Vimentin is a 57 kDa class III intermediate filament (IF) protein that belongs to the intermediate filament family. It is the predominant IF in cells of mesenchymal origin such as vascular endothelium and blood cells (1‑3). The human Vimentin cDNA encodes a 466 amino acid (aa) protein that contains head and tail regions with multiple regulatory Ser/Thr phosphorylation sites, and a central rod domain with three coiled-coil regions separated by linkers (1, 2). Human Vimentin shares 97‑98% aa identity with mouse, rat, ovine, bovine and canine Vimentin. Sixteen Vimentin coiled-coil dimers self-assemble to form intermediate (10‑12 nm wide) filaments (4). These filaments then anneal longitudinally to form non-polarized fibers that support cell structure and withstand stress (4). IF fibers are highly dynamic, and half-life depends on the balance between kinase and phosphatase activity. For example, phosphorylation followed by dephosphorylation drives IF disintegration, followed by reorganization during mitosis (1, 5, 6). Interactions of head and tail domains link IFs with other structures such as actin and microtubule cytoskeletons (7). Vimentin is involved in positioning autophagosomes, lysosomes and the Golgi complex within the cell (8). It facilitates cell migration and motility by recycling internalized trailing edge integrins back to the cell surface at the leading edge (9‑11). Vimentin helps maintain the lipid composition of cellular membranes, and caspase cleavage of Vimentin is a key event in apoptosis (8, 12). Phosphorylation promotes secretion of Vimentin by TNF-alpha -stimulated macrophages (13). Extracellular Vimentin has been shown to associate with several microbes, and appears to promote an antimicrobial oxidative burst (13, 14). Cell-associated Vimentin can also interact with NKp46 to recruit NK cells to tuberculosis-infected monocytes (15).
Human/Mouse/Rat Vimentin Antibody
R&D Systems | Catalog # AF2105

Key Product Details
Validated by
Knockout/Knockdown, Biological Validation
Species Reactivity
Validated:
Human, Mouse, Rat
Cited:
Human, Mouse, Transgenic Mouse, Xenograft
Applications
Validated:
Knockout Validated, Immunohistochemistry, Western Blot, Immunocytochemistry, Simple Western
Cited:
Immunohistochemistry, Immunohistochemistry-Paraffin, Western Blot, Flow Cytometry, Immunocytochemistry, Immunoprecipitation, Binding Assay, ELISA Development
Label
Unconjugated
Antibody Source
Polyclonal Goat IgG
Loading...
Product Specifications
Immunogen
E. coli-derived recombinant human Vimentin
Ser2-Glu466
Accession # P08670
Ser2-Glu466
Accession # P08670
Specificity
Detects human, mouse, and rat Vimentin in Western blots.
Clonality
Polyclonal
Host
Goat
Isotype
IgG
Scientific Data Images for Human/Mouse/Rat Vimentin Antibody
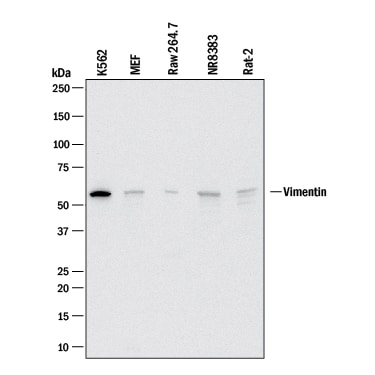
Detection of Human, Mouse, and Rat Vimentin by Western Blot.
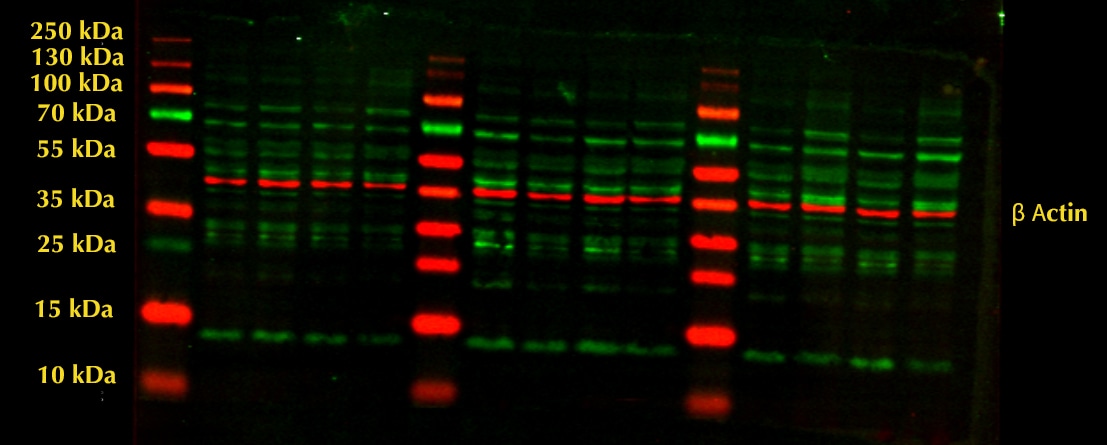
Western blot shows lysates of K562 human chronic myelogenous leukemia cell line, MEF mouse embryonic feeder cells, RAW 264.7 mouse monocyte/macrophage cell line, NR8383 rat alveolar macrophage cell line, and Rat‑2 rat embryonic fibroblast cell line. PVDF membrane was probed with 2 µg/mL of Goat Anti-Human/Mouse/Rat Vimentin Antigen Affinity-purified Polyclonal Antibody (Catalog # AF2105) followed by HRP-conjugated Anti-Goat IgG Secondary Antibody (HAF017). A specific band was detected for Vimentin at approximately 55 kDa (as indicated). This experiment was conducted under reducing conditions and using Western Blot Buffer Group 1.Vimentin in Human Skin.
Vimentin was detected in immersion fixed paraffin-embedded sections of human skin using 10 µg/mL Goat Anti-Human/Mouse/Rat Vimentin Antigen Affinity-purified Polyclonal Antibody (Catalog # AF2105) overnight at 4 °C. Tissue was stained with the Anti-Goat HRP-DAB Cell & Tissue Staining Kit (brown; Catalog # CTS008) and counterstained with hematoxylin (blue). View our protocol for Chromogenic IHC Staining of Paraffin-embedded Tissue Sections.Vimentin in HeLa Human Cell Line.
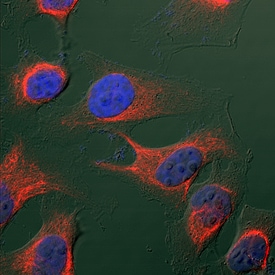
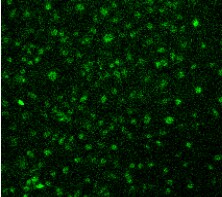
Vimentin was detected in immersion fixed HeLa human cervical epithelial carcinoma cell line using Goat Anti-Human/Mouse/Rat Vimentin Antigen Affinity-purified Polyclonal Antibody (Catalog # AF2105) at 1.7 µg/mL for 3 hours at room temperature. Cells were stained using the NorthernLights™ 557-conjugated Anti-Goat IgG Secondary Antibody (red; Catalog # NL001) and counterstained with DAPI (blue). Specific staining was localized to intermediate filaments in cytoplasm. View our protocol for Fluorescent ICC Staining of Cells on Coverslips.Detection of Human Vimentin by Simple WesternTM.
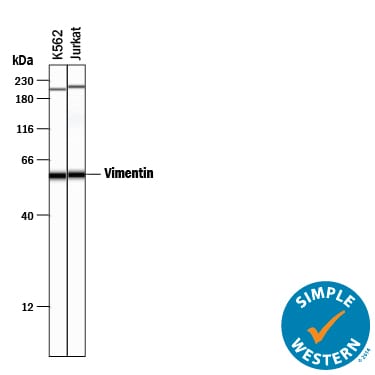
Simple Western lane view shows lysates of K562 human chronic myelogenous leukemia cell line and Jurkat human acute T cell leukemia cell line, loaded at 0.2 mg/mL. A specific band was detected for Vimentin at approximately 59 kDa (as indicated) using 10 µg/mL of Goat Anti-Human/Mouse/Rat Vimentin Antigen Affinity-purified Polyclonal Antibody (Catalog # AF2105) followed by 1:50 dilution of HRP-conjugated Anti-Goat IgG Secondary Antibody (Catalog # HAF109). This experiment was conducted under reducing conditions and using the 12-230 kDa separation system. Non-specific interaction with the 230 kDa Simple Western standard may be seen with this antibody.Vimentin Specificity is Shown by Immunocytochemistry in Knockout Cell Line.
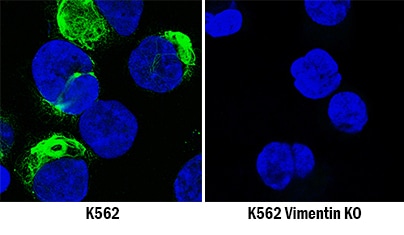
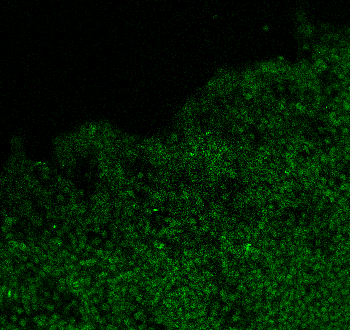
Vimentin was detected in immersion fixed K562 human chronic myelogenous leukemia cell line but is not detected in Vimentin knockout (KO) K562 Human Cell Line cell line using Goat Anti-Human/Mouse/Rat Vimentin Antigen Affinity-purified Polyclonal Antibody (Catalog # AF2105) at 5 µg/mL for 3 hours at room temperature. Cells were stained using the NorthernLights™ 493-conjugated Anti-Goat IgG Secondary Antibody (green; Catalog # NL003) and counterstained with DAPI (blue). Specific staining was localized to cytoplasm. Staining was performed using our Fluorescent ICC Staining of Non-adherent Cells protocol.Western Blot Shows Human Vimentin Specificity by Using Knockout Cell Line.
Western blot shows lysates of K562 human chronic myelogenous leukemia parental cell line and Vimentin knockout K562 cell line (KO). PVDF membrane was probed with 2 µg/mL of Goat Anti-Human/Mouse/Rat Vimentin Antigen Affinity-purified Polyclonal Antibody (Catalog # AF2105) followed by HRP-conjugated Anti-Goat IgG Secondary Antibody (Catalog # HAF017). A specific band was detected for Vimentin at approximately 55 kDa (as indicated) in the parental K562 cell line, but is not detectable in knockout K562 cell line. GAPDH (AF5718) is shown as a loading control. This experiment was conducted under reducing conditions and using Western Blot Buffer Group 1.Detection of Human Vimentin by Western Blot
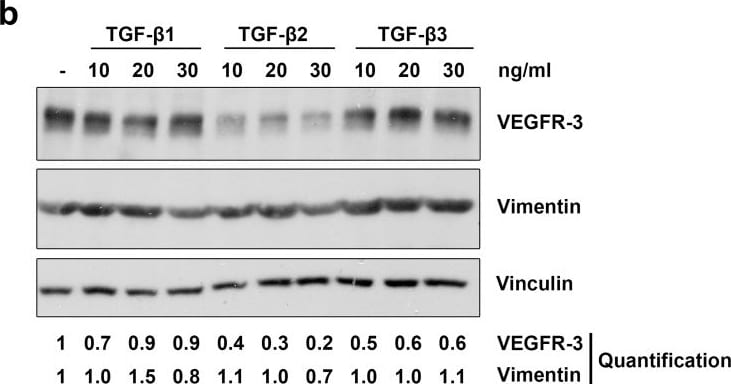
TGF-beta 1, -beta 2 and -beta 3 reduce lymphatic marker expression in LECs.Primary human LECs were treated with 10, 20 or 30 ng/ml TGF-beta 1, -beta 2 and -beta 3 for 72 hours (a) or 100 hours (b). Untreated cells served as a control. Lysates were prepared and analysed by Western blot using antibodies specific for Lyve-1, Prox-1, VEGFR-3 or vimentin. Vinculin served as loading control. The experiment was performed twice with equivalent results. For densitometry evaluation, protein bands were analysed using the software ImageJ. Bands for the Prox-1, Lyve-1, vimentin and VEGFR-3 proteins were normalized to the corresponding loading control and are displayed as the expression level relative to the untreated control samples. Image collected and cropped by CiteAb from the following publication (https://dx.plos.org/10.1371/journal.pone.0162221), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human Vimentin by Western Blot
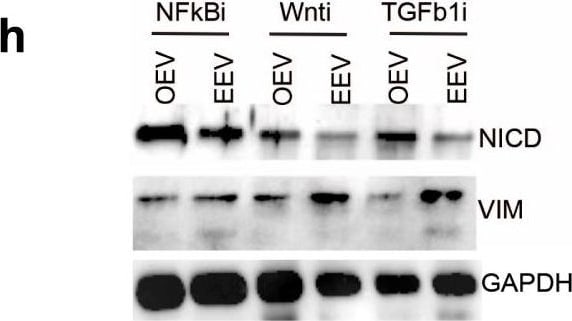
Extracellular vesicles impact multiple signaling elements of the glioma stem cell program.a Mass spectrometry quantification (n = 3) for the presence of MMPs in HUVEC-EVs (EEVs), proneural-GSC157 cells, GSC157 cells treated with CMO and GSC157 cells treated with HUVEC-CM (CME). Comparisons are made relative to untreated GSC157 cells (b, c). MMP activity of GSC157 EVs, HUVEC-EVs and HBEC5i EVs over 1 h (n = 3; b) and cumulatively (n = 3; c). d Cumulative MMP activity for GSC157 cells, GSC157 cells treated with their own EVs (OEVs), HBEC5i EVs and HUVEC-EVs over 1 h (n = 3). e Cumulative MMP activity of GSC157 cells treated with HBEC5i-EVs and either DMSO control or MMP inhibitor, BB94 (n = 3). f Expression of NICD following treatment of proneural-GSC157 cells with OEVs or EEVs in the absence or presence of MMP inhibitors, AG3340 and BB94. g DAVID analysis of the top pathways enriched in proneural-GSC157 cells treated with CME (HUVEC-conditioned media). h Expression of proneural-(NICD) and mesenchymal-(VIM) hallmarks after pharmacological inhibition of: NF kappa B pathway (Bay11-7082), Wnt pathway (LGK974) and TGF beta pathway (LY2157299). i Immunocytochemistry and quantification of phospho-P65 (p-P65) after treatment of GSC157 cells with EEVs (HUVEC-EVs). Densitometry quantifications of p-P65 (j) and total P65 (k) in GSC157 cells treated with HBEC5i-EVs (EEVs) relative to OEVs (n = 3). l Expression of activated (phospho) and total p65 in the presence of Bay11-7082 in GSC157s. m Expression of activated (phospho) and total p65 in the presence of AG3340 and BB94 in GSC157 cells exposed to EEVs. n Quantification of wound healing assay for OEV, EEV or EEVs+Bay 11-7082 treated proneural-GSC157 cells. o Schematic of the different donor EVs (OEVs and EEVs) competing for being taken up by proneural-GSC157. p Either 5,000 or 50,000 GSC157 cells exposed to a fixed amount of 30μg of HBEC5i-EVs (EEVs). q Expression of NICD in the presence of OEVs, HUVEC-EVs (EEVs), OEV:EEV (1:1) and OEV:EEV (2:1). NICD Notch intracellular domain, CMO own conditioned meida, CME endothelial conditioned media, EV extracellular vesicles, OEVs own EVs, EEVs endothelial cell derived EVs, MMP matrix metaloprotinase; HUVEC human umbilical vein endothelial cells, HBEC5i immortalized human brain brain endothelial cells. Source data are provided as a Source Data file. Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/36123372), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human Vimentin by Western Blot
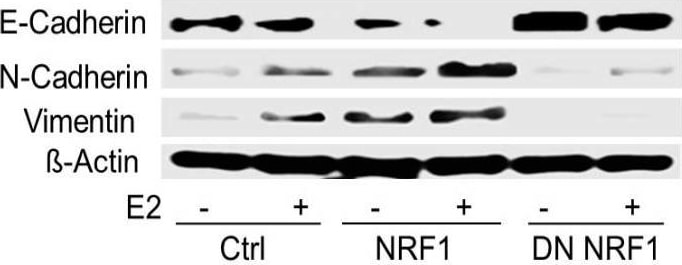
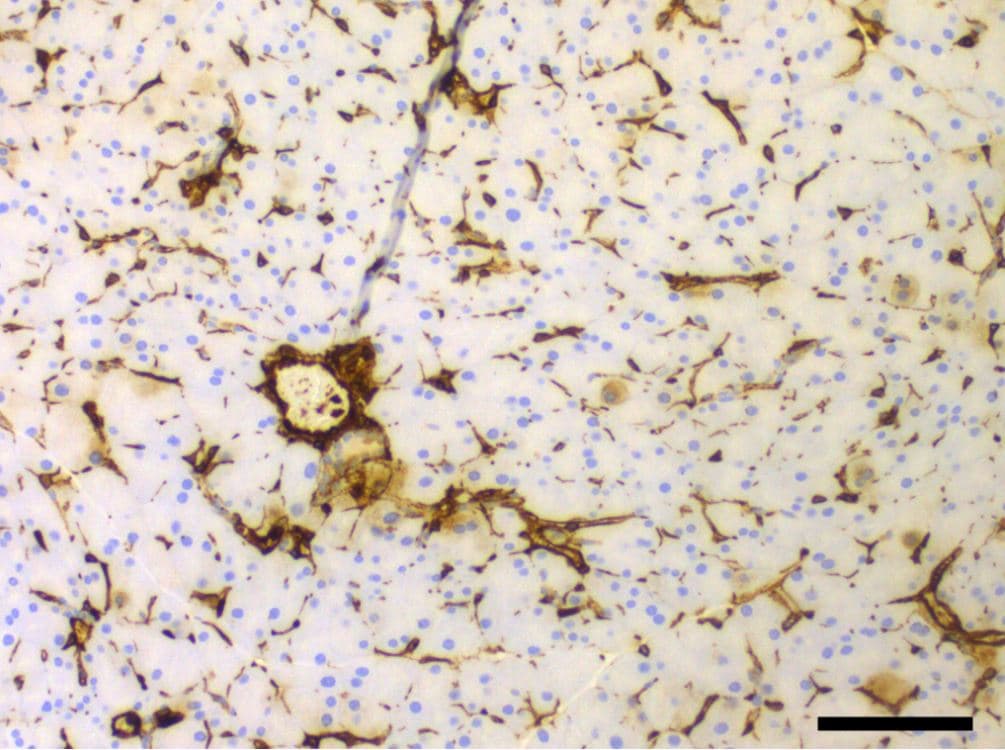
NRF1 and/or E2 re-programming contributed to the overexpression of pluripotency and epithelial-mesenchymal transition (EMT) markers and differentiation of tumor initiating breast cancer cells to other-types of cells. Experimental conditions were the same as Figure 1. (A) Immunofluorescent and flow cytometry detections of MCF10A (T) tumor initiating pluripotent stem cells markers (SOX2, Oct4, and Nanog) in transformed breast epithelial cells of MCF10A [MCF-10A (T)] overexpressing vector (control) or NRF1 treated with DMSO and E2. (B) Immunofluorescent and flow cytometry detections of pluripotent stem cells markers (SOX2 and Oct4) in transformed breast epithelial cells of MCF10A [MCF-10A (T)] overexpressing vector (control) or NRF1 treated with DMSO and E2. (C) Immunofluorescent and Western detections of the expression of EMT markers E-cadherin, N-cadherin, and vimentin in transformed breast epithelial cells of MCF10A [MCF-10A (T)] overexpressing vector (control) or NRF1 treated with DMSO and E2. (D) Differentiation of NRF1 tumor initiating stem cells to chondrocytes, neurons, and smooth muscle cells compared to the vector. The vector did not show any differentiation to other cells. Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/30486409), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse Vimentin by Immunohistochemistry
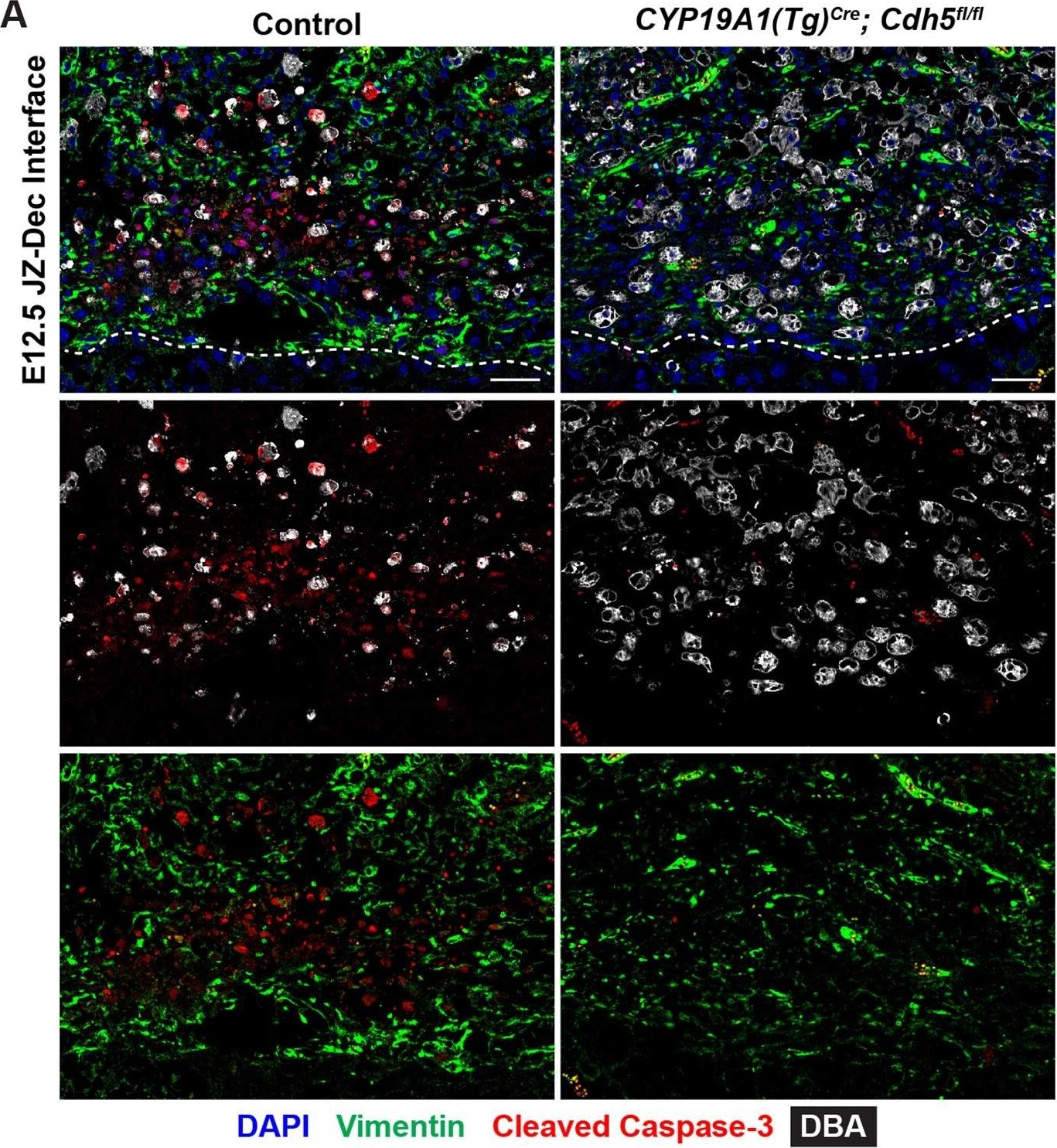
Persistent uterine natural killer (uNK) cells at the junctional zone-decidual interface of Cdh5 knockout placentas.(A) Immunofluorescence staining for DBA (a lectin that specifically binds uterine natural killer (uNK) cells, gray), vimentin (green), and cleaved caspase-3 (red).Dotted line demarcates the border of the junctional zone and decidua. Scale bars = 50 μm. (B, C) Quantification of DBA+ cell density and cleaved caspase-3+ cell density in the decidua. (D) Gene ontology (GO) analysis of upregulated biological process terms related to NK cells. Statistical analysis was performed using two-tailed, unpaired Welch’s t-test. Data are shown as means ± SD.Figure 4—figure supplement 2—source data 1.Excel file containing quantification for uterine natural killer cell density and apoptotic cell density in Figure 4—figure supplement 2.Excel file containing quantification for uterine natural killer cell density and apoptotic cell density in Figure 4—figure supplement 2. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/35486098), licensed under a CC-BY license. Not internally tested by R&D Systems.Applications for Human/Mouse/Rat Vimentin Antibody
Application
Recommended Usage
Immunocytochemistry
5-15 µg/mL
Sample: Immersion fixed A549 human lung carcinoma cell line untreated or treated with 10 ng/ML TGF-beta (Catalog # 240-B) for 48 hours, and immersion fixed HeLa human cervical epithelial carcinoma cell line
Sample: Immersion fixed A549 human lung carcinoma cell line untreated or treated with 10 ng/ML TGF-beta (Catalog # 240-B) for 48 hours, and immersion fixed HeLa human cervical epithelial carcinoma cell line
Immunohistochemistry
5-15 µg/mL
Sample: Immersion fixed paraffin-embedded sections of human skin
Sample: Immersion fixed paraffin-embedded sections of human skin
Knockout Validated
Vimentin is specifically detected in K562 human chronic myelogenous leukemia cell line but is not detectable in Vimentin knockout K562 cell line.
Simple Western
10 µg/mL
Sample: K562 human chronic myelogenous leukemia cell line and Jurkat human acute T cell leukemia cell line
Sample: K562 human chronic myelogenous leukemia cell line and Jurkat human acute T cell leukemia cell line
Western Blot
2 µg/mL
Sample: K562 human chronic myelogenous leukemia cell line, MEF mouse embryonic feeder cells, RAW 264.7 mouse monocyte/macrophage cell line, NR8383 rat alveolar macrophage cell line, and Rat‑2 rat embryonic fibroblast cell line
Sample: K562 human chronic myelogenous leukemia cell line, MEF mouse embryonic feeder cells, RAW 264.7 mouse monocyte/macrophage cell line, NR8383 rat alveolar macrophage cell line, and Rat‑2 rat embryonic fibroblast cell line
Reviewed Applications
Read 10 reviews rated 4 using AF2105 in the following applications:
Formulation, Preparation, and Storage
Purification
Antigen Affinity-purified
Reconstitution
Reconstitute at 0.2 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: Vimentin
References
- Omary, M.B. et al. (2006) Trends Biochem. Sci. 31:383.
- Ivaska, J. et al. (2007) Exp. Cell Res. 313:2050.
- Ferrari, S. et al. (1986) Mol. Cell. Biol. 6:3614.
- Sokolova, A.V. et al. (2006) Proc. Natl. Acad. Sci. USA 103:16206.
- Eriksson, J.E. et al. (2004) J. Cell Sci. 117:919.
- Li, Q.-F. et al. (2006) J. Biol. Chem. 281:34716.
- Esue, O. et al. (2006) J. Biol. Chem. 281:30393.
- Styers, M.L. et al. (2005) Traffic 6:359.
- McInroy, L. and A. Maata (2007) Biochem. Biophys. Res. Commun. 360:109.
- Nieminen, M. et al. (2006) Nat. Cell Biol. 8:156.
- Ivaska, J. et al. (2005) EMBO J. 24:3834.
- Byun, Y. et al. (2001) Cell Death Differ. 8:443.
- Mor-Vaknin, N. et al. (2003) Nat. Cell Biol. 5:59.
- Zou, Y. et al. (2006) Biochem. Biophys. Res. Commun. 351:625.
- Garg, A. et al. (2006) J. Immunol. 177:6192.
Alternate Names
VIM
Gene Symbol
VIM
UniProt
Additional Vimentin Products
Product Documents for Human/Mouse/Rat Vimentin Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human/Mouse/Rat Vimentin Antibody
For research use only
Citations for Human/Mouse/Rat Vimentin Antibody
Customer Reviews for Human/Mouse/Rat Vimentin Antibody (10)
4 out of 5
10 Customer Ratings
Have you used Human/Mouse/Rat Vimentin Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
Showing
1
-
5 of
10 reviews
Showing All
Filter By:
-
Application: Simple WesternSample Tested: fibroblastsSpecies: HumanVerified Customer | Posted 08/28/2024Vimentin in HPFHPF treated with cigarette smoke extract
-
Application: Western BlotSample Tested: HepG2 human hepatocellular carcinoma cell lineSpecies: HumanVerified Customer | Posted 12/20/2022Ab concentration 2 ug/mL, no specific band detectedBio-Techne ResponseThank you for reviewing our product. We are sorry to hear that this product did not perform as expected. We have been in touch with the customer to resolve this issue according to our Product Guarantee and to the customer’s satisfaction.
-
Application: Immunocytochemistry/ImmunofluorescenceSample Tested: Cancer TissueSpecies: MouseVerified Customer | Posted 07/02/2021
-
Application: ImmunohistochemistrySample Tested: Pancreatic cancer tissueSpecies: MouseVerified Customer | Posted 02/20/2021
-
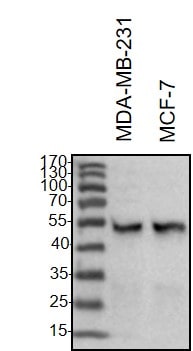
Application: Western BlotSample Tested: Breast cancer cellsSpecies: HumanVerified Customer | Posted 02/20/2021Western Blot: MDA-MB-231 and MCF-7 whole cell lysates were loaded with 30 ug/lane. 10% SDS-PAGE. Vimentin Antibody (AF2105) primary antibody: 1:1000, 4℃, overnight.
-
Application: Immunocytochemistry/ImmunofluorescenceSample Tested: Pancreas tissueSpecies: MouseVerified Customer | Posted 06/27/2018
-
Application: Immunocytochemistry/ImmunofluorescenceSample Tested: Bladder tissueSpecies: HumanVerified Customer | Posted 12/19/2017Bio-Techne ResponseTechnical Service will be following up. The customer followed up reporting the poor staining may have been caused by storing the product outside of our guarantee.
-
Application: Immunocytochemistry/ImmunofluorescenceSample Tested: Kidney cancer tissueSpecies: HumanVerified Customer | Posted 12/10/2017
-
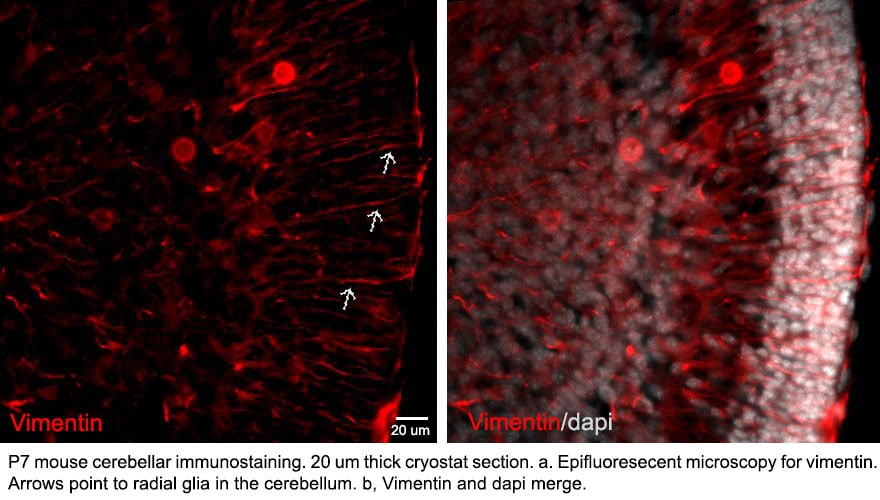
Application: Immunocytochemistry/ImmunofluorescenceSample Tested: Cerebellum tissueSpecies: MouseVerified Customer | Posted 07/22/2016P7 MOUSE CEREBELLUM. Radial Glia
-
Application: ImmunofluorescenceSample Tested: See PMID 23658023Species: HumanVerified Customer | Posted 01/06/2015
There are no reviews that match your criteria.
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- ICC Cell Smear Protocol for Suspension Cells
- ICC Immunocytochemistry Protocol Videos
- ICC for Adherent Cells
- IHC Sample Preparation (Frozen sections vs Paraffin)
- Immunocytochemistry (ICC) Protocol
- Immunocytochemistry Troubleshooting
- Immunofluorescence of Organoids Embedded in Cultrex Basement Membrane Extract
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Fluorescent ICC Staining of Cell Smears - Graphic
- Protocol for the Fluorescent ICC Staining of Cultured Cells on Coverslips - Graphic
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent ICC Staining of Cells on Coverslips
- Protocol for the Preparation and Fluorescent ICC Staining of Non-adherent Cells
- Protocol for the Preparation and Fluorescent ICC Staining of Stem Cells on Coverslips
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- Protocol for the Preparation of a Cell Smear for Non-adherent Cell ICC - Graphic
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Immunohistochemistry
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars