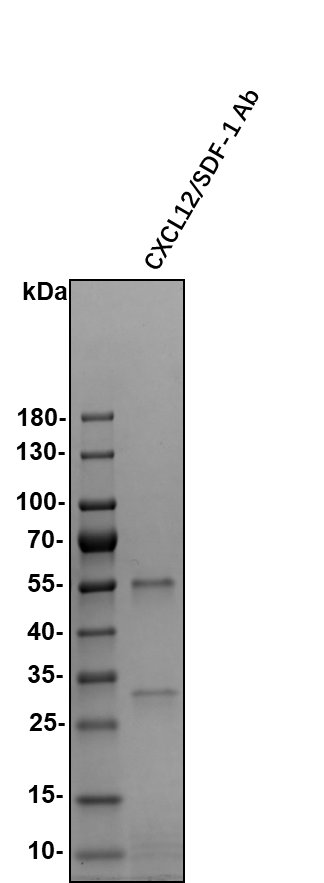
CXCL12, also known as SDF-1, is a CXC chemokine that interacts with CXCR4. SDF-1 alpha and SDF-1 beta are alternatively spliced isoforms of CXCL12. SDF-1 beta has an additional 4 amino acids at the carboxy-terminus.
Human/Mouse CXCL12/SDF‑1 Antibody
R&D Systems | Catalog # MAB350

Key Product Details
Validated by
Orthogonal Validation
Species Reactivity
Validated:
Human, Mouse
Cited:
Human, Mouse, Rat, Canine, Feline, Rabbit, Transgenic Mouse, Xenograft
Applications
Validated:
Multiplex Immunofluorescence, Immunohistochemistry, ELISA Capture (Matched Antibody Pair), Intracellular Staining by Flow Cytometry, Dual RNAscope ISH-IHC Compatible, Immunocytochemistry, COMET, CyTOF-ready
Cited:
Immunohistochemistry, Immunohistochemistry-Paraffin, Immunohistochemistry-Frozen, Western Blot, Neutralization, Flow Cytometry, Immunocytochemistry, CyTof, ELISA Capture, ELISA Development, ELISA Development (Capture), ELISA Development (Detection), In vivo assay, Functional Assay, Luminex Development, Mass Spectrometry
Label
Unconjugated
Antibody Source
Monoclonal Mouse IgG1 Clone # 79018
Loading...
Product Specifications
Immunogen
E. coli-derived recombinant human CXCL12/SDF‑1 alpha
Lys22-Lys89
Accession # Q6ICW0
Lys22-Lys89
Accession # Q6ICW0
Specificity
Detects human and mouse CXCL12/SDF-1 alpha and CXCL12/SDF-1 beta in ELISAs.
Clonality
Monoclonal
Host
Mouse
Isotype
IgG1
Scientific Data Images for Human/Mouse CXCL12/SDF‑1 Antibody
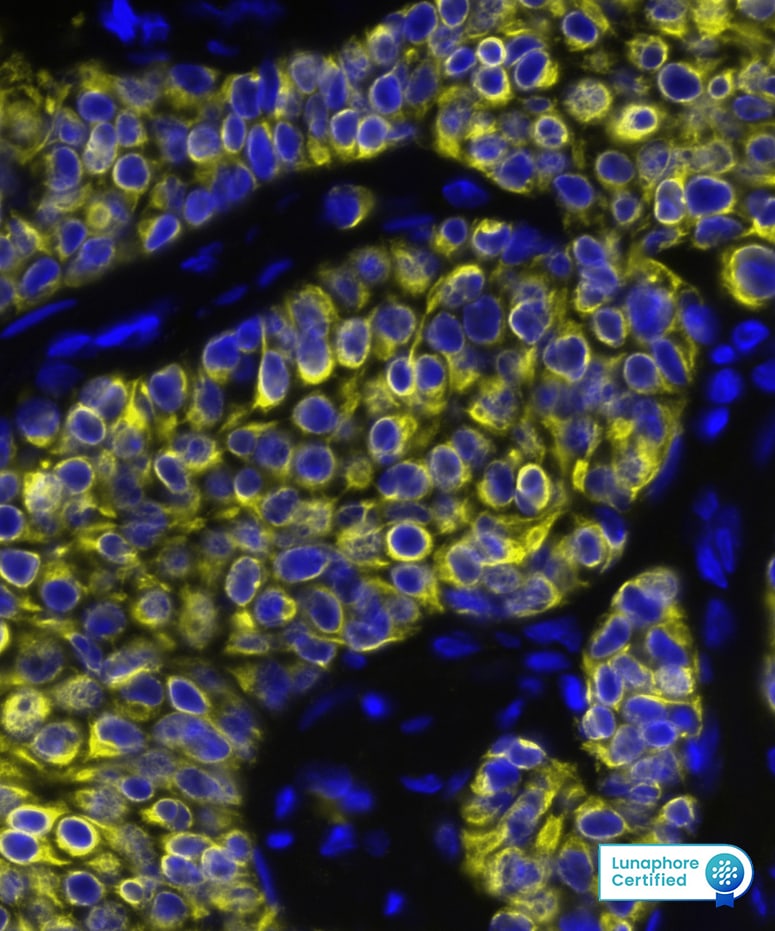
CXCL12/SDF-1 in Human breast tumor via seqIF™ staining on COMET™
CXCL12/SDF-1 was detected in immersion fixed paraffin-embedded sections of human breast tumor using Mouse Anti-Human CXCL12/SDF-1, Monoclonal Antibody (Catalog #MAB350) at 15ug/mL at 37° Celsius for 4 minutes. Before incubation with the primary antibody, tissue underwent an all-in-one dewaxing and antigen retrieval preprocessing using PreTreatment Module (PT Module) and Dewax and HIER Buffer H (pH 9; Epredia Catalog # TA-999-DHBH). Tissue was stained using the Alexa Fluor™ 647 Goat anti-Mouse IgG Secondary Antibody at 1:200 at 37 ° Celsius for 2 minutes. (Yellow; Lunaphore Catalog # DR647MS) and counterstained with DAPI (blue; Lunaphore Catalog # DR100). Specific staining was localized to the cytoplasm. Protocol available in COMET™ Panel Builder.CXCL12/SDF‑1 in Human PBMCs and K562 Human Cell Line.
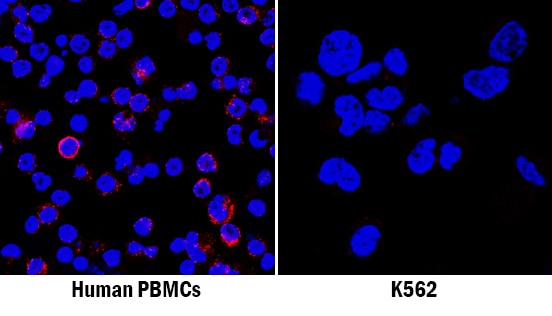
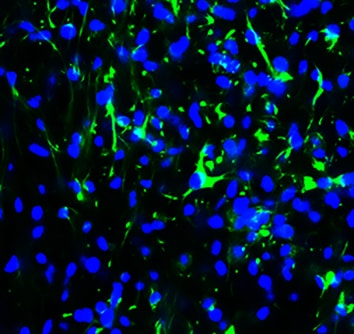
CXCL12/SDF-1 was detected in immersion fixed human peripheral blood mononuclear cells (PBMCs; positive staining) and K562 human chronic myelogenous leukemia cell line (negative staining) using Mouse Anti-Human/Mouse CXCL12/SDF-1 Monoclonal Antibody (Catalog # MAB350) at 8 µg/mL for 3 hours at room temperature. Cells were stained using the NorthernLights™ 557-conjugated Anti-Mouse IgG Secondary Antibody (red; Catalog # NL007) and counterstained with DAPI (blue). Specific staining was localized to cell membranes. View our protocol for Fluorescent ICC Staining of Non-adherent Cells.CXCL12/SDF‑1 in Human Tonsil.
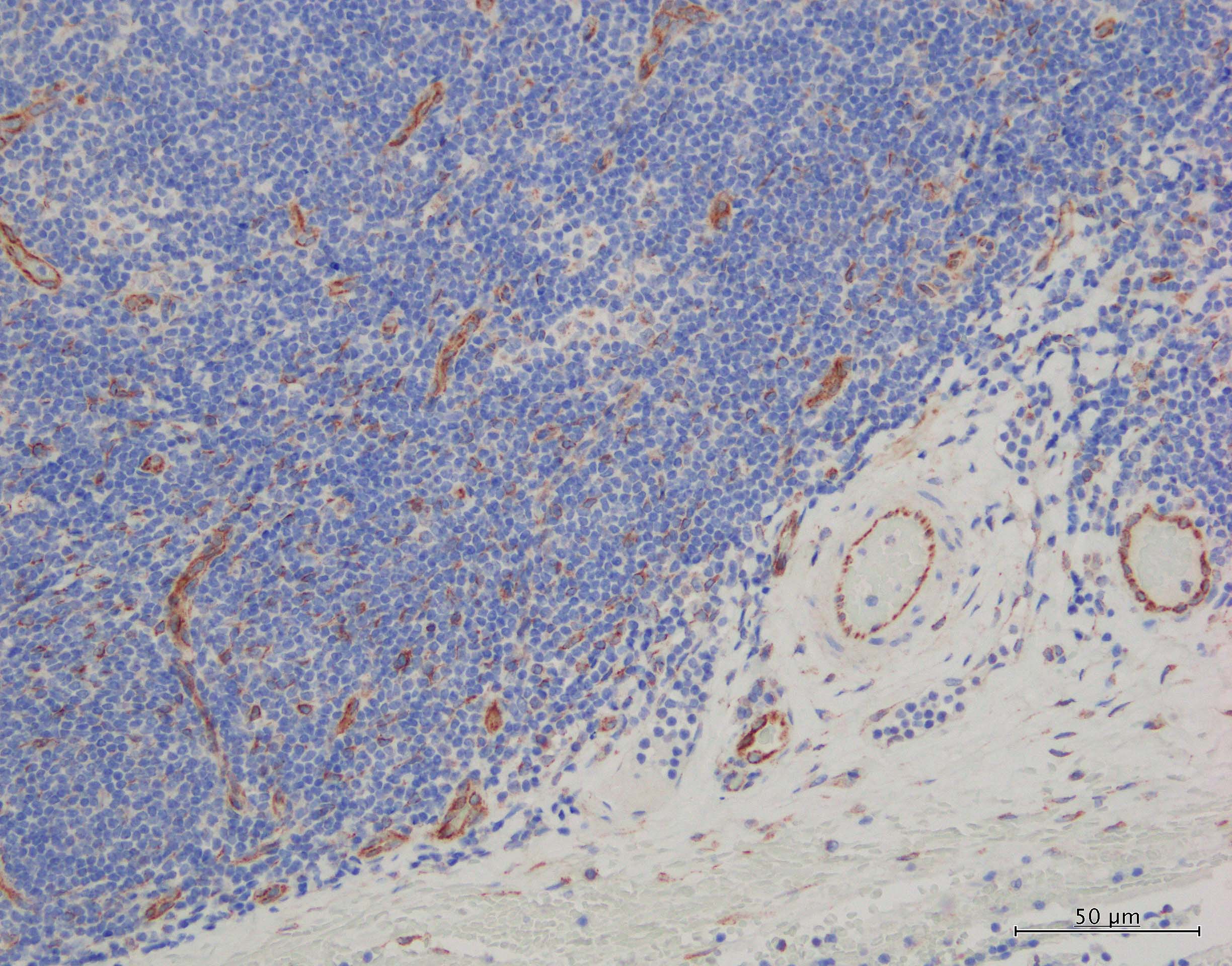
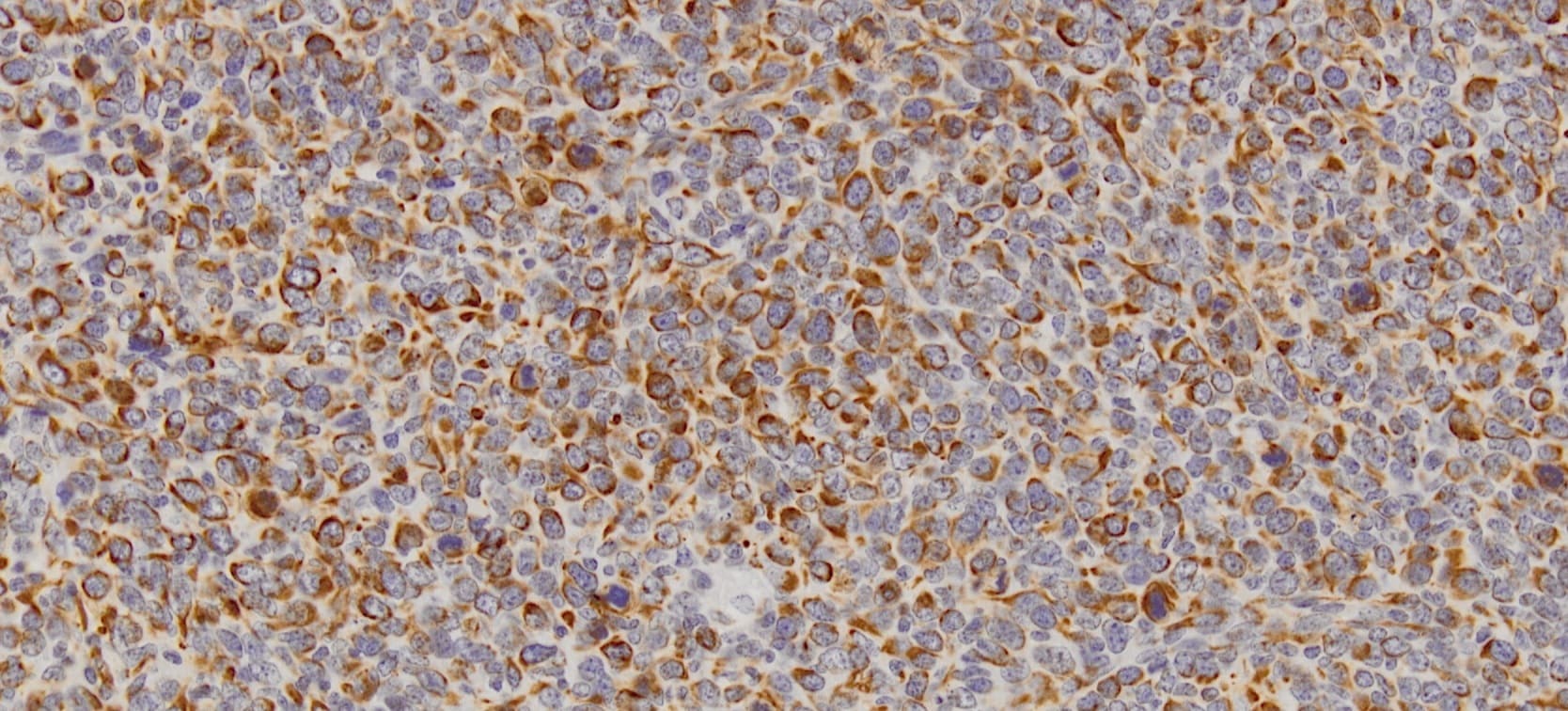
CXCL12/SDF‑1 was detected in immersion fixed paraffin-embedded sections of human tonsil using 15 µg/mL Human/Mouse CXCL12/SDF‑1 Monoclonal Antibody (Catalog # MAB350) overnight at 4 °C. Tissue was stained with the Anti-Mouse HRP-DAB Cell & Tissue Staining Kit (brown; Catalog # CTS002) and counterstained with hematoxylin (blue). Specific labeling was localized to epithelial cells in cripts. View our protocol for Chromogenic IHC Staining of Paraffin-embedded Tissue Sections.CXCL12/SDF‑1 in Human Tonsil.
CXCL12/SDF‑1 was detected in immersion fixed paraffin-embedded sections of human tonsil using Human/Mouse CXCL12/SDF‑1 Monoclonal Antibody (Catalog # MAB350) at 25 µg/mL overnight at 4 °C. Tissue was stained using the Anti-Mouse HRP-DAB Cell & Tissue Staining Kit (brown; Catalog # CTS002) and counterstained with hematoxylin (blue). View our protocol for Chromogenic IHC Staining of Paraffin-embedded Tissue Sections.Detection of CXCL12/SDF‑1 in HT1080 Human Cell Line by Flow Cytometry.
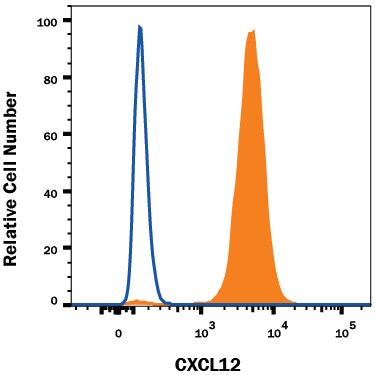
HT1080 human fibrosarcoma cell line was stained with Mouse Anti-Human/Mouse CXCL12/SDF-1 Monoclonal Antibody (Catalog # MAB350, filled histogram) or isotype control antibody (Catalog # MAB002, open histogram), followed by Phycoerythrin-conjugated Anti-Mouse IgG Secondary Antibody (Catalog # F0102B). To facilitate intracellular staining, cells were fixed and permeabilized with FlowX FoxP3 Fixation & Permeabilization Buffer Kit (Catalog # FC012). View our protocol for Staining Intracellular Molecules.CXCL12/SDF‑1 alpha in Human Tonsil Using Dual RNAscope®ISH and IHC.
CXCL12/SDF-1a mRNA was detected in formalin-fixed paraffin-embedded tissue sections of human tonsil probed with ACD RNAScope®Probe (Catalog # 422991) and stained using ACD RNAscope®2.5 HD Detection Reagents-Red (top image, Catalog # 32260). Adjacent tissue section was processed for immunohistochemistry using R&D Systems Mouse Anti-Human CXCL12/SDF-1a Monoclonal Antibody (Catalog # MAB350) at 5 ug/mL for 1 hour at room temperature followed by incubation with the Anti-Mouse IgG VisUCyte HRP Polymer Antibody (R&D Systems, Catalog # VC001) and DAB chromogen (lower image, yellow-brown). Tissues were counterstained with hematoxylin (blue).Detection of Human CXCL12/SDF-1 by Immunocytochemistry/Immunofluorescence
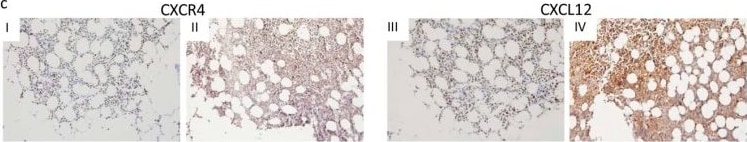
CXCR4 and CXCL12 expression and BM infiltration. (a) Expression analysis of CXCR4 and CXCL12 in uninfiltrated and infiltrated bone marrow specimens at the time of diagnosis by RQ-PCR. (b) Expression analysis of CXCR4 and CXCL12 in infiltrated bone marrow biopsies and the respective paired sample in patients under remission by RQ-PCR. (c) Representative immunohistochemical stains of CXCR4 (I–II) and CXCL12 (III–IV) on selected bone marrow specimens of DLBCL patients (magnification 20×). (I) and (III) represent the CXCR4 and CXCL12 staining of uninfiltrated bone marrow specimens, and (II) and (IV) represent those of the infiltrated bone marrow specimens. mRNA expression levels were calculated as a relative expression in comparison to uninfiltrated bone marrow specimens. Each bar represents the mean values of expression levels ± standard error of the mean (SEM). The comparison of the expression levels was performed by using the Mann–Whitney U-test or the Student’s t-test. All images were captured using an Olympus BX51 microscope and an Olympus E-330 camera. Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/31554271), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse CXCL12/SDF-1 by Immunohistochemistry
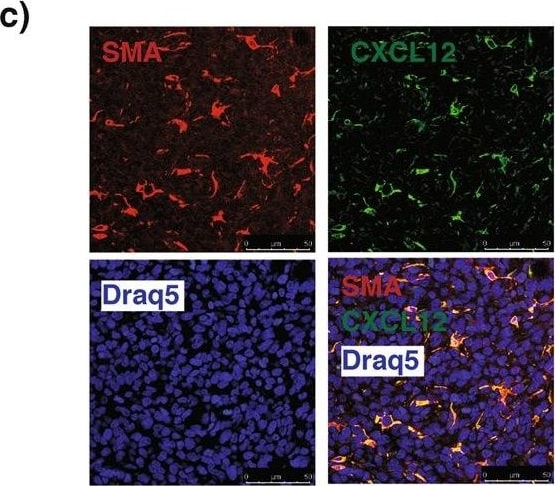
RhoA modulates the CXCR4-CXCL12 axis in breast tumors. (a) Representative immunohistochemical (IHC) staining images of breast primary tumor sections of mice orthotopically injected with 4T1 GFP-LUC cells carrying indicated lentiviral modifications, stained with antibodies for CD31 (top, left), F4/80 (middle, left) and alpha -SMA (bottom, left). CD31+ area (top, right), F4/80+ cells (middle, right) and alpha -SMA+ area (bottom, right) per tumor field of view (FOV) (mean ± SE) quantifications of IHC images. N = 4. (b) Representative IHC staining images of breast primary tumor sections of above mice stained with CXCL12 antibody (left). CXCL12+ area per tumor FOV (mean ± SE) quantification of IHC images. N = 4. (c) Representative immunofluorescent images of breast primary tumor sections of above mice co-stained with alpha -SMA, CXCL12 and DRAQ5 nuclear stain, showing the co-localization of alpha -SMA and CXCL12 signals. Scale bar: 50 µm. (d) Representative IHC staining images of breast primary tumor sections of above mice stained with CXCR4 antibody (left). CXCR4+ area per tumor FOV (mean ± SE) quantification of IHC images. N = 4. (e) Relative mCXCR4 mRNA levels normalized with mActin (mean ± SE), as quantified by qRT-PCR in primary breast tumors of above mice (top) and in 4T1 GFP-LUC cells carrying indicated lentiviral modifications (bottom). N = 4, ns- not significant *P < 0.05, unpaired Student’s t-test (two-tailed). Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/31705019), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human CXCL12/SDF-1 by Immunocytochemistry/Immunofluorescence
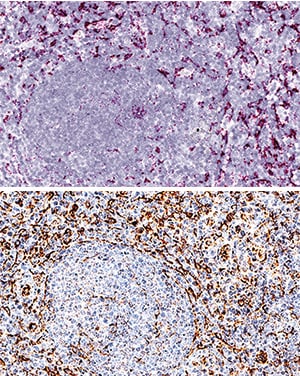
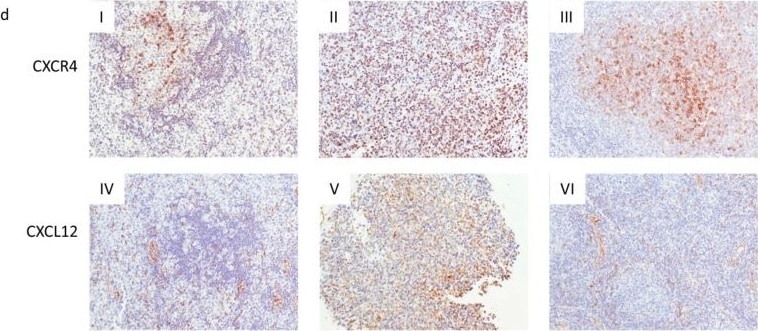
CXCR4 and CXCL12 expression in DLBCL. (a) Expression analysis of CXCR4 and CXCL12 in non-neoplastic control germinal center B cells (GC-B) and diffuse large B cell lymphoma cells (DLBCL) consisting of DLBCL-NGCB and DLCBL-GCB, by RQ-PCR. GCB-DLBCL were further subdivided into primary (DLBCL-pGCB) and transformed DLBCL (DLBCL-pGCB) originating from follicular lymphoma. (b) Expression analysis of CXCR4 and CXCL12 in DLBCL samples with early (stage 1) and advanced stage (stage 2–4) (left graphs) and DLBCL samples with and without bone marrow infiltration (right graphs) by RQ-PCR. (c) Probability of 5-year-survival in DLBCL patients (our cohort left panel and the cohort of Lenz et al. right [20]) stratified by the third quartile of CXCR4 expression, respectively. (d) Representative immunohistochemical stains of CXCR4 (I–III) and CXCL12 (IV–VI) on DLBCL samples (magnification 20×). mRNA expression levels were calculated as a relative expression in comparison to the GC-B cells. All images were captured using an Olympus BX51 microscope and an Olympus E-330 camera. Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/31554271), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human Human/Mouse CXCL12/SDF-1 Antibody by Immunohistochemistry
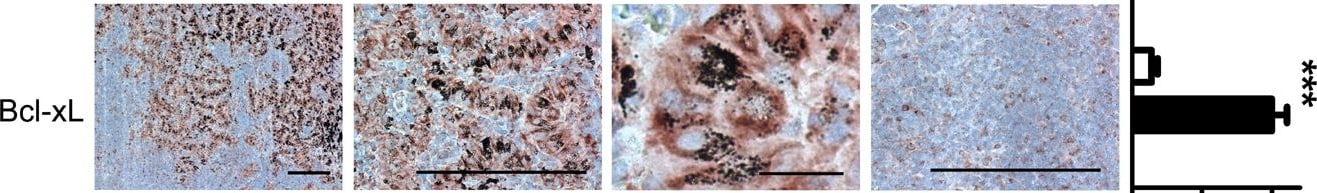
Tumor-promoting phenotype of the myeloid clusters.IHC staining demonstrating the expression of CD163, IL-6, IL-10, VEGF-A, MMP-9, SDF-1 and Bcl-xL by the myeloid cells associated with anthracosis. For each protein, representative images showing positive and negative staining areas were selected from the same slide. The quantification was performed by analyzing random images of myeloid cluster areas associated with anthracosis and other areas (10 images for each group) from 10 patients. Two-tailed Student's t-test was used for statistical analysis. Shown are means ± SEM, *** P<0.001. (Scale bar, 200 µm for 100× and 400×; 25 µm for 1600×) Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/23717691), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse CXCL12/SDF-1 by Immunohistochemistry
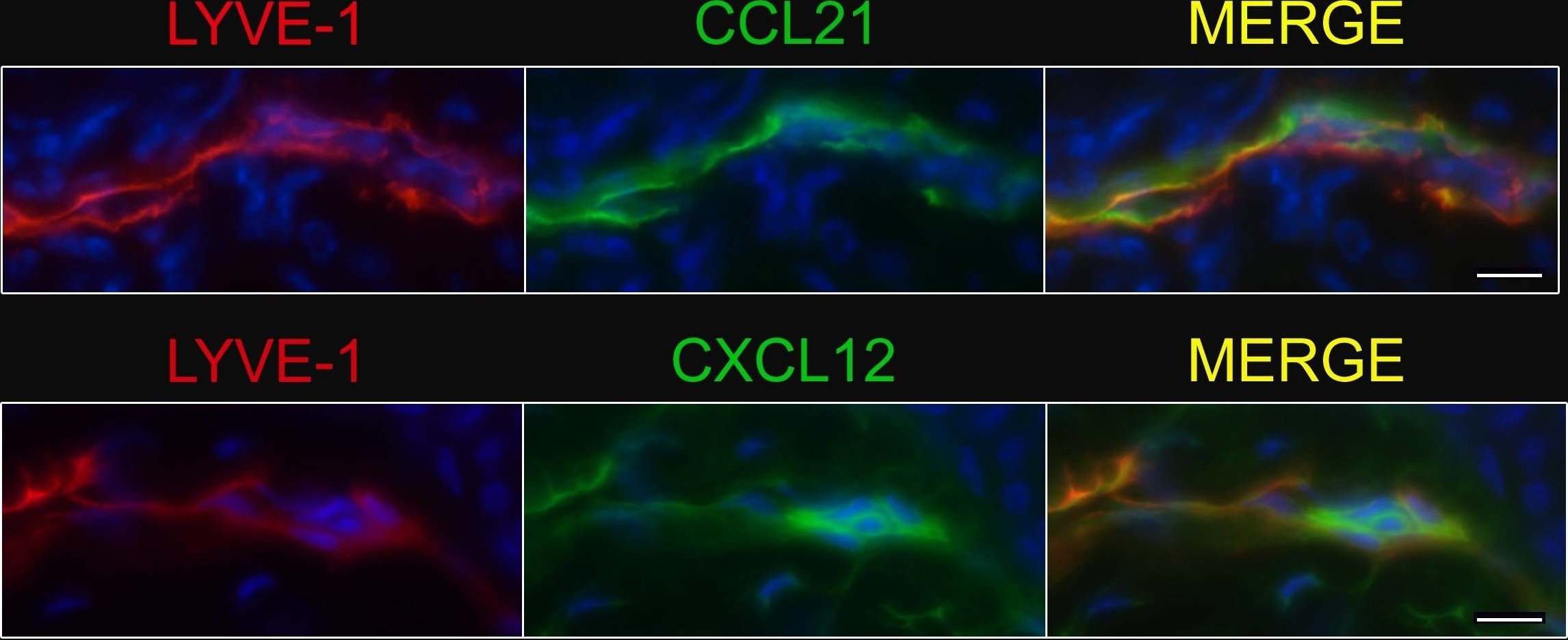
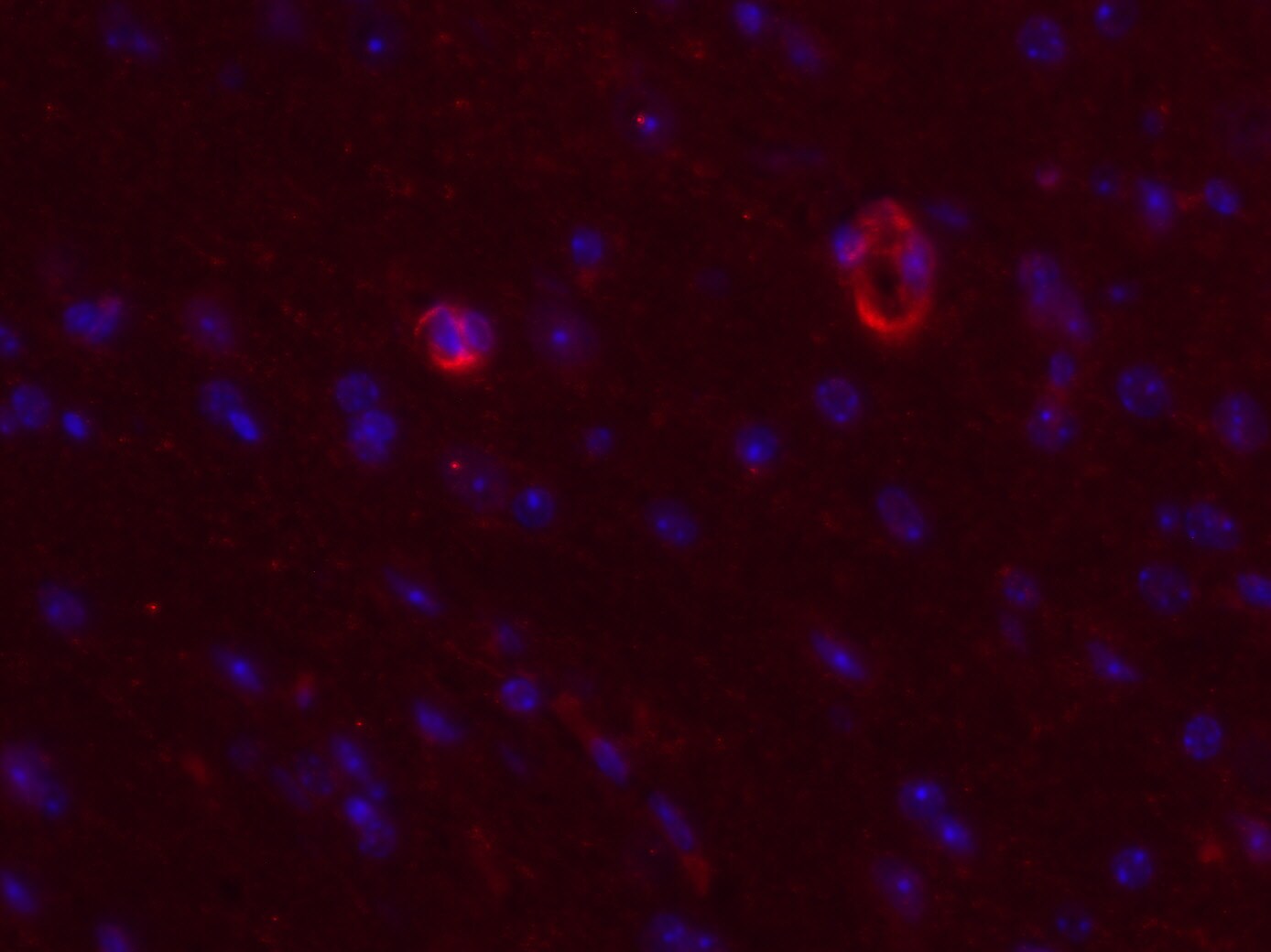
Afferent lymph vessels in chronically inflamed skin express CXCL12.Immunofluorescence staining of frozen sections of chronically inflamed skin (21 days after inflammation elicited by subcutaneous injection of CFA) for CXC12 and CCL21 by LYVE-1+ lymph vessels using DAPI counterstaining. One representative image of lymph vessels analyzed in 5 mice is shown. Scale bars, 10 µm. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/24752354), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of CXCL12/SDF-1 by Western Blot
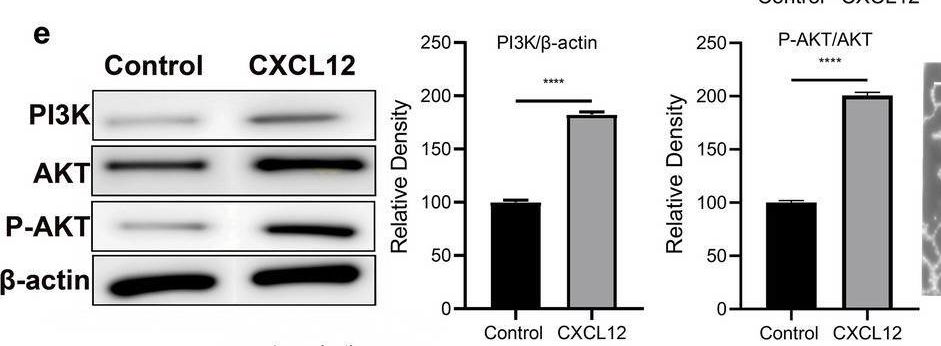
CXCR4/CXCL12 signaling promotes lymphangiogenesis in TSCC. a Cell proliferation of human lymphatic endothelial cells (HLECs) treated with CXCL12 (100 ng/ml) or vehicle control, assessed by CCK-8 assay. b Wound healing assay showing the effect of CXCL12 on HLEC migration. Representative images (left) and quantification (right). Scale bars, 100 μm. c Tube formation assay of HLECs treated with CXCL12 (100 ng/ml) or vehicle control. Representative images (left) and quantification (right). Scale bars, 100 μm. d Western blot analysis of lymphangiogenic factors (VEGF-C, VEGFR-3, and Prox1) in HLECs treated with CXCL12 (100 ng/ml) or vehicle control. Representative blots (left) and quantification (right). e Western blot analysis of PI3K, p-AKT, and AKT in HLECs treated with CXCL12 (100 ng/ml) or vehicle control. Representative blots (left) and quantification (right). f Tube formation assay of HLECs treated with CXCL12 (100 ng/ml), LY294002 (20 μM), or CXCL12 + LY294002. Representative images (left) and quantification (right). Scale bars, 100 μm. g Schematic representation of the co-culture system used to investigate the interaction between TSCC cells and HLECs. h Transwell migration assay of HLECs co-cultured with CAL-27 cells with different CXCR4 expression levels. Representative images (left) and quantification (right). Scale bars, 100 μm. Data are presented as mean ± SD from three independent experiments. ***p < 0.001, ****p < 0.0001(compared to Control); ####p < 0.0001(compared to CXCL12) Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/40629333), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of CXCL12/SDF-1 by Immunohistochemistry
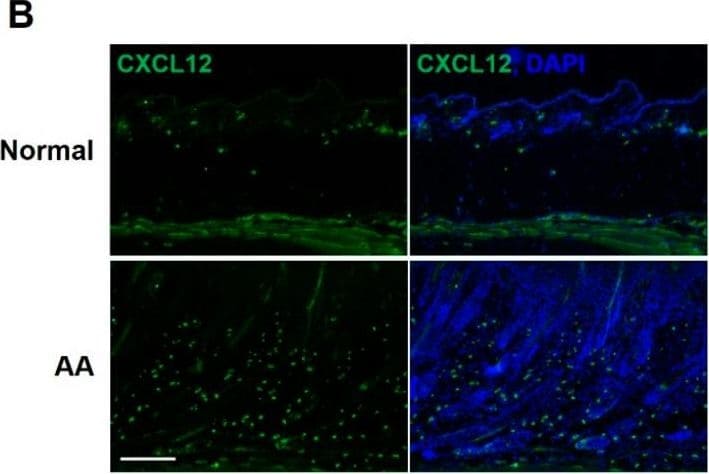
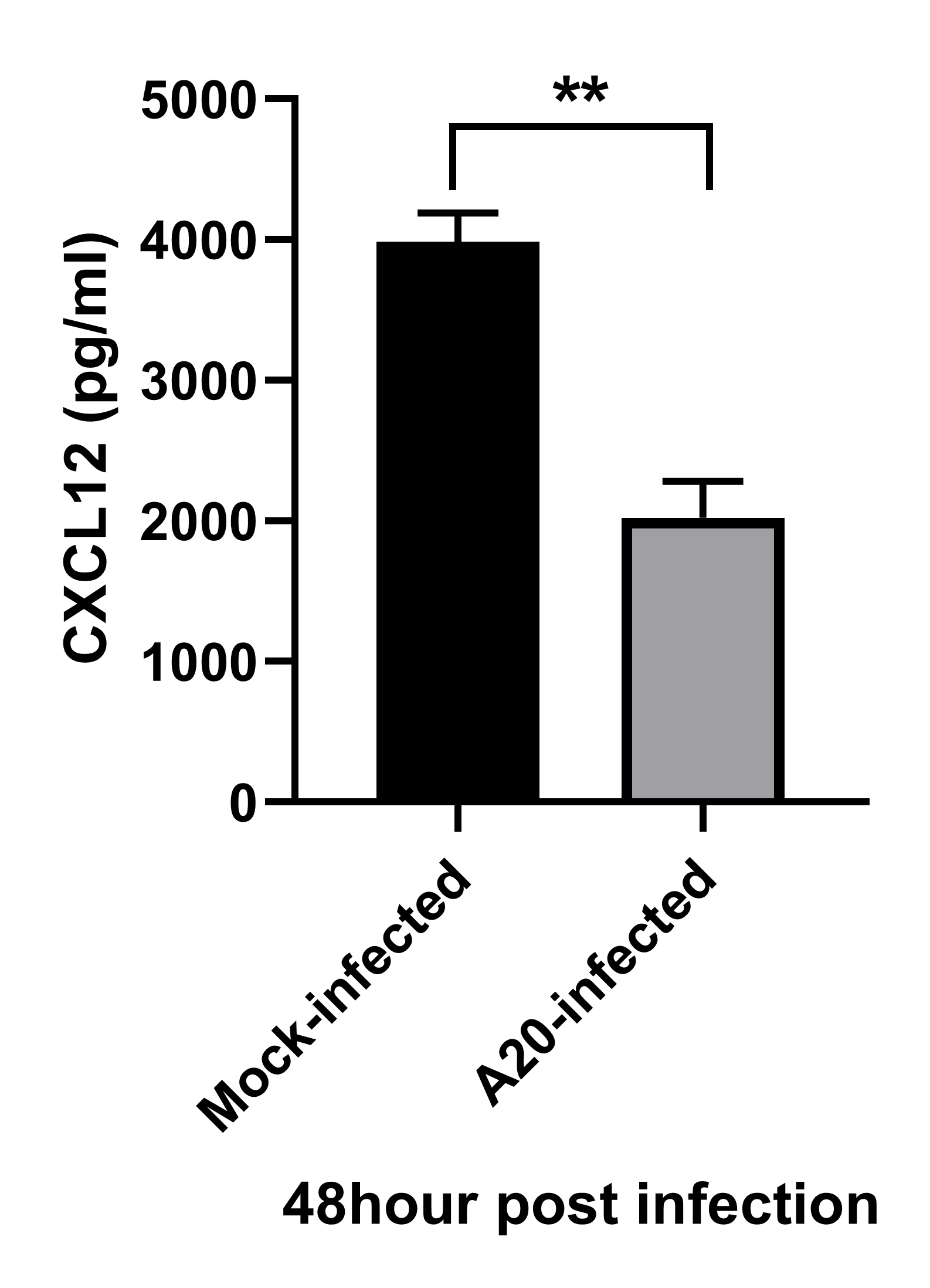
CXCL12 neutralization improves alopecia areata. (A) In this experimental design, skin-draining lymph node (SDLN) cells were isolated from AA-affected C3H/HeN female mice and intradermally injected into the dorsal skin of naïve mice to induce AA. Severe hair loss was observed after 12 weeks, and alpha CXCL12 (20 μg) was subcutaneously injected twice in a 2-week interval. (B) Skin sections from normal and AA mice were stained with an anti-CXCL12 antibody (green). The expression of CXCL12 increased in the AA model. The scale bar is set at 500 μm. (C) The administration of alpha CXCL12 in AA mice significantly reduced the areas of hair loss, which are marked in the images with dotted lines. The extent of hair loss in both the control and alpha CXCL12-treated mice was quantified using Image J (v1.53t). *** p < 0.001 vs. control, n = 3. (D) Skin sections of AA and alpha CXCL12-treated mice were stained with anti-CD8, anti-MHC-I, or anti-MHC-II antibodies. The alpha CXCL12-treated group had reduced expression of these immune reaction markers (green; white arrows). DAPI staining (blue) indicates cell nuclei. The scale bar is set at 100 μm. An asterisk indicates a statistical difference using Student’s t-test. Image collected and cropped by CiteAb from the following open publication (https://www.mdpi.com/1422-0067/25/3/1705), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of CXCL12/SDF-1 by Western Blot
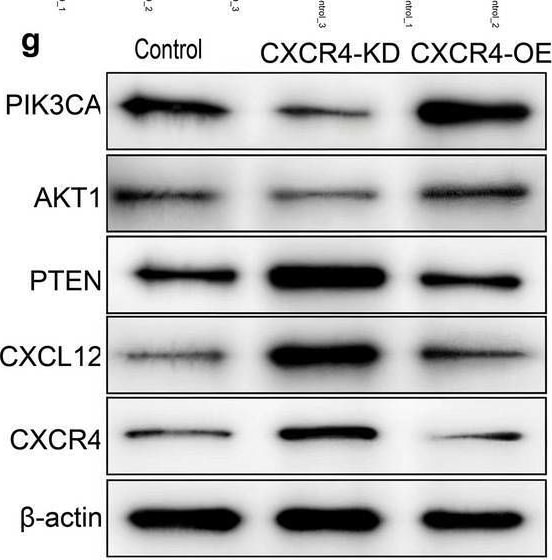
Transcriptomic analysis of CXCR4/CXCL12-regulated gene expression in TSCC. a Principal component analysis (PCA) of RNA-seq data from CAL-27 cells with different CXCR4 expression levels (Control, CXCR4-KD, and CXCR4-OE). b Volcano plot showing differentially expressed genes (DEGs) between Control and CXCR4-KD CAL-27 cells. c Volcano plot showing DEGs between Control and CXCR4-OE CAL-27 cells. d Heatmap of DEGs between Control and CXCR4-KD CAL-27 cells. e Heatmap of DEGs between Control and CXCR4-OE CAL-27 cells. f qRT-PCR validation of selected DEGs (PI3K3CA, AKT1, PTEN, CXCL12, and CXCR4) in CAL-27 cells with different CXCR4 expression levels. g Western blot validation of selected DEGs in CAL-27 cells with different CXCR4 expression levels. Representative blots (left) and quantification (right). h Proposed mechanistic model illustrating how the CXCR4/CXCL12 axis promotes TSCC lymphatic metastasis through the PI3K/AKT pathway. Data are presented as mean ± SD from three independent experiments. *p < 0.05, **p < 0.01, ***p < 0.001, ****p < 0.0001 (compared to Control); ##p < 0.01, ###p < 0.001, ####p < 0.0001(compared to CXCR4-OE) Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/40629333), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of CXCL12/SDF-1 by Immunohistochemistry
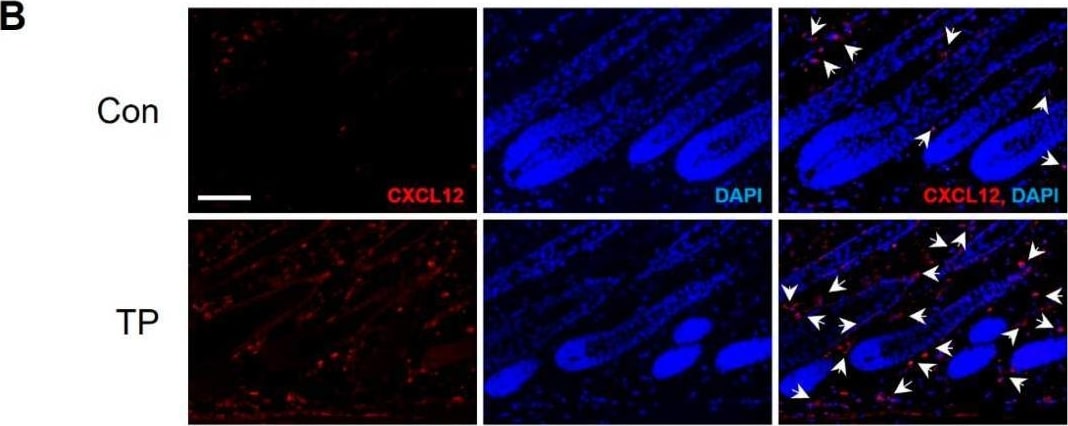
Neutralization of CXCL12 promotes hair growth in a testosterone-induced AGA model. (A) The back skin of 7-week-old C3H male mice was shaved and subcutaneous (s.c.) injections of testosterone propionate (TP) were used to induce the AGA. (B) The expression of CXCL12 in the dorsal skin of control and TP-treated mice was detected using immunofluorescence. CXCL12+ cells (red) are indicated by white arrows, and DAPI staining (blue) indicates cell nuclei. The scale bar is set at 100 μm. (C) Different doses of CXCL12 neutralizing antibody ( alpha CXCL12, 5, and 20 μg) were subcutaneously injected twice a week for three weeks. A daily topical treatment of 0.05% dutasteride served as a positive control. alpha CXCL12 administration accelerated hair growth in TP-treated C3H mice and the hair weight was measured. ### p < 0.001 vs. control, * p < 0.05, ** p < 0.01 vs. TP-treated, n = 7. (D) alpha CXCL12 (10, 100, or 1000 ng/mL) treatment increased the length of mouse vibrissa follicles in the AGA mimic ex vivo model. ## p < 0.01 vs. control, * p < 0.05 vs. TP-treated, n = 8. + or -: treated with or without TP, alpha CXCL12. The asterisk and sharp symbols indicate statistical differences using Student’s t-test. Image collected and cropped by CiteAb from the following open publication (https://www.mdpi.com/1422-0067/25/3/1705), licensed under a CC-BY license. Not internally tested by R&D Systems.Applications for Human/Mouse CXCL12/SDF‑1 Antibody
Application
Recommended Usage
COMET
Optimal dilutions of this antibody should be experimentally determined.
CyTOF-ready
Ready to be labeled using established conjugation methods. No BSA or other carrier proteins that could interfere with conjugation.
Dual RNAscope ISH-IHC Compatible
5-25 µg/mL
Sample: Immersion fixed paraffin-embedded sections of human tonsil
Sample: Immersion fixed paraffin-embedded sections of human tonsil
Immunocytochemistry
8-15 µg/mL
Sample: Immersion fixed human peripheral blood mononuclear cells (PBMCs) and K562 human chronic myelogenous leukemia cell line
Sample: Immersion fixed human peripheral blood mononuclear cells (PBMCs) and K562 human chronic myelogenous leukemia cell line
Immunohistochemistry
8-25 µg/mL
Sample: Immersion fixed paraffin-embedded sections of human tonsil
Sample: Immersion fixed paraffin-embedded sections of human tonsil
Intracellular Staining by Flow Cytometry
0.25 µg/106 cells
Sample: HT1080 human fibrosarcoma cell line fixed and permeabilized with FlowX FoxP3 Fixation & Permeabilization Buffer Kit (Catalog # FC012)
Sample: HT1080 human fibrosarcoma cell line fixed and permeabilized with FlowX FoxP3 Fixation & Permeabilization Buffer Kit (Catalog # FC012)
Multiplex Immunofluorescence
15 µg/mL
Sample: Immersion fixed paraffin-embedded sections of Human Breast Tumor
Sample: Immersion fixed paraffin-embedded sections of Human Breast Tumor
Human CXCL12/SDF-1 beta Sandwich Immunoassay
ELISA Capture (Matched Antibody Pair)
Human/Mouse CXCL12/SDF-1 Sandwich Immunoassay
ELISA Capture (Matched Antibody Pair)
Recommended Concentration: 2-8 µg/mL
Use in combination with these reagents:
Use in combination with these reagents:
Please Note: Optimal dilutions of this antibody should be experimentally determined.
Reviewed Applications
Read 12 reviews rated 4.8 using MAB350 in the following applications:
Flow Cytometry Panel Builder
Bio-Techne Knows Flow Cytometry
Save time and reduce costly mistakes by quickly finding compatible reagents using the Panel Builder Tool.
Advanced Features
- Spectra Viewer - Custom analysis of spectra from multiple fluorochromes
- Spillover Popups - Visualize the spectra of individual fluorochromes
- Antigen Density Selector - Match fluorochrome brightness with antigen density
Formulation, Preparation, and Storage
Purification
Protein A or G purified from hybridoma culture supernatant
Reconstitution
Reconstitute at 0.5 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. See Certificate of Analysis for details.
*Small pack size (-SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
*Small pack size (-SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: CXCL12/SDF-1
Alternate Names
PBSF, SDF-1, SDF1
Gene Symbol
CXCL12
UniProt
Additional CXCL12/SDF-1 Products
Product Documents for Human/Mouse CXCL12/SDF‑1 Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human/Mouse CXCL12/SDF‑1 Antibody
For research use only
Citations for Human/Mouse CXCL12/SDF‑1 Antibody
Customer Reviews for Human/Mouse CXCL12/SDF‑1 Antibody (12)
4.8 out of 5
12 Customer Ratings
Have you used Human/Mouse CXCL12/SDF‑1 Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
Showing
1
-
5 of
12 reviews
Showing All
Filter By:
-
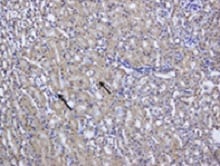
Application: ImmunohistochemistrySample Tested: Kidney tissueSpecies: MouseVerified Customer | Posted 08/02/2021
-
Application: Immunocytochemistry/ImmunofluorescenceSample Tested: Mast cellsSpecies: MouseVerified Customer | Posted 07/30/2021
-
Application: ImmunohistochemistrySample Tested: Tonsil tissueSpecies: HumanVerified Customer | Posted 08/25/2020Epitope retrieval: Leica BOND ER2 (high-pH) 100C 40min Primary Ab: 5ug/mL in BSA/TBS for 30min Detection system: Leica BOND-RX autostainer with Leica BOND refine detection kit
-
Application: MicroarraysSample Tested: EDTA PlasmaSpecies: HumanVerified Customer | Posted 06/10/2020
-
Application: ELISASample Tested: A549 human lung carcinoma cell lineSpecies: HumanVerified Customer | Posted 02/16/2020
-
Application: Western BlotSample Tested: Recombinant proteinSpecies: HumanVerified Customer | Posted 09/28/2019
-
Application: Immunocytochemistry/ImmunofluorescenceSample Tested: Spinal cordSpecies: MouseVerified Customer | Posted 05/11/2019
-
Application: Immunohistochemistry-FrozenSample Tested: Adult brainSpecies: MouseVerified Customer | Posted 04/18/2019Murine brain cryosection was stained with anti-CXCL12 and anti-mouse Alexa Fluor 555. Single positive cells were observed (also inner surface of vessels), looks quite specific although signal could be more intense (exposure time 1.5 s).
-
Application: ImmunohistochemistrySample Tested: 4T1 mouse breast cancer cell lineSpecies: MouseVerified Customer | Posted 03/05/20191:100 Citrate buffer antigen retrieval FFPE tissues
-
Application: MicroarraySample Tested: EDTA PlasmaSpecies: HumanVerified Customer | Posted 11/09/2018
-
Application: MicroarraysSample Tested: EDTA PlasmaSpecies: HumanVerified Customer | Posted 11/07/2018
-
Application: ImmunohistochemistrySample Tested: E6.5 mouse embryo fixed in 4% PFASpecies: MouseVerified Customer | Posted 04/13/2018
There are no reviews that match your criteria.
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- 7-Amino Actinomycin D (7-AAD) Cell Viability Flow Cytometry Protocol
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Extracellular Membrane Flow Cytometry Protocol
- Flow Cytometry Protocol for Cell Surface Markers
- Flow Cytometry Protocol for Staining Membrane Associated Proteins
- Flow Cytometry Staining Protocols
- Flow Cytometry Troubleshooting Guide
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- ICC Cell Smear Protocol for Suspension Cells
- ICC Immunocytochemistry Protocol Videos
- ICC for Adherent Cells
- IHC Sample Preparation (Frozen sections vs Paraffin)
- ISH-IHC Protocol for Chromogenic Detection on Formalin Fixed Paraffin Embedded (FFPE) Tissue
- Immunocytochemistry (ICC) Protocol
- Immunocytochemistry Troubleshooting
- Immunofluorescence of Organoids Embedded in Cultrex Basement Membrane Extract
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Intracellular Flow Cytometry Protocol Using Alcohol (Methanol)
- Intracellular Flow Cytometry Protocol Using Detergents
- Intracellular Nuclear Staining Flow Cytometry Protocol Using Detergents
- Intracellular Staining Flow Cytometry Protocol Using Alcohol Permeabilization
- Intracellular Staining Flow Cytometry Protocol Using Detergents to Permeabilize Cells
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Propidium Iodide Cell Viability Flow Cytometry Protocol
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Liperfluo
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Characterization of Human Th22 Cells
- Protocol for the Characterization of Human Th9 Cells
- Protocol for the Fluorescent ICC Staining of Cell Smears - Graphic
- Protocol for the Fluorescent ICC Staining of Cultured Cells on Coverslips - Graphic
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent ICC Staining of Cells on Coverslips
- Protocol for the Preparation and Fluorescent ICC Staining of Non-adherent Cells
- Protocol for the Preparation and Fluorescent ICC Staining of Stem Cells on Coverslips
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- Protocol for the Preparation of a Cell Smear for Non-adherent Cell ICC - Graphic
- Protocol: Annexin V and PI Staining by Flow Cytometry
- Protocol: Annexin V and PI Staining for Apoptosis by Flow Cytometry
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Fluorokine Flow Cytometry Kits
- Troubleshooting Guide: Immunohistochemistry
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...
Associated Pathways
 Jak/STAT Signaling Pathway
Jak/STAT Signaling Pathway