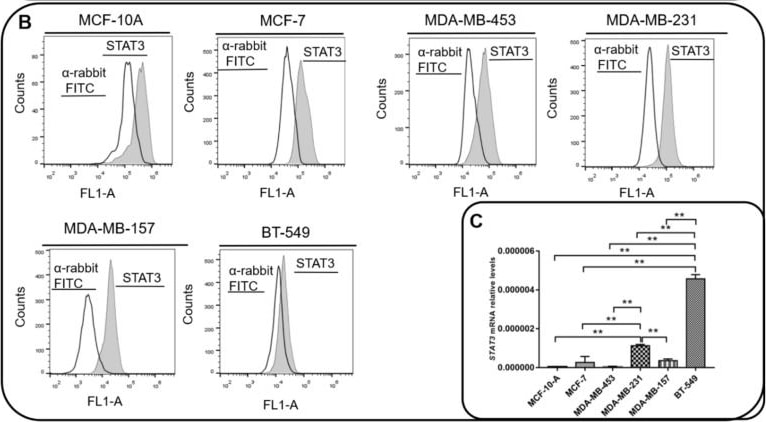
Interleukin 6 (IL-6) and STAT3 expression in BC cells. (A) IL-6 levels were measured by ELISA assay in supernatants of MCF-10, MCF-7, MDA-MB-231, BT-549, MDA-MB-157 and MDA-MB-453 cells. IL-6 mRNA relative levels of MCF-10, MCF-7, MDA-MB-231, BT-549, MDA-MB-157 and MDA-MB-453 cells were also recorded. (B) Expression of activated STAT3 (STAT3pY705) in MCF-10, MCF-7, MDA-MB-231, BT-549, MDA-MB-157 and MDA-MB-453 cells was analyzed by flow cytometry (gray peak). As the negative control, the secondary antibody alone was used (white peak). (C) STAT3 mRNA relative levels of MCF-10, MCF-7, MDA-MB-231, BT-549, MDA-MB-157 and MDA-MB-453 cells. (D) STAT3 phosphorylation levels (STAT3pY705-dark gray peak with tracing line) were analyzed in MDA-MB-231 and MDA-MB-157 cells treated with Tocilizumab (TCZ), recombinant IL-6 (rIL-6) and STAT3 inhibitor, STATTIC (STC). As the negative control, the secondary antibody alone was used (white peak). * p < 0.05; ** p < 0.001. Three independent experiments were performed. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/35626741), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Phospho-STAT3 (Y705) by Flow Cytometry
Interleukin 6 (IL-6) and STAT3 expression in BC cells. (A) IL-6 levels were measured by ELISA assay in supernatants of MCF-10, MCF-7, MDA-MB-231, BT-549, MDA-MB-157 and MDA-MB-453 cells. IL-6 mRNA relative levels of MCF-10, MCF-7, MDA-MB-231, BT-549, MDA-MB-157 and MDA-MB-453 cells were also recorded. (B) Expression of activated STAT3 (STAT3pY705) in MCF-10, MCF-7, MDA-MB-231, BT-549, MDA-MB-157 and MDA-MB-453 cells was analyzed by flow cytometry (gray peak). As the negative control, the secondary antibody alone was used (white peak). (C) STAT3 mRNA relative levels of MCF-10, MCF-7, MDA-MB-231, BT-549, MDA-MB-157 and MDA-MB-453 cells. (D) STAT3 phosphorylation levels (STAT3pY705-dark gray peak with tracing line) were analyzed in MDA-MB-231 and MDA-MB-157 cells treated with Tocilizumab (TCZ), recombinant IL-6 (rIL-6) and STAT3 inhibitor, STATTIC (STC). As the negative control, the secondary antibody alone was used (white peak). * p < 0.05; ** p < 0.001. Three independent experiments were performed. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/35626741), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Phospho-STAT3 (Y705) by Western Blot
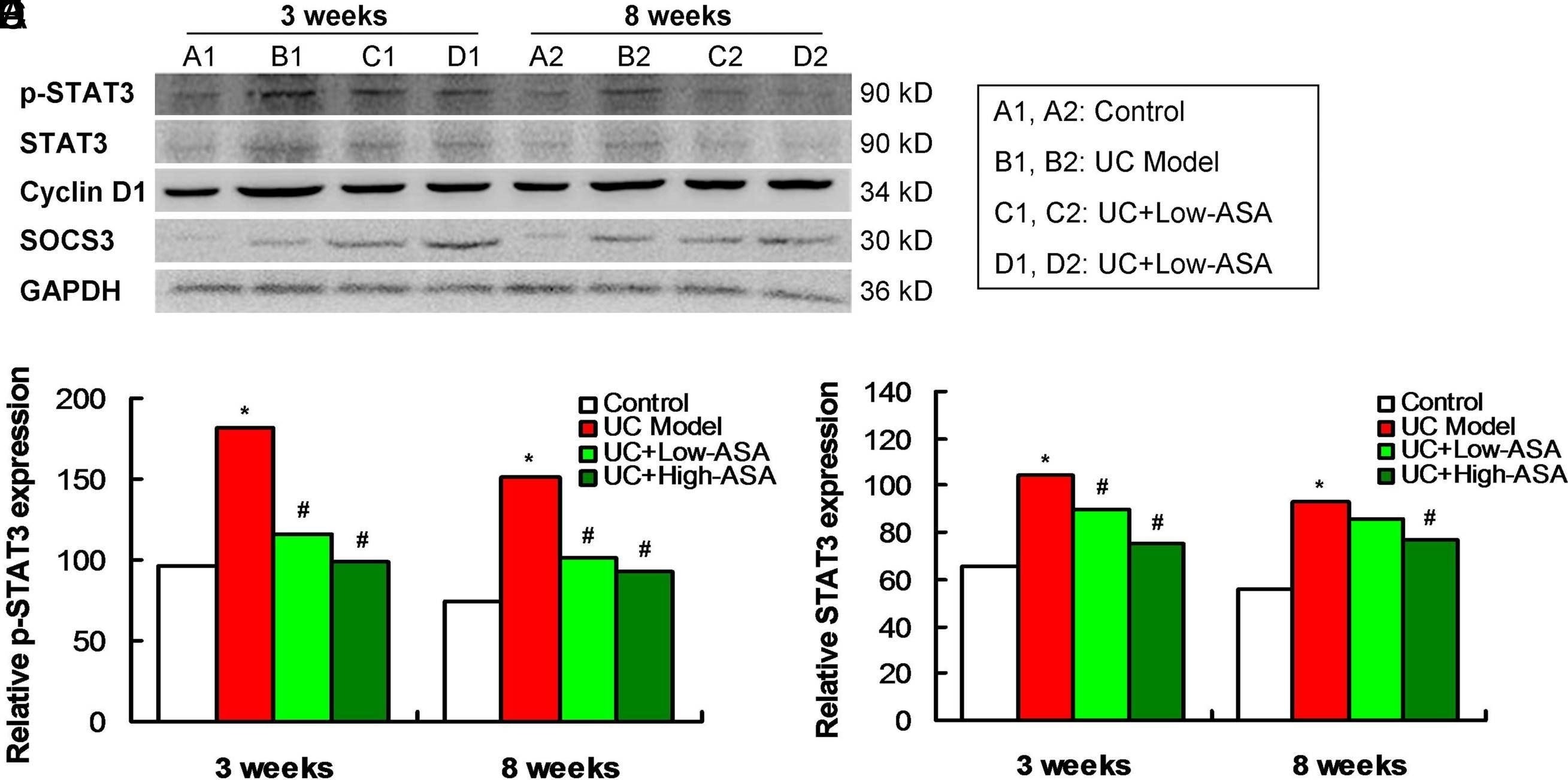
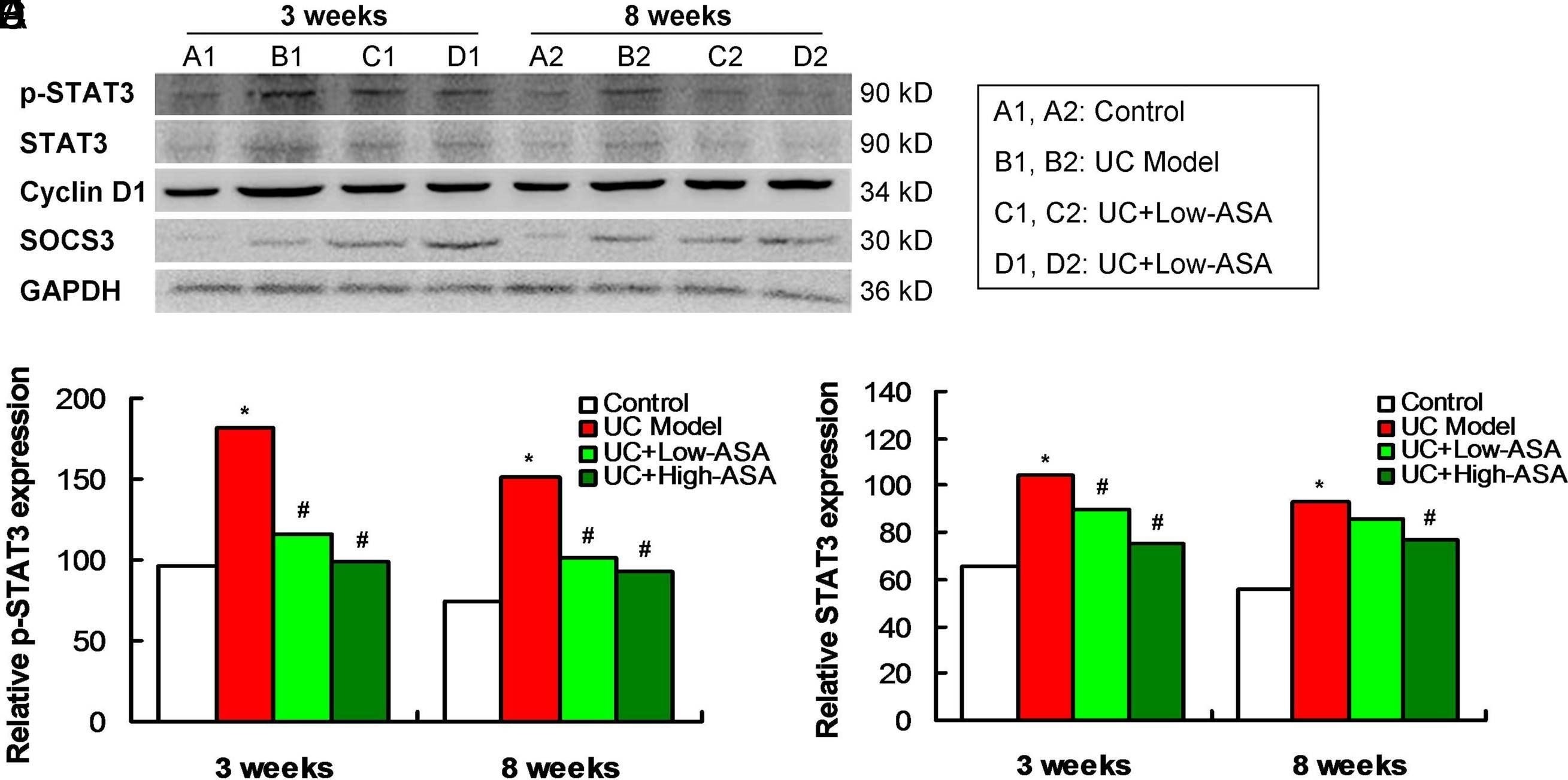
Aspirin modulated the JAK/p-STAT3 signaling pathway in atypical hyperplastic intestinal mucosal cells of UC mice (n = 4 for each group). (A) Western blotting images for JAK/p-STAT3 signaling pathway-associated molecules expression. (B) Statistical analysis and comparison for p-STAT3 expression. (C) Statistical analysis and comparison for STAT3 expression. (D) Statistical analysis and comparison for cyclin D1 expression. (E) Statistical analysis and comparison for SOCS3 expression. *P <.05 versus control group. #P <.05 versus UC model group. JAK, Janus kinase; UC, ulcerative colitis; p-STAT3, phosphorylated-STAT3; STAT3, signal transducer and activator of transcription 3; SOCS3, suppressor of cytokine signaling 3. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/35946886), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Phospho-STAT3 (Y705) by Western Blot
Aspirin modulated the JAK/p-STAT3 signaling pathway in atypical hyperplastic intestinal mucosal cells of UC mice (n = 4 for each group). (A) Western blotting images for JAK/p-STAT3 signaling pathway-associated molecules expression. (B) Statistical analysis and comparison for p-STAT3 expression. (C) Statistical analysis and comparison for STAT3 expression. (D) Statistical analysis and comparison for cyclin D1 expression. (E) Statistical analysis and comparison for SOCS3 expression. *P <.05 versus control group. #P <.05 versus UC model group. JAK, Janus kinase; UC, ulcerative colitis; p-STAT3, phosphorylated-STAT3; STAT3, signal transducer and activator of transcription 3; SOCS3, suppressor of cytokine signaling 3. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/35946886), licensed under a CC-BY license. Not internally tested by R&D Systems.Applications for Human Phospho-STAT3 (Y705) Antibody
Application
Recommended Usage
CyTOF-ready
Ready to be labeled using established conjugation methods. No BSA or other carrier proteins that could interfere with conjugation.
Flow Cytometry
0.25 µg/106 cells
Sample: Daudi human Burkitt's lymphoma cell line treated with IFN-alpha, fixed with paraformaldehyde and permeabilized with methanol
Sample: Daudi human Burkitt's lymphoma cell line treated with IFN-alpha, fixed with paraformaldehyde and permeabilized with methanol
Immunocytochemistry
5-15 µg/mL
Sample: Immersion fixed IFN-alpha treated Daudi human Burkitt's lymphoma cell line
Sample: Immersion fixed IFN-alpha treated Daudi human Burkitt's lymphoma cell line
Simple Western
10 µg/mL
Sample: Daudi human Burkitt's lymphoma cell line untreated (-) or treated (+) with 50 µg/mL Recombinant Human IL-22 (Catalog # 782-IL) for 15 minutes
Sample: Daudi human Burkitt's lymphoma cell line untreated (-) or treated (+) with 50 µg/mL Recombinant Human IL-22 (Catalog # 782-IL) for 15 minutes
Western Blot
0.25-0.5 µg/mL
Sample: Daudi human Burkitt's lymphoma cell line treated with Recombinant Human IFN‑ alpha A (Catalog # 11100-1) and HepG2 human hepatocellular carcinoma cell line treated with Recombinant Human IL‑22 (Catalog # 782-IL)
Sample: Daudi human Burkitt's lymphoma cell line treated with Recombinant Human IFN‑ alpha A (Catalog # 11100-1) and HepG2 human hepatocellular carcinoma cell line treated with Recombinant Human IL‑22 (Catalog # 782-IL)
Reviewed Applications
Read 3 reviews rated 4.7 using AF4607 in the following applications:
Flow Cytometry Panel Builder
Bio-Techne Knows Flow Cytometry
Save time and reduce costly mistakes by quickly finding compatible reagents using the Panel Builder Tool.
Advanced Features
- Spectra Viewer - Custom analysis of spectra from multiple fluorochromes
- Spillover Popups - Visualize the spectra of individual fluorochromes
- Antigen Density Selector - Match fluorochrome brightness with antigen density
Formulation, Preparation, and Storage
Purification
Antigen Affinity-purified
Reconstitution
Reconstitute at 0.2 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: STAT3
Long Name
Signal Transducer and Activator of Transcription 3
Alternate Names
Acute-phase response factor, APRFMGC16063, DNA-binding protein APRF, FLJ20882, HIES, signal transducer and activator of transcription 3, signal transducer and activator of transcription 3 (acute-phase responsefactor)
Gene Symbol
STAT3
Additional STAT3 Products
Product Documents for Human Phospho-STAT3 (Y705) Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human Phospho-STAT3 (Y705) Antibody
For research use only
Citations for Human Phospho-STAT3 (Y705) Antibody
Customer Reviews for Human Phospho-STAT3 (Y705) Antibody (3)
4.7 out of 5
3 Customer Ratings
Have you used Human Phospho-STAT3 (Y705) Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
Showing
1
-
3 of
3 reviews
Showing All
Filter By:
-
Application: Western BlotSample Tested: HEK293 human embryonic kidney cell lineSpecies: HumanVerified Customer | Posted 07/21/2018
-
Application: ImmunoprecipitationSample Tested: IPS2 induced pluripotent stem cellsSpecies: HumanVerified Customer | Posted 01/17/2018
-
Application: Western BlotSample Tested: Whole cell extracts from bone marrow derived macrophagesSpecies: MouseVerified Customer | Posted 04/05/2017
There are no reviews that match your criteria.
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- 7-Amino Actinomycin D (7-AAD) Cell Viability Flow Cytometry Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Extracellular Membrane Flow Cytometry Protocol
- Flow Cytometry Protocol for Cell Surface Markers
- Flow Cytometry Protocol for Staining Membrane Associated Proteins
- Flow Cytometry Staining Protocols
- Flow Cytometry Troubleshooting Guide
- ICC Cell Smear Protocol for Suspension Cells
- ICC Immunocytochemistry Protocol Videos
- ICC for Adherent Cells
- Immunocytochemistry (ICC) Protocol
- Immunocytochemistry Troubleshooting
- Immunofluorescence of Organoids Embedded in Cultrex Basement Membrane Extract
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Intracellular Flow Cytometry Protocol Using Alcohol (Methanol)
- Intracellular Flow Cytometry Protocol Using Detergents
- Intracellular Nuclear Staining Flow Cytometry Protocol Using Detergents
- Intracellular Staining Flow Cytometry Protocol Using Alcohol Permeabilization
- Intracellular Staining Flow Cytometry Protocol Using Detergents to Permeabilize Cells
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Propidium Iodide Cell Viability Flow Cytometry Protocol
- Protocol for Liperfluo
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Characterization of Human Th22 Cells
- Protocol for the Characterization of Human Th9 Cells
- Protocol for the Fluorescent ICC Staining of Cell Smears - Graphic
- Protocol for the Fluorescent ICC Staining of Cultured Cells on Coverslips - Graphic
- Protocol for the Preparation and Fluorescent ICC Staining of Cells on Coverslips
- Protocol for the Preparation and Fluorescent ICC Staining of Non-adherent Cells
- Protocol for the Preparation and Fluorescent ICC Staining of Stem Cells on Coverslips
- Protocol for the Preparation of a Cell Smear for Non-adherent Cell ICC - Graphic
- Protocol: Annexin V and PI Staining by Flow Cytometry
- Protocol: Annexin V and PI Staining for Apoptosis by Flow Cytometry
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Fluorokine Flow Cytometry Kits
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...
Associated Pathways
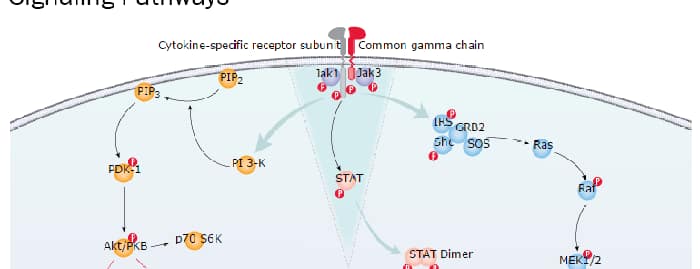 Embryonic and Induced Pluripotent Stem Cell Differentiation Pathways & Lineage-specific Markers
Embryonic and Induced Pluripotent Stem Cell Differentiation Pathways & Lineage-specific Markers
 IL-2 Signaling Pathways
IL-2 Signaling Pathways
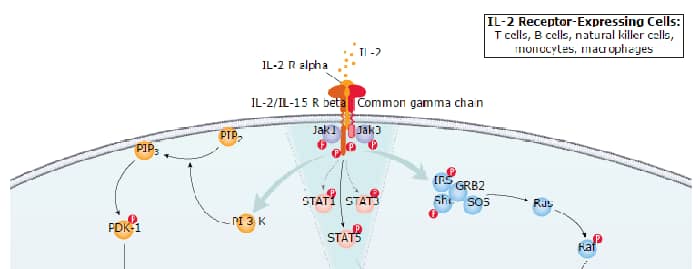 IL-7 Signaling Pathways
IL-7 Signaling Pathways
 IL-9 Signaling Pathways
IL-9 Signaling Pathways

IL-15 Signaling Pathways
 IL-21 Signaling Pathways
IL-21 Signaling Pathways
 IL-21 Signaling Pathways and their Primary Biological Effects in Different Immune Cell Types
IL-21 Signaling Pathways and their Primary Biological Effects in Different Immune Cell Types
 Jak/STAT Signaling Pathway
Jak/STAT Signaling Pathway
 Th1 Differentiation Pathway
Th1 Differentiation Pathway
 Th17 Differentiation Pathway
Th17 Differentiation Pathway
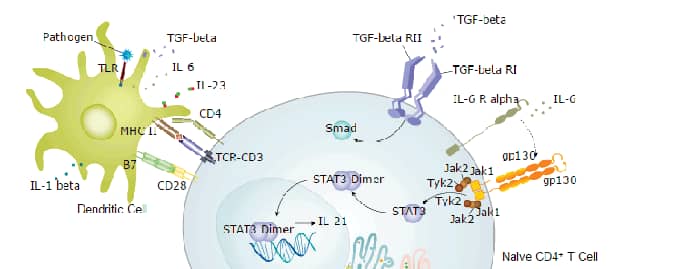 VEGF - VEGF R2 Signaling Pathways
VEGF - VEGF R2 Signaling Pathways

 IL-21 Signaling Pathways
IL-21 Signaling Pathways
 IL-21 Signaling Pathways and their Primary Biological Effects in Different Immune Cell Types
IL-21 Signaling Pathways and their Primary Biological Effects in Different Immune Cell Types
 Jak/STAT Signaling Pathway
Jak/STAT Signaling Pathway
 Th1 Differentiation Pathway
Th1 Differentiation Pathway
 Th17 Differentiation Pathway
Th17 Differentiation Pathway
 VEGF - VEGF R2 Signaling Pathways
VEGF - VEGF R2 Signaling Pathways