Endoglin (CD105) is a 90 kDa type I transmembrane glycoprotein of the zona pellucida (ZP) family of proteins (1-3). Endoglin and Betaglycan/T beta RIII are type III receptors for TGF beta superfamily ligands, sharing 71% aa identity in the transmembrane (TM) and cytoplasmic domains. Endoglin is highly expressed on proliferating vascular endothelial cells, chondrocytes, and syncytiotrophoblasts of term placenta, with lower amounts on hematopoietic, mesenchymal and neural crest stem cells, activated monocytes, and lymphoid and myeloid leukemic cells (2-5). Human endoglin cDNA encodes 658 amino acids including a 25 amino acid (aa) signal sequence, a 561 aa extracellular domain (ECD) with an orphan domain and a two-part ZP domain, a TM domain, and a 47 aa cytoplasmic domain (1-3). An isoform with a 14 aa cytoplasmic domain (S-endoglin) can oppose effects of long (L) endoglin (6, 7). The human endoglin ECD shares 65-72% aa identity with mouse, rat, bovine, porcine, and canine endoglin. Endoglin homodimers interact with TGF-beta 1 and TGF-beta 3 (but not TGF-beta 2) but only after binding T beta RII (8). Similarly, they interact with activin-A and BMP-7 via activin type IIA or B receptors, and with BMP-2 via BMPR-1A/ALK-3 or BMPR-1B/ALK-6 (9). BMP-9, however, is reported to bind endoglin directly (10). Endoglin modifies ligand-induced signaling in multiple ways. For example, expression of endoglin can inhibit TGF-beta 1 signals but enhance BMP7 signals in the same myoblast cell line (11). In endothelial cells, endoglin inhibits T beta RI/ALK5, but enhances ALK1-mediated activation (12). Deletion of mouse endoglin causes lethal vascular and cardiovascular defects, and human endoglin haploinsufficiency can a cause the vascular disorder, hereditary hemorrhagic telangiectasia type I (13, 14). These abnormalities confirm the essential function of endoglin in differentiation of smooth muscle, angiogenesis, and neovascularization (2-4, 12-14). In preeclampsia of pregnancy, high levels of proteolytically generated soluble endoglin and VEGF R1 (sFlt-1), along with low Placental Growth Factor (PlGF), are pathogenic due to antiangiogenic activity (15).

Key Product Details
Validated by
Knockout/Knockdown, Biological Validation
Species Reactivity
Validated:
Human
Cited:
Human, Mouse
Applications
Validated:
Knockout Validated, Immunohistochemistry, Western Blot, Flow Cytometry, Immunocytochemistry, Simple Western, CyTOF-ready
Cited:
Immunohistochemistry, Immunohistochemistry-Paraffin, Western Blot, Flow Cytometry, Immunocytochemistry, Immunoprecipitation, IHC-
Label
Unconjugated
Antibody Source
Polyclonal Goat IgG
Loading...
Product Specifications
Immunogen
Mouse myeloma cell line NS0-derived recombinant human Endoglin/CD105
Glu26-Gly586
Accession # Q5T9B9
Glu26-Gly586
Accession # Q5T9B9
Specificity
Detects human Endoglin in direct ELISAs and Western blots.
Clonality
Polyclonal
Host
Goat
Isotype
IgG
Scientific Data Images for Human Endoglin/CD105 Antibody
Detection of Human Endoglin/CD105 by Western Blot.
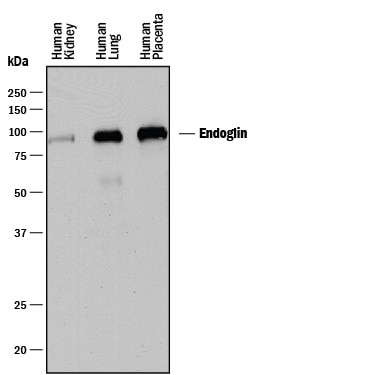
Western blot shows lysates of human kidney tissue, human lung tissue, human placenta tissue. PVDF membrane was probed with 0.25 µg/mL of Goat Anti-Human Endoglin/CD105 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF1097) followed by HRP-conjugated Anti-Goat IgG Secondary Antibody (HAF017). A specific band was detected for Endoglin/CD105 at approximately 90 kDa (as indicated). This experiment was conducted under reducing conditions and using Immunoblot Buffer Group 1.Endoglin/CD105 in U937 Human Cell Line.
Endoglin/CD105 was detected in immersion fixed U937 human histiocytic lymphoma cell line using Goat Anti-Human Endoglin/CD105 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF1097) at 10 µg/mL for 3 hours at room temperature. Cells were stained using the NorthernLights™ 557-conjugated Anti-Goat IgG Secondary Antibody (red; NL001) and counterstained with DAPI (blue). View our protocol for Fluorescent ICC Staining of Cells on Coverslips.Endoglin/CD105 in Human Prostate.
Endoglin/CD105 was detected in immersion fixed paraffin-embedded sections of human prostate using Goat Anti-Human Endoglin/CD105 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF1097) at 1.7 µg/mL overnight at 4 °C. Tissue was stained using the Anti-Goat HRP-DAB Cell & Tissue Staining Kit (brown; CTS008) and counterstained with hematoxylin (blue). View our protocol for Chromogenic IHC Staining of immersion fixed paraffin-embedded Tissue Sections.Endoglin/CD105 in Mouse Kidney.
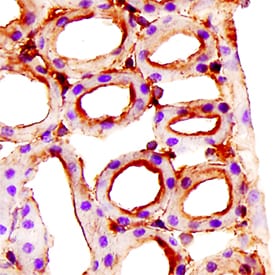
Endoglin/CD105 was detected in immersion fixed paraffin-embedded sections of mouse kidney using Goat Anti-Human Endoglin/CD105 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF1097) at 15 µg/mL overnight at 4 °C. Tissue was stained using the Anti-Goat HRP-DAB Cell & Tissue Staining Kit (brown; CTS008) and counterstained with hematoxylin (blue). Specific staining was localized to plasma membranes in tubules. View our protocol for Chromogenic IHC Staining of Paraffin-embedded Tissue Sections.Detection of Human Endoglin/CD105 by Simple WesternTM.
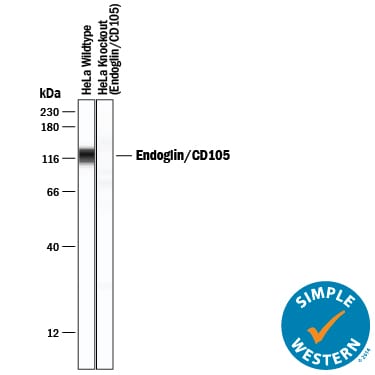
Simple Western lane view shows lysates of HeLa human cervical epithelial carcinoma parental cell line and Endoglin/CD105 knockout HeLa cell line (KO), loaded at 0.2 mg/mL. A specific band was detected for Endoglin/CD105 at approximately 121 kDa (as indicated) in the parental HeLa cell line, but is not detectable in knockout HeLa cell line. Goat Anti-Human Endoglin/CD105 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF1097) was used at 20 µg/mL followed by 1:50 dilution of HRP-conjugated Anti-Goat IgG Secondary Antibody (HAF019). This experiment was conducted under reducing conditions and using the 12-230 kDa separation system.Western Blot Shows Human Endoglin/CD105 Specificity by Using Knockout Cell Line.
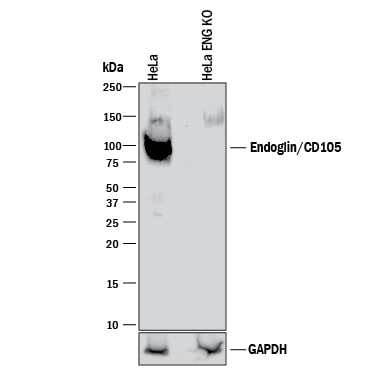
Western blot shows lysates of HeLa human cervical epithelial carcinoma parental cell line and Endoglin/CD105 knockout HeLa cell line (KO). PVDF membrane was probed with 0.25 µg/mL of Goat Anti-Human Endoglin/CD105 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF1097) followed by HRP-conjugated Anti-Goat IgG Secondary Antibody (HAF017). A specific band was detected for Endoglin/CD105 at approximately 90 kDa (as indicated) in the parental HeLa cell line, but is not detectable in knockout HeLa cell line. GAPDH (AF5718) is shown as a loading control. This experiment was conducted under reducing conditions and using Immunoblot Buffer Group 1.Detection of Human Endoglin/CD105 by Immunocytochemistry/Immunofluorescence
Overview of Multi-dimensional Microscopic Molecular Profiling (MMMP).The overall MMMP approach is depicted using an example tissue section from normal human duodenum (sample #1.9.7). (a) Slides were subjected to repeated cycles of staining and imaging with fluorescent primary antibodies and DAPI. At the end of each cycle, fluorescent signal was removed by a chemical bleaching process, and slides were again imaged, before proceeding to the next round of this iterative procedure. After the final antibody stain (#15 Sma), slides were analyzed with a series of histochemical stains. (b) A set of tiling images spanning each tissue section was initially generated by the microscope system. The tiling images were then computationally ‘stitched’ together to produce a single image per staining cycle for each sample. (c) Image registration was performed to align images from the same tissue section across cycles. Mean intensities of the DAPI signal from all immuno-fluorescence images are shown from before (Unregistered) and after (Registered) the image registration procedure was completed. (d) Following registration, signal intensities from the relevant channels for each image (columns) in the MMMP series were extracted for each pixel (rows) within the tissue section and compiled into a large data matrix of in situ molecular profiles. Image collected and cropped by CiteAb from the following publication (https://dx.plos.org/10.1371/journal.pone.0128975), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human Endoglin/CD105 by Flow Cytometry
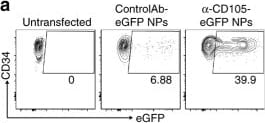
Transfection of stem cells with mRNA nanoparticles can promote their expansion and self-renewal. a Targeting of CD105 enables specific transfection of HSC CD34+ cells. Cells were left untreated, or transfected with eGFP-encoding mRNA in nanoparticles (NPs) coated with PGA coupled to a control antibody or anti-CD105. Transfection efficiency was assayed by flow cytometry after 24 h. b NP transfection efficiency in CD34+ samples from three independent donors. Viability is shown in c. d Expansion of CD34 + PBSCs after NP-transfection. e Phenotypical characterization of PBSC-derived CD34+ subpopulations after 2 days in culture. Cells were either transfected with eGFP NPs on day 1 or left unmodified. Gating is indicated in brackets on top of each column. f Summary bar graph showing mean frequencies and SE of primitive Hematopoietic Stem Cells (HSCs), Multipotent Progenitors (MPPs), Lymphoid-primed Multipotent Progenitors (LMPs), and Early Myeloid Progenitors (EMPs). PBSCs from four independent donors were analyzed. Error bars represent mean ± SE. g Colony output of sort-purified GFP-NP transfected versus unmodified CD34+ cells from day 7 cultures (n = 3 cultures from independent donors); n.s., non-significant. Arising colonies were identified as colony forming unit (CFU) granulocyte (CFU-G), macrophage (CFU-M), granulocyte-macrophage (CFU-GM) and burst forming unit-erythrocyte (BFU-E). Colonies consisting of erythroid and myeloid cells were scored as CFU-MIX; n.s., non-significant. Error bars represent mean ± SE. h Representative images of CFU-MIX colonies from untransfected and GFP-NP-transfected CD34+ cells (×4-magnification; scale bar 1000 µm). i qPCR measurements of NP-delivered Musashi-2 (MSI2) mRNA expression over time. Error bars represent mean ± SE. j Comparison of CD133 and CD34 expression in HSCs transfected with control GFP mRNA-NPs versus MSI2 mRNA-NPs, assessed by flow cytometry 8 days after NP exposure and cell expansion. Data represent two independent experiments conducted in triplicate. k Cellular fold expansion of CD34− (differentiated) and CD34+ CD133+ (progenitor) cells. Bar graphs show mean and SE of three independent experiments. Data represent two independent experiments conducted in triplicate. l Colony forming unit outputs of untransfected versus MSI2-NP-transfected HSCs (n = 3 cultures from independent donors); Pairwise differences between groups were analyzed with the unpaired, two-tailed Student’s t Test. *P = 0.049, **P = 0.012, ***P = 0.011; n.s., non-significant. Error bars represent mean ± SE. m Representative images of colonies from untransfected and MSI2-NP-transfected CD34+ cells (scale bar, 300 µm) Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/28855514), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human Endoglin/CD105 by Immunocytochemistry/Immunofluorescence
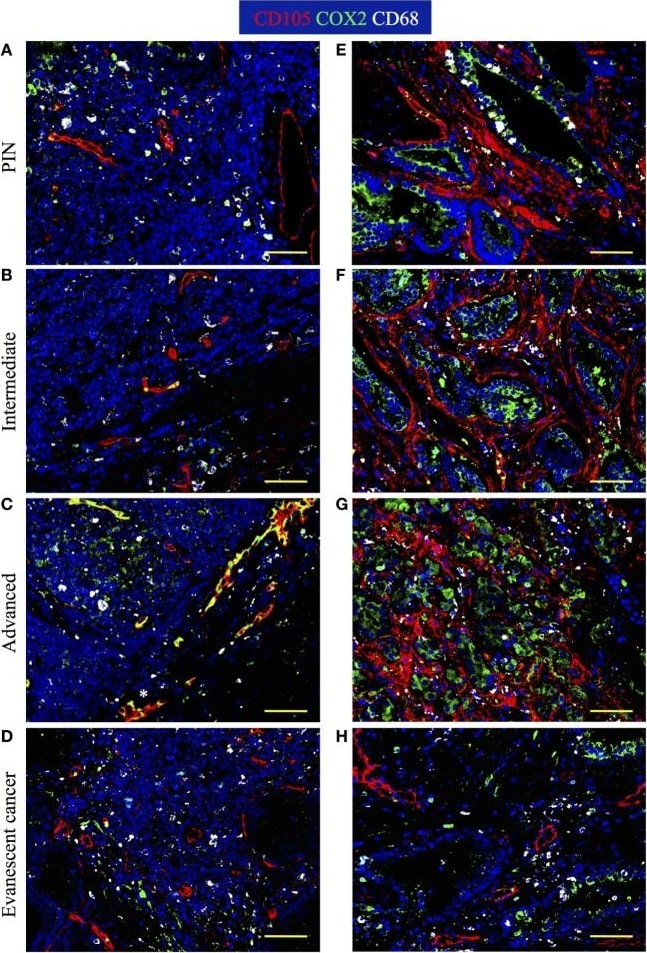
Spatiotemporal changes in tumor vascularization and COX2 coverage at different stages of prostate cancer progression/regression. The 5-μm thick paraffin serial sections were stained with antibodies against endoglin (CD105, red), cyclooxygenase 2 (COX2, green), and CD68 (white). (A–D) Representative 200× pictures are showing changes in vascularization and COX2 expression in tertiary lymphoid organs (TLO), or (E–H) tumor areas. (A) CD105+ vessels, CD68+COX2+ macrophages, and CD68−COX2+ cells are detected in TLO from prostatic intraepithelial neoplasia (PIN) patients. (B) Smaller CD105+ vessels and CD68−COX2+ cells are located on the border of TLO from intermediate prostate cancer patient. (C) In a prostatectomy specimen from advanced carcinoma patient, CD105+ vessels with cuboidal morphology are located outside TLO, while numerous CD68−COX2+ cells are found in the center of a TLO. (D) Oval-shaped and spindle-shaped COX2+ cells are located on the border of a TLO. (E) Vascularity and COX2+ epithelial cells are modestly increased in tumor areas from PIN patients. (F,G) Abundant blood vessels with abnormal morphology and aberrant organization were located in close proximity to strongly labeled COX2+ transformed epithelium in tumors of intermediate and advanced prostate cancer patients. (D,H) Preserved vascular morphology and organization, as well as reduced COX2+ density were observed in TLO and tumor areas from patients with spontaneous prostate cancer regression. CD105+ vessels with a high endothelial venule-like morphology were detected in TLO from patients at late stages of prostate cancer (white asterisk). Representative 200× magnification pictures of TLO and tumor areas were taken with a Zeiss Axioplan Microscope and recorded with a Hamamatsu Camera. Scale bars represent 100 μm. Image collected and cropped by CiteAb from the following publication (https://journal.frontiersin.org/article/10.3389/fimmu.2017.00563/full), licensed under a CC-BY license. Not internally tested by R&D Systems.Applications for Human Endoglin/CD105 Antibody
Application
Recommended Usage
CyTOF-ready
Ready to be labeled using established conjugation methods. No BSA or other carrier proteins that could interfere with conjugation.
Flow Cytometry
0.25 µg/106 cells
Sample: U937 human histiocytic lymphoma cell line
Sample: U937 human histiocytic lymphoma cell line
Immunocytochemistry
5-15 µg/mL
Sample: Immersion fixed U937 human histiocytic lymphoma cell line
Sample: Immersion fixed U937 human histiocytic lymphoma cell line
Immunohistochemistry
5-15 µg/mL
Sample: Immersion fixed paraffin-embedded sections of human kidney cancer tissue, human prostate, and mouse kidney
Sample: Immersion fixed paraffin-embedded sections of human kidney cancer tissue, human prostate, and mouse kidney
Knockout Validated
Endoglin/CD105
is specifically detected in HeLa human cervical epithelial carcinoma parental cell line but is not detectable in
Endoglin/CD105 knockout HeLa cell line.
Simple Western
20 µg/mL
Sample: HeLa human cervical epithelial carcinoma cell line
Sample: HeLa human cervical epithelial carcinoma cell line
Western Blot
0.25 µg/mL
Sample: Human kidney tissue, human lung tissue, human placenta tissue
Sample: Human kidney tissue, human lung tissue, human placenta tissue
Reviewed Applications
Read 2 reviews rated 4.5 using AF1097 in the following applications:
Flow Cytometry Panel Builder
Bio-Techne Knows Flow Cytometry
Save time and reduce costly mistakes by quickly finding compatible reagents using the Panel Builder Tool.
Advanced Features
- Spectra Viewer - Custom analysis of spectra from multiple fluorochromes
- Spillover Popups - Visualize the spectra of individual fluorochromes
- Antigen Density Selector - Match fluorochrome brightness with antigen density
Formulation, Preparation, and Storage
Purification
Antigen Affinity-purified
Reconstitution
Reconstitute at 0.2 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: Endoglin/CD105
References
- Gougos, A. and Letarte, M. (1990) J. Biol. Chem. 265:8361.
- ten Dijke, P. et al. (2008) Angiogenesis 11:79.
- Bernabeu, C. et al. (2007) J. Cell. Biochem. 102:1375.
- Mancini, M.L. et al. (2007) Dev. Biol. 308:520.
- Moody, J.L. et al. (2007) Stem Cells 25:2809.
- Velasco, S. et al. (2008) J. Cell Sci. 121:913.
- Perez-Gomez, E. et al. (2005) Oncogene 24:4450.
- Cheifetz, S, et al. (1992) J. Biol. Chem. 267:19027.
- Barbara, N.P. et al. (1999) J. Biol. Chem. 274:584.
- Scharpfenecker, M. et al. (2007) J. Cell Sci. 120:964.
- Scherner, O. et al. (2007) J. Biol. Chem. 282:13934.
- Pece-Barbara, N. et al. (2005) J. Biol. Chem. 280:27800.
- Arthur, H.M. et al. (2000) Dev. Biol. 217:42.
- Lebrin, F. and C.L. Mummery (2008) Trends Cardiovasc. Med. 18:25.
- Venkatesha, S. et al. (2006) Nat. Med. 12:642.
Alternate Names
CD105, ENG
Gene Symbol
ENG
UniProt
Additional Endoglin/CD105 Products
Product Documents for Human Endoglin/CD105 Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human Endoglin/CD105 Antibody
For research use only
Citations for Human Endoglin/CD105 Antibody
Customer Reviews for Human Endoglin/CD105 Antibody (2)
4.5 out of 5
2 Customer Ratings
Have you used Human Endoglin/CD105 Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
Showing
1
-
2 of
2 reviews
Showing All
Filter By:
-
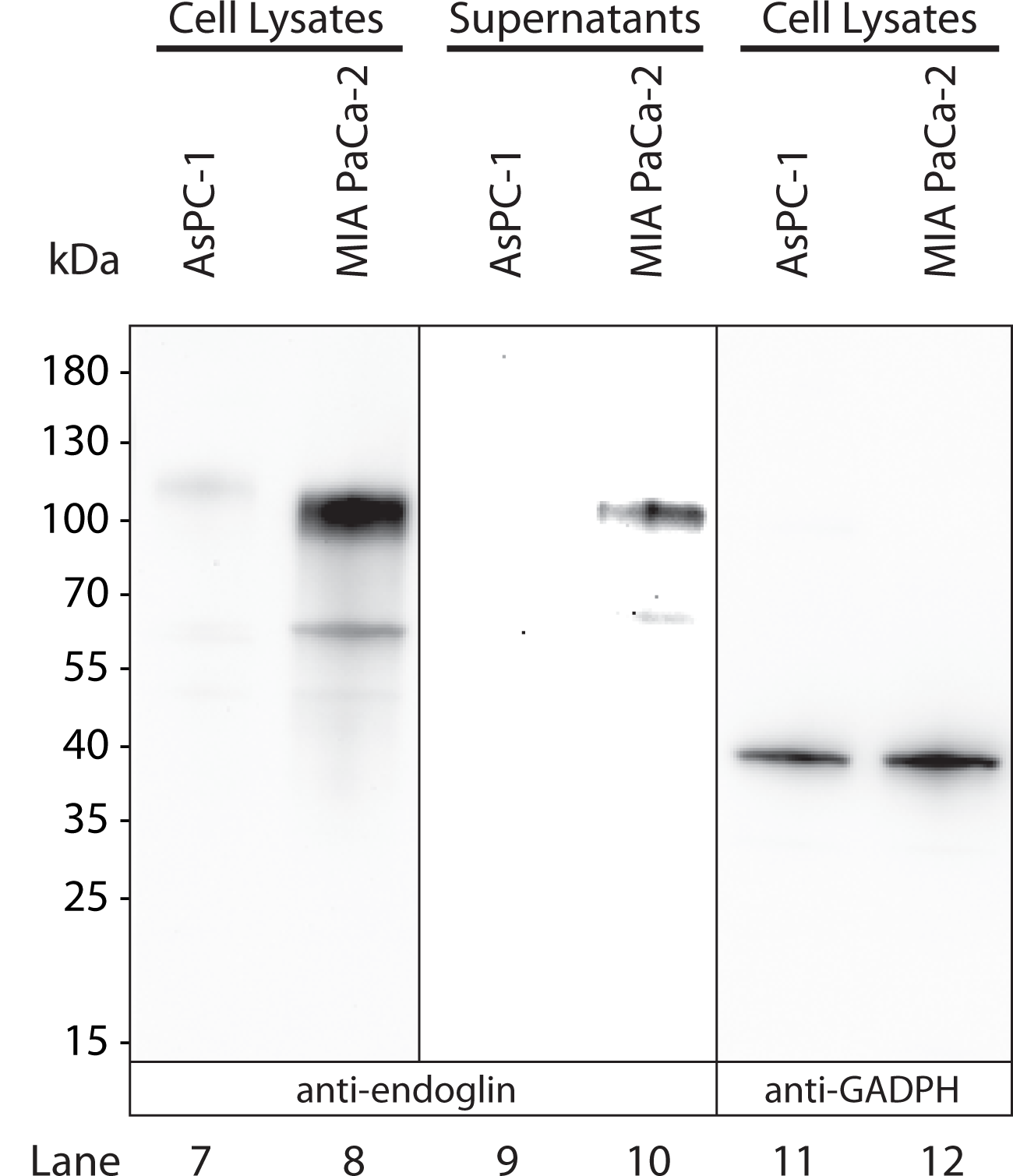
Application: Western BlotSample Tested: Pancreatic cancer cellsSpecies: HumanVerified Customer | Posted 12/11/2019Lysates and supernatants of pancreatic cancer cells AsPC-1 and MIA PaCa-2 were examined under denaturing/reducing conditions by SDS-PAGE/western blots and subsequent antibody stainings. Nitrocellulose membrane was probed with 0.13 ug/ml. This antibody stains endoglin as expected.
-
Application: ImmunofluorescenceSample Tested: See PMID 23623496Species: HumanVerified Customer | Posted 01/05/2015
There are no reviews that match your criteria.
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- 7-Amino Actinomycin D (7-AAD) Cell Viability Flow Cytometry Protocol
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Extracellular Membrane Flow Cytometry Protocol
- Flow Cytometry Protocol for Cell Surface Markers
- Flow Cytometry Protocol for Staining Membrane Associated Proteins
- Flow Cytometry Staining Protocols
- Flow Cytometry Troubleshooting Guide
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- ICC Cell Smear Protocol for Suspension Cells
- ICC Immunocytochemistry Protocol Videos
- ICC for Adherent Cells
- IHC Sample Preparation (Frozen sections vs Paraffin)
- Immunocytochemistry (ICC) Protocol
- Immunocytochemistry Troubleshooting
- Immunofluorescence of Organoids Embedded in Cultrex Basement Membrane Extract
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Intracellular Flow Cytometry Protocol Using Alcohol (Methanol)
- Intracellular Flow Cytometry Protocol Using Detergents
- Intracellular Nuclear Staining Flow Cytometry Protocol Using Detergents
- Intracellular Staining Flow Cytometry Protocol Using Alcohol Permeabilization
- Intracellular Staining Flow Cytometry Protocol Using Detergents to Permeabilize Cells
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Propidium Iodide Cell Viability Flow Cytometry Protocol
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Liperfluo
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Characterization of Human Th22 Cells
- Protocol for the Characterization of Human Th9 Cells
- Protocol for the Fluorescent ICC Staining of Cell Smears - Graphic
- Protocol for the Fluorescent ICC Staining of Cultured Cells on Coverslips - Graphic
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent ICC Staining of Cells on Coverslips
- Protocol for the Preparation and Fluorescent ICC Staining of Non-adherent Cells
- Protocol for the Preparation and Fluorescent ICC Staining of Stem Cells on Coverslips
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- Protocol for the Preparation of a Cell Smear for Non-adherent Cell ICC - Graphic
- Protocol: Annexin V and PI Staining by Flow Cytometry
- Protocol: Annexin V and PI Staining for Apoptosis by Flow Cytometry
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Fluorokine Flow Cytometry Kits
- Troubleshooting Guide: Immunohistochemistry
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars