Osteopontin (OPN, previously also referred to as transformation-associated secreted phosphoprotein, bone sialoprotein I, 2ar, 2B7, early T lymphocyte activation 1 protein, minopotin, calcium oxalate crystal growth inhibitor protein), is a secreted, highly acidic, calcium-binding, RGD-containing, phosphorylated glycoprotein originally isolated from bone matrix (1). Subsequently, OPN has been found in kidney, placenta, blood vessels and various tumor tissues. Many cell types (including macrophages, osteoclasts, activated T cells, fibroblasts, epithelial cells, vascular smooth muscle cells, and natural killer cells) can express OPN in response to activation by cytokines, growth factors or inflammatory mediators. Elevated expression of OPN has also been associated with numerous pathobiological conditions such as atherosclerotic plaques, renal tubulointerstitial fibrosis, granuloma formations in tuberculosis and silicosis, neointimal formation associated with balloon catheterization, metastasizing tumors, and cerebral ischemia. Human OPN cDNA encodes a 314 amino acid (aa) residue precursor protein with a 16 aa residue predicted signal peptide that is cleaved to yield a 298 aa residue mature protein with an integrin binding sequence (RGD), and N- and O-glycosylation sites. By alternative splicing, at least three human OPN isoforms exist. OPN has been shown to bind to different cell types through RGD-mediated interaction with the integrins alpha v beta 1, alpha v beta 3, alpha v beta 5, and non-RGD-mediated interaction with CD44 and the integrins alpha 8 beta 1 or alpha 9 beta 1. OPN exists both as a component of extracellular matrix and as a soluble molecule. Functionally, OPN is chemotactic for macrophages, smooth muscle cells, endothelial cells and glial cells. OPN has also been shown to inhibit nitric oxide production and cytotoxicity by activated macrophages. Human, mouse, rat, pig, and bovine OPN share from approximately 40-80% amino acid sequence identity. Osteopontin is a substrate for proteolytic cleavage by thrombin, enterokinase, MMP-3, and MMP-7. The functions of OPN in a variety of cell types were shown to be modified as a result of proteolytic cleavage (2, 3).
Human Osteopontin/OPN Antibody
R&D Systems | Catalog # AF1433

Key Product Details
Species Reactivity
Validated:
Cited:
Applications
Validated:
Cited:
Label
Antibody Source
Product Specifications
Immunogen
Specificity
Clonality
Host
Isotype
Endotoxin Level
Scientific Data Images for Human Osteopontin/OPN Antibody
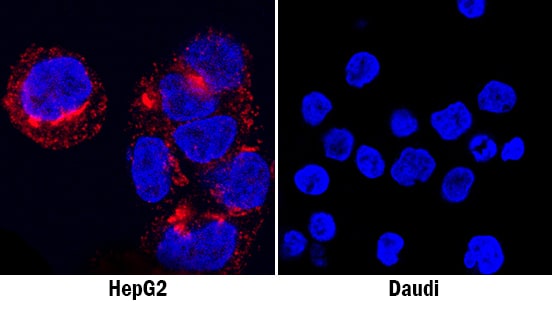
Osteopontin/OPN in HepG2 Human Cell Line.
Osteopontin/OPN was detected in immersion fixed HepG2 human hepatocellular carcinoma cell line (positive staining; left panel) and Daudi human Burkitt's lymphoma cell line (negative staining; right panel) using Goat Anti-Human Osteopontin/OPN Antigen Affinity-purified Polyclonal Antibody (Catalog # AF1433) at 1.7 µg/mL for 3 hours at room temperature. Cells were stained using the NorthernLights™ 557-conjugated Anti-Goat IgG Secondary Antibody (red; Catalog # NL001) and counterstained with DAPI (blue). Specific staining was localized to cytoplasm. View our protocol for Fluorescent ICC Staining of Cells on Coverslips.Osteopontin/OPN in Human Breast Cancer Tissue.
Osteopontin/OPN was detected in immersion fixed paraffin-embedded sections of human breast cancer tissue using 8 µg/mL Goat Anti-Human Osteopontin/OPN Antigen Affinity-purified Polyclonal Antibody (Catalog # AF1433) overnight at 4 °C. Tissue was stained with the Anti-Goat HRP-DAB Cell & Tissue Staining Kit (brown; Catalog # CTS008) and counterstained with hematoxylin (blue). Specific labeling was localized to the surface of epithelial cells in the intralobular duct. View our protocol for Chromogenic IHC Staining of Paraffin-embedded Tissue Sections.Osteopontin/OPN in Human Breast Cancer Tissue.
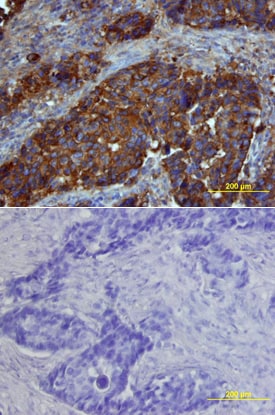
Osteopontin/OPN was detected in immersion fixed paraffin-embedded sections of human breast cancer tissue using Goat Anti-Human Osteopontin/OPN Antigen Affinity-purified Polyclonal Antibody (Catalog # AF1433) at 15 µg/mL overnight at 4 °C. Tissue was stained using the Anti-Goat HRP-DAB Cell & Tissue Staining Kit (brown; Catalog # CTS008) and counterstained with hematoxylin (blue). Lower panel shows a lack of labeling if primary antibodies are omitted and tissue is stained only with secondary antibody followed by incubation with detection reagents. View our protocol for Chromogenic IHC Staining of Paraffin-embedded Tissue Sections.Cell Adhesion Mediated by Osteopontin/OPN and Neutralization by Human Osteopontin/OPN Antibody.
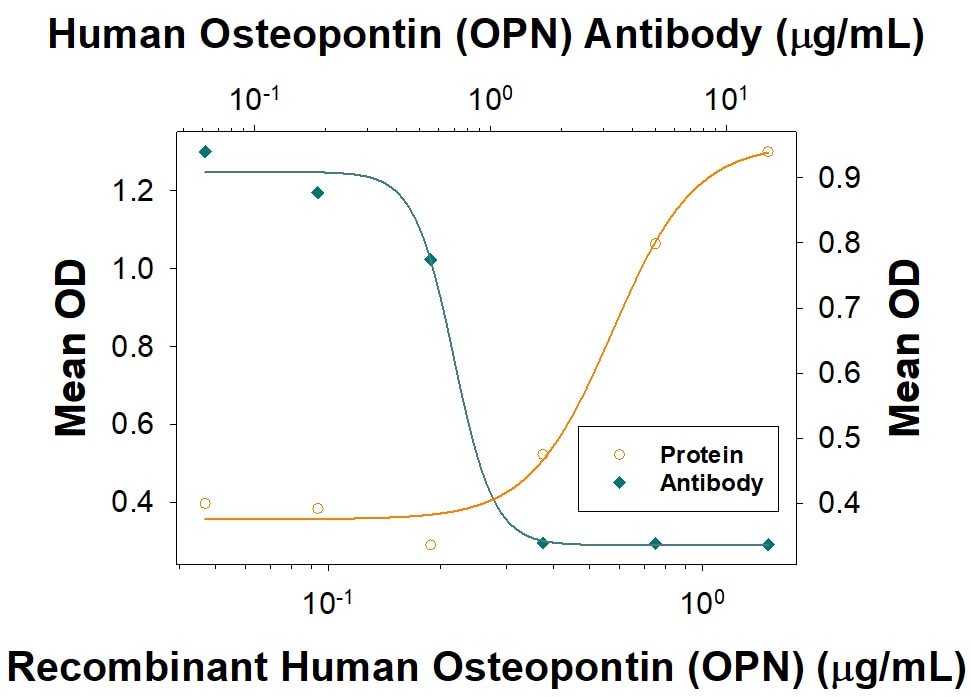
Recombinant Human Osteopontin/OPN (Catalog # 1433-OP), immobilized onto a microplate, supports the adhesion of the HEK293 human embryonic kidney cell line in a dose-dependent manner (orange line). Adhesion elicited by Recombinant Human Osteopontin/OPN (1 µg/mL) is neutralized (green line) by increasing concentrations of Goat Anti-Human Osteopontin/OPN Antigen Affinity-purified Polyclonal Antibody (Catalog # AF1433). The ND50 is typically 0.300 - 3.60 µg/mL.Human Osteopontin/OPN ELISA Standard Curve.
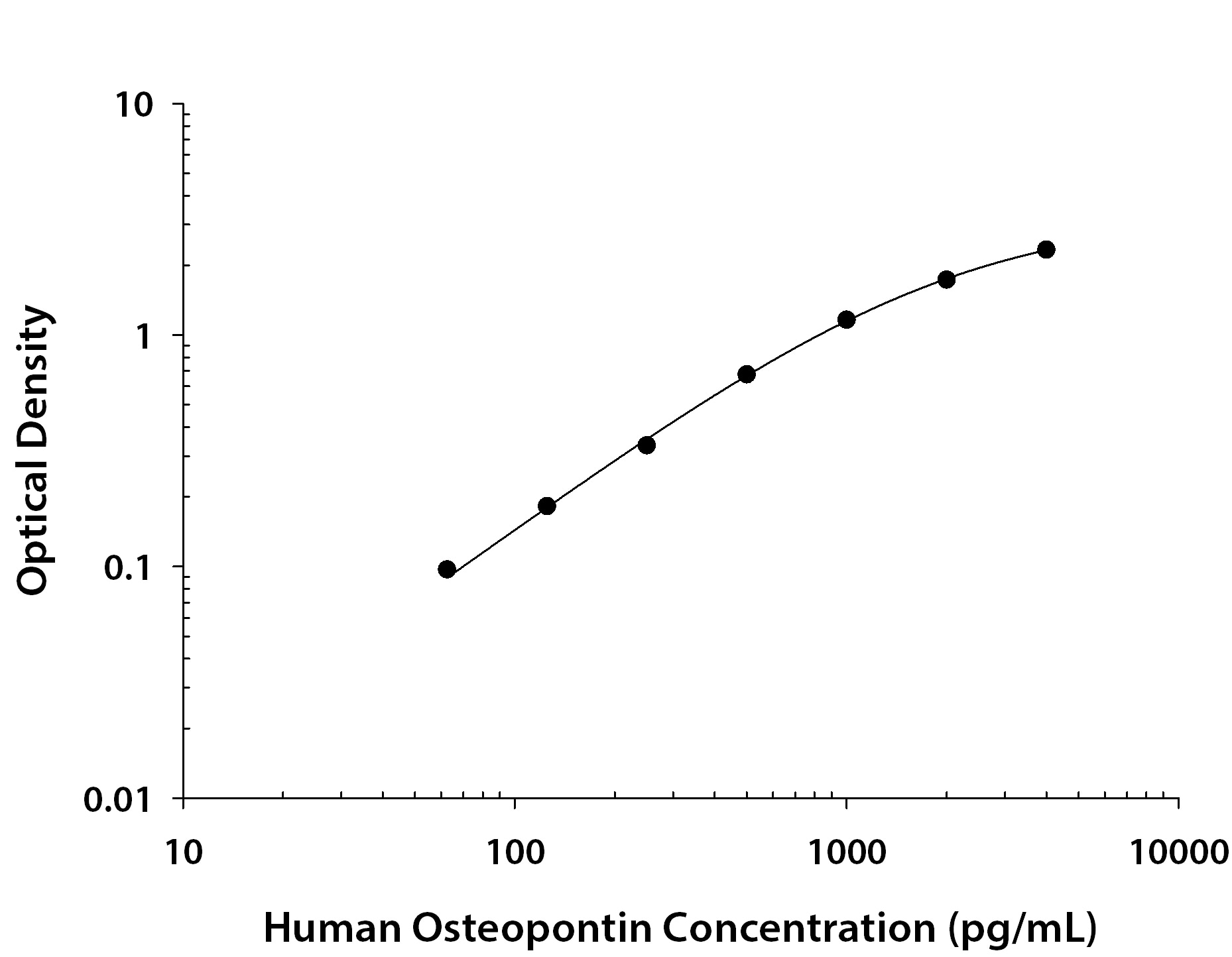
Recombinant Human Osteopontin/OPN protein was serially diluted 2-fold and captured by Mouse Anti-Human Osteopontin/OPN Monoclonal Antibody (Catalog # MAB14332R) coated on a Clear Polystyrene Microplate (Catalog # DY990). Goat Anti-Human Osteopontin/OPN Antigen Affinity-purified Polyclonal Antibody (Catalog # AF1433) was biotinylated and incubated with the protein captured on the plate. Detection of the standard curve was achieved by incubating Streptavidin-HRP (Catalog # DY998) followed by Substrate Solution (Catalog # DY999) and stopping the enzymatic reaction with Stop Solution (Catalog # DY994).Detection of Osteopontin/OPN in Human Kidney.
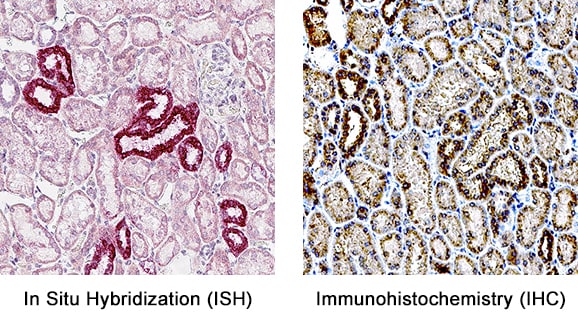
Formalin-fixed paraffin-embedded tissue sections of human kidney were probed for OPN mRNA (ACD RNAScope Probe, catalog #420101; Fast Red chromogen, ACD catalog # 322750). Adjacent tissue section was processed for immunohistochemistry using goat anti-human OPN polyclonal antibody (R&D Systems catalog # AF1433) at 1ug/mL with overnight incubation at 4 degrees Celsius followed by incubation with anti-goat IgG VisUCyte HRP Polymer Antibody (Catalog # VC004) and DAB chromogen (yellow-brown). Tissue was counterstained with hematoxylin (blue). Specific staining was localized to cytoplasm in tubules.Detection of Human Osteopontin/OPN by Western Blot
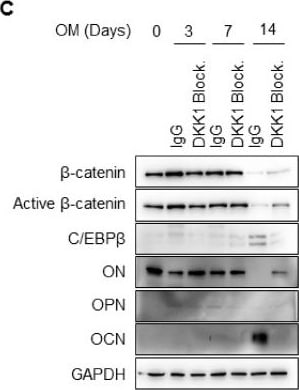
DKK1 blockade inhibits mineralization of osteoblast differentiation. Osteoblasts were treated with 1 μg anti-DKK1 or IgG as controls during osteoblast differentiation (n = 3). At indicated days, analysis of (A) results of ALP and ARS staining; scale bar is 200 μm, (B) hydroxyapatite staining; BF, Bright Field; HA, Hydroxyapatite; Scale bar is 500 μm, (C) immunoblotting for proteins, and (D) qPCR for mRNA. Representative data are shown (n = 3). * p < 0.05 (mean ± SD; n = 3). Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/31963554), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Osteopontin/OPN by Western Blot
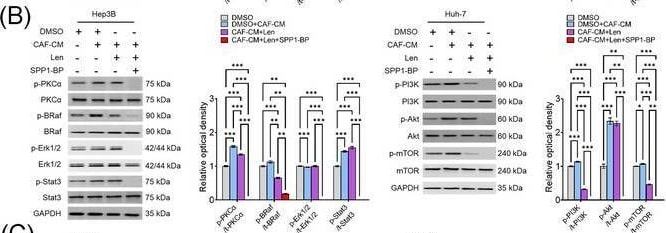
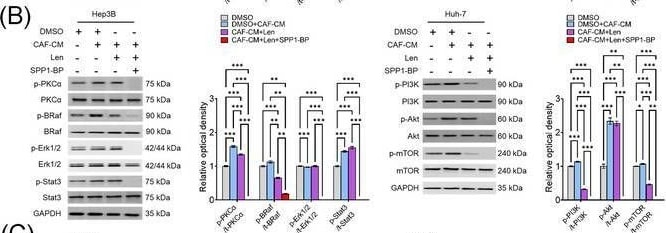
Molecular mechanism underlying CAF‐derived SPP1‐induced resistance to sorafenib or lenvatinib. (A‐B) Changes in the expression levels of PKC alpha, BRAF/ERK/STAT3, and PI3K/AKT/mTOR pathway proteins in CAF‐CM‐incubated Huh‐7 cells treated with sorafenib (15 μmol/L; A) or lenvatinib alone (5 μmol/L; B) and in combination with SPP1‐BP. (C‐D) Changes in the phosphorylation of AKT, mTOR, BRAF, and ERK1/2 under ITGB1 and/or ITGB5 silencing in Hep3B and Huh‐7 cells treated with sorafenib (C) or lenvatinib (D) in combination with CAF‐CM. (Mean ± SEM; Two‐way ANOVA test; *P < 0.05; **P < 0.01; ***P < 0.001).Abbreviations: DMSO, dimethyl sulfoxide; CAF, cancer‐associated fibroblast; CM, culture medium; Sor, sorafenib; SPP1‐BP, SPP1‐blocking peptide, PKC alpha, protein kinase C alpha ; BRAF,v‐Raf murine sarcoma viral oncogene homolog B; ERK, extracellular signal‐related kinase; STAT3, signal transducer and activator of transcription 3; GAPDH, glyceraldehyde 3‐phosphate dehydrogenase; PI3K, phosphatidylinositol‐3‐kinase; AKT, protein kinase B; mTOR, mammalian target of rapamycin; Len, lenvatinib; siCtrl, negative control; ITGB1, integrin subunit beta 1; ITGB5, integrin subunit beta 5. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/36919193), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Osteopontin/OPN by Immunohistochemistry
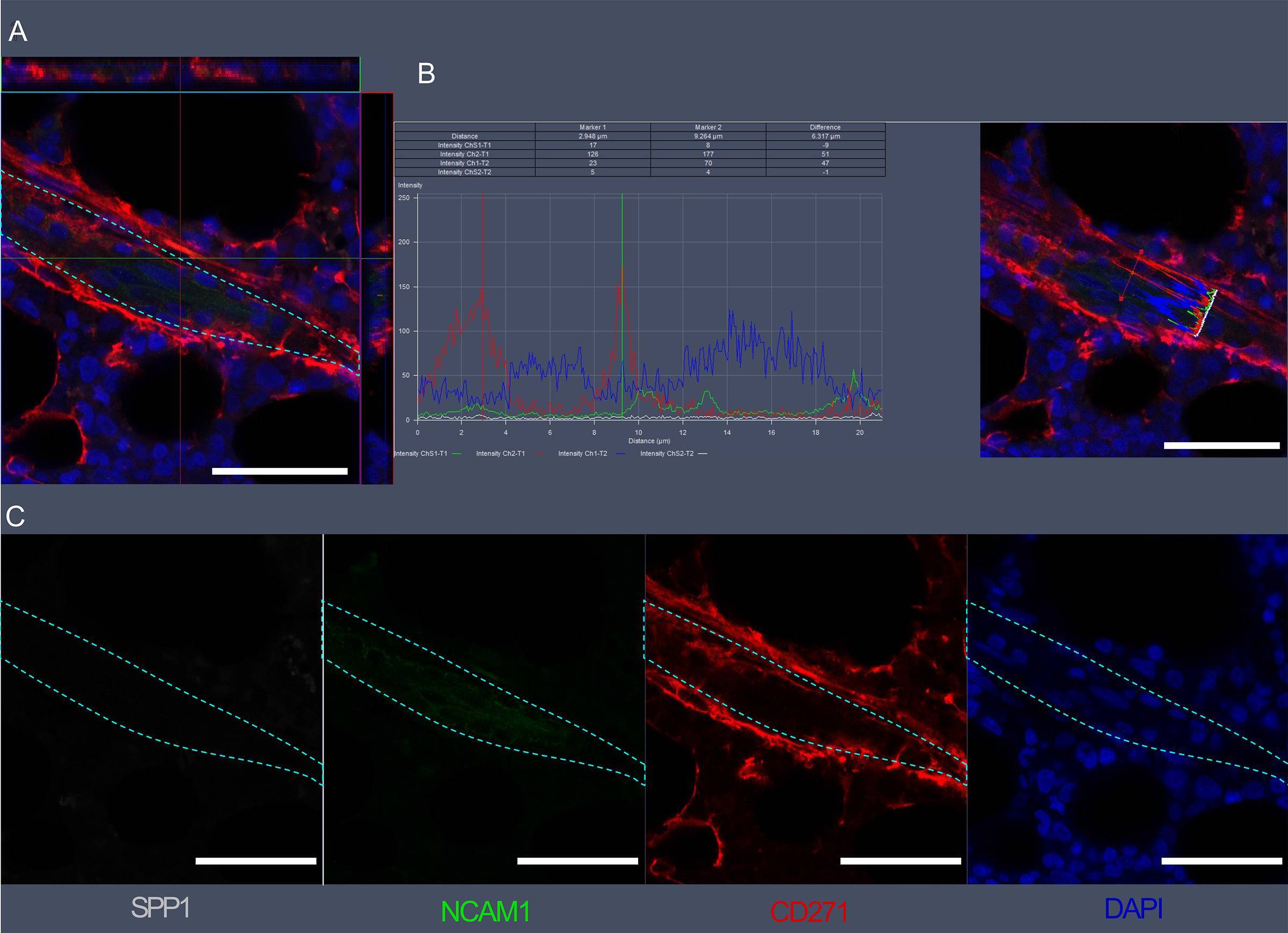
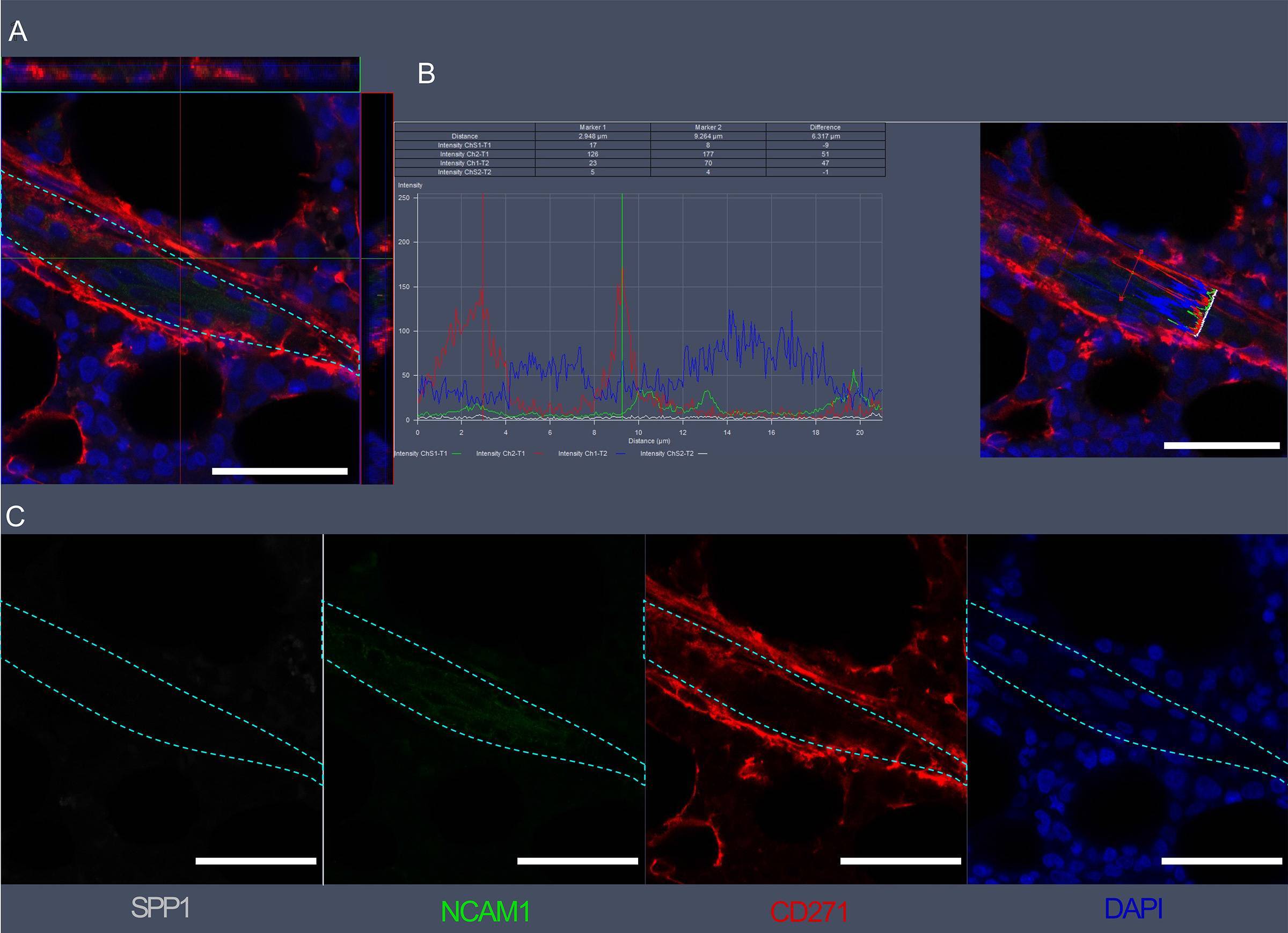
Visualization of SPP1, NCAM1, and CD271 expression in bone marrow vascular regions by confocal microscopy.(A) Confocal scan of vascular region in BM biopsies with 3D orthographic cross-section view, co-stained with rabbit anti-NCAM1, mouse anti-CD271, goat anti-SPP1, and DAPI. (B) Intensity profile for all channels in A across a cell of interest in a representative z plane. (C) Single channel data for the florescent markers in A. Scale bars represent 50 μm. Cyan dashed lines indicate vessel surface. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/36876630), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Osteopontin/OPN by Immunohistochemistry
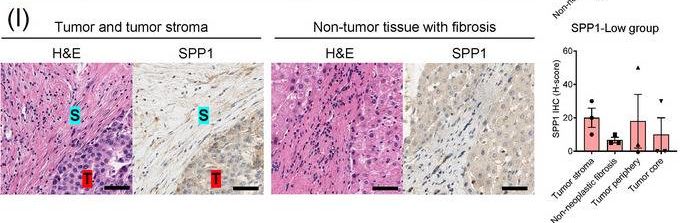
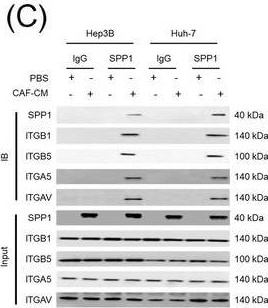
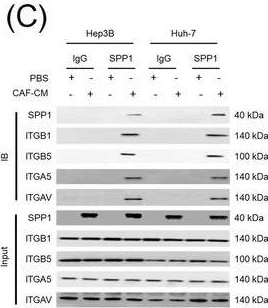
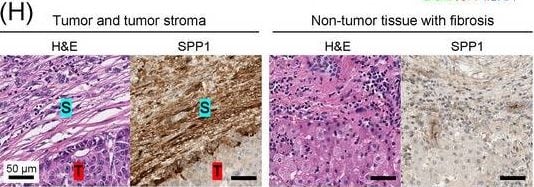
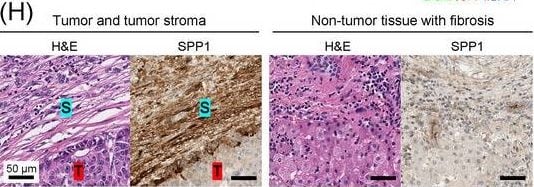
Identification of the CAF‐derived molecules that induce resistance to sorafenib or lenvatinib in patients with HCC. (A) Heat map of 790 genes overexpressed in CAFs. (B) GSEA of the genes overexpressed in CAFs. (C) Process of candidate gene selection. (D) Expression levels of the 9 candidate genes according to sorafenib response in the GSE109211 dataset. (E) Comparison of SPP1 expression between sorafenib responders and non‐responders in the GSE109211 and GSE143233 datasets. (F) Immunofluorescence staining of alpha ‐SMA and SPP1 in tumor tissues from patients with HCC. (G) Comparison of SPP1 expression between CAFs and their paired para‐cancer fibroblasts in the WTS data from the 9 pairs of CAF and para‐cancer fibroblasts. (H) Representative H&E and SPP1 IHC images of the tumor (T), tumor stroma (S) (left), and non‐tumor fibrous tissue (middle) in an SPP1‐high patient. Scale bar, 50 μm. Comparison of SPP1 IHC staining intensity according to tumor location in the SPP1‐high group (right). (I) Representative H&E and SPP1 IHC images of tumor (T), tumor stroma (S) (left), and non‐tumor fibrous tissue (middle) in an SPP1‐low patient. Comparison of SPP1 IHC staining intensity according to tumor location in the SPP1‐low group (right). (Mean ± SEM; unpaired Welch's t‐test; *P < 0.05; **P < 0.01; ***P < 0.001).Abbreviations: HCC, hepatocellular carcinoma; NF, normal fibroblast; CAFs, cancer‐associated fibroblasts; ELISA, enzyme linked immunosorbent assay; H&E, hematoxylin and eosin; alpha ‐SMA, alpha smooth muscle actin; SPP1, secreted phosphoprotein 1; DAPI, 4′, 6‐diamidino‐2‐phenylindole; PAF, para‐cancer fibroblast; S, tumor stroma; T, tumor; IHC, immunohistochemistry. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/36919193), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Osteopontin/OPN by Western Blot
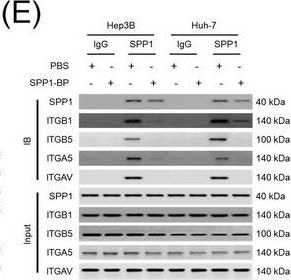
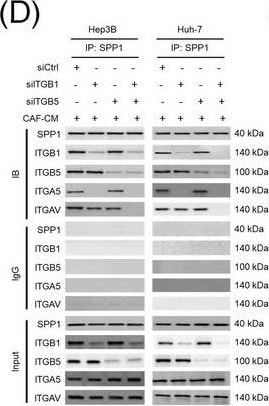
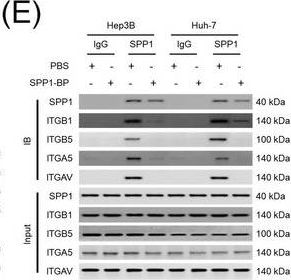
Integrin complexes (integrin alpha V beta 5, alpha 5 beta 1, and alpha V beta 1) on HCC cells were identified as binding proteins of SPP1. (A) Western blotting of SPP1 receptor‐binding protein expression in HCC cells. (B) Measurement of CD44 and integrin expression in Hep3B and Huh‐7 cells by fluorescent intensity. (C‐E) Co‐IP of SPP1 after CAF‐CM treatment, followed by Western blotting for SPP1, ITGA5, ITGAV, ITGB1, and ITGB5 in HCC cells (C), HCC cells silenced for ITGB1 and/or ITGB5 (D), and HCC cells incubated with/without SPP1‐BP (E).Abbreviations: ITGA4, integrin subunit alpha 4; ITGA5, integrin subunit alpha 5; ITGA8, integrin subunit alpha 8; ITGA9, integrin subunit alpha 9; ITGAV, integrin subunit alpha V; ITGB1, integrin subunit beta 1; ITGB3, integrin subunit beta 3; ITGB5, integrin subunit beta ; GAPDH, glyceraldehyde 3‐phosphate dehydrogenase; RFU, relative fluorescence units; IgG, immunoglobulin G; SPP1, secreted phosphoprotein 1; CAF, cancer‐associated fibroblast; CM, culture medium; IB, immunoblotting; siCtrl, negative control; SPP1‐BP, SPP1‐blocking peptide. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/36919193), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Osteopontin/OPN by Western Blot
Integrin complexes (integrin alpha V beta 5, alpha 5 beta 1, and alpha V beta 1) on HCC cells were identified as binding proteins of SPP1. (A) Western blotting of SPP1 receptor‐binding protein expression in HCC cells. (B) Measurement of CD44 and integrin expression in Hep3B and Huh‐7 cells by fluorescent intensity. (C‐E) Co‐IP of SPP1 after CAF‐CM treatment, followed by Western blotting for SPP1, ITGA5, ITGAV, ITGB1, and ITGB5 in HCC cells (C), HCC cells silenced for ITGB1 and/or ITGB5 (D), and HCC cells incubated with/without SPP1‐BP (E).Abbreviations: ITGA4, integrin subunit alpha 4; ITGA5, integrin subunit alpha 5; ITGA8, integrin subunit alpha 8; ITGA9, integrin subunit alpha 9; ITGAV, integrin subunit alpha V; ITGB1, integrin subunit beta 1; ITGB3, integrin subunit beta 3; ITGB5, integrin subunit beta ; GAPDH, glyceraldehyde 3‐phosphate dehydrogenase; RFU, relative fluorescence units; IgG, immunoglobulin G; SPP1, secreted phosphoprotein 1; CAF, cancer‐associated fibroblast; CM, culture medium; IB, immunoblotting; siCtrl, negative control; SPP1‐BP, SPP1‐blocking peptide. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/36919193), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Osteopontin/OPN by Western Blot
Integrin complexes (integrin alpha V beta 5, alpha 5 beta 1, and alpha V beta 1) on HCC cells were identified as binding proteins of SPP1. (A) Western blotting of SPP1 receptor‐binding protein expression in HCC cells. (B) Measurement of CD44 and integrin expression in Hep3B and Huh‐7 cells by fluorescent intensity. (C‐E) Co‐IP of SPP1 after CAF‐CM treatment, followed by Western blotting for SPP1, ITGA5, ITGAV, ITGB1, and ITGB5 in HCC cells (C), HCC cells silenced for ITGB1 and/or ITGB5 (D), and HCC cells incubated with/without SPP1‐BP (E).Abbreviations: ITGA4, integrin subunit alpha 4; ITGA5, integrin subunit alpha 5; ITGA8, integrin subunit alpha 8; ITGA9, integrin subunit alpha 9; ITGAV, integrin subunit alpha V; ITGB1, integrin subunit beta 1; ITGB3, integrin subunit beta 3; ITGB5, integrin subunit beta ; GAPDH, glyceraldehyde 3‐phosphate dehydrogenase; RFU, relative fluorescence units; IgG, immunoglobulin G; SPP1, secreted phosphoprotein 1; CAF, cancer‐associated fibroblast; CM, culture medium; IB, immunoblotting; siCtrl, negative control; SPP1‐BP, SPP1‐blocking peptide. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/36919193), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Osteopontin/OPN by Western Blot
Molecular mechanism underlying CAF‐derived SPP1‐induced resistance to sorafenib or lenvatinib. (A‐B) Changes in the expression levels of PKC alpha, BRAF/ERK/STAT3, and PI3K/AKT/mTOR pathway proteins in CAF‐CM‐incubated Huh‐7 cells treated with sorafenib (15 μmol/L; A) or lenvatinib alone (5 μmol/L; B) and in combination with SPP1‐BP. (C‐D) Changes in the phosphorylation of AKT, mTOR, BRAF, and ERK1/2 under ITGB1 and/or ITGB5 silencing in Hep3B and Huh‐7 cells treated with sorafenib (C) or lenvatinib (D) in combination with CAF‐CM. (Mean ± SEM; Two‐way ANOVA test; *P < 0.05; **P < 0.01; ***P < 0.001).Abbreviations: DMSO, dimethyl sulfoxide; CAF, cancer‐associated fibroblast; CM, culture medium; Sor, sorafenib; SPP1‐BP, SPP1‐blocking peptide, PKC alpha, protein kinase C alpha ; BRAF,v‐Raf murine sarcoma viral oncogene homolog B; ERK, extracellular signal‐related kinase; STAT3, signal transducer and activator of transcription 3; GAPDH, glyceraldehyde 3‐phosphate dehydrogenase; PI3K, phosphatidylinositol‐3‐kinase; AKT, protein kinase B; mTOR, mammalian target of rapamycin; Len, lenvatinib; siCtrl, negative control; ITGB1, integrin subunit beta 1; ITGB5, integrin subunit beta 5. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/36919193), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Osteopontin/OPN by Western Blot
Integrin complexes (integrin alpha V beta 5, alpha 5 beta 1, and alpha V beta 1) on HCC cells were identified as binding proteins of SPP1. (A) Western blotting of SPP1 receptor‐binding protein expression in HCC cells. (B) Measurement of CD44 and integrin expression in Hep3B and Huh‐7 cells by fluorescent intensity. (C‐E) Co‐IP of SPP1 after CAF‐CM treatment, followed by Western blotting for SPP1, ITGA5, ITGAV, ITGB1, and ITGB5 in HCC cells (C), HCC cells silenced for ITGB1 and/or ITGB5 (D), and HCC cells incubated with/without SPP1‐BP (E).Abbreviations: ITGA4, integrin subunit alpha 4; ITGA5, integrin subunit alpha 5; ITGA8, integrin subunit alpha 8; ITGA9, integrin subunit alpha 9; ITGAV, integrin subunit alpha V; ITGB1, integrin subunit beta 1; ITGB3, integrin subunit beta 3; ITGB5, integrin subunit beta ; GAPDH, glyceraldehyde 3‐phosphate dehydrogenase; RFU, relative fluorescence units; IgG, immunoglobulin G; SPP1, secreted phosphoprotein 1; CAF, cancer‐associated fibroblast; CM, culture medium; IB, immunoblotting; siCtrl, negative control; SPP1‐BP, SPP1‐blocking peptide. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/36919193), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Osteopontin/OPN by Immunohistochemistry
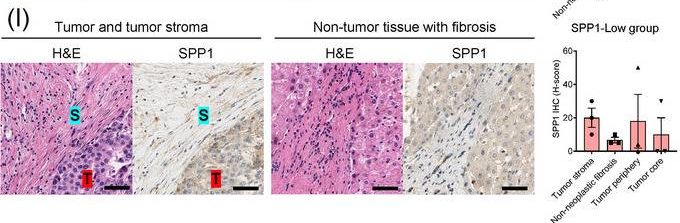
Identification of the CAF‐derived molecules that induce resistance to sorafenib or lenvatinib in patients with HCC. (A) Heat map of 790 genes overexpressed in CAFs. (B) GSEA of the genes overexpressed in CAFs. (C) Process of candidate gene selection. (D) Expression levels of the 9 candidate genes according to sorafenib response in the GSE109211 dataset. (E) Comparison of SPP1 expression between sorafenib responders and non‐responders in the GSE109211 and GSE143233 datasets. (F) Immunofluorescence staining of alpha ‐SMA and SPP1 in tumor tissues from patients with HCC. (G) Comparison of SPP1 expression between CAFs and their paired para‐cancer fibroblasts in the WTS data from the 9 pairs of CAF and para‐cancer fibroblasts. (H) Representative H&E and SPP1 IHC images of the tumor (T), tumor stroma (S) (left), and non‐tumor fibrous tissue (middle) in an SPP1‐high patient. Scale bar, 50 μm. Comparison of SPP1 IHC staining intensity according to tumor location in the SPP1‐high group (right). (I) Representative H&E and SPP1 IHC images of tumor (T), tumor stroma (S) (left), and non‐tumor fibrous tissue (middle) in an SPP1‐low patient. Comparison of SPP1 IHC staining intensity according to tumor location in the SPP1‐low group (right). (Mean ± SEM; unpaired Welch's t‐test; *P < 0.05; **P < 0.01; ***P < 0.001).Abbreviations: HCC, hepatocellular carcinoma; NF, normal fibroblast; CAFs, cancer‐associated fibroblasts; ELISA, enzyme linked immunosorbent assay; H&E, hematoxylin and eosin; alpha ‐SMA, alpha smooth muscle actin; SPP1, secreted phosphoprotein 1; DAPI, 4′, 6‐diamidino‐2‐phenylindole; PAF, para‐cancer fibroblast; S, tumor stroma; T, tumor; IHC, immunohistochemistry. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/36919193), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Osteopontin/OPN by Western Blot
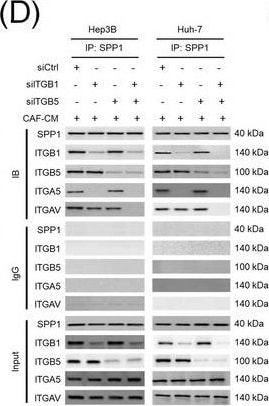
Integrin complexes (integrin alpha V beta 5, alpha 5 beta 1, and alpha V beta 1) on HCC cells were identified as binding proteins of SPP1. (A) Western blotting of SPP1 receptor‐binding protein expression in HCC cells. (B) Measurement of CD44 and integrin expression in Hep3B and Huh‐7 cells by fluorescent intensity. (C‐E) Co‐IP of SPP1 after CAF‐CM treatment, followed by Western blotting for SPP1, ITGA5, ITGAV, ITGB1, and ITGB5 in HCC cells (C), HCC cells silenced for ITGB1 and/or ITGB5 (D), and HCC cells incubated with/without SPP1‐BP (E).Abbreviations: ITGA4, integrin subunit alpha 4; ITGA5, integrin subunit alpha 5; ITGA8, integrin subunit alpha 8; ITGA9, integrin subunit alpha 9; ITGAV, integrin subunit alpha V; ITGB1, integrin subunit beta 1; ITGB3, integrin subunit beta 3; ITGB5, integrin subunit beta ; GAPDH, glyceraldehyde 3‐phosphate dehydrogenase; RFU, relative fluorescence units; IgG, immunoglobulin G; SPP1, secreted phosphoprotein 1; CAF, cancer‐associated fibroblast; CM, culture medium; IB, immunoblotting; siCtrl, negative control; SPP1‐BP, SPP1‐blocking peptide. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/36919193), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Osteopontin/OPN by Western Blot
Integrin complexes (integrin alpha V beta 5, alpha 5 beta 1, and alpha V beta 1) on HCC cells were identified as binding proteins of SPP1. (A) Western blotting of SPP1 receptor‐binding protein expression in HCC cells. (B) Measurement of CD44 and integrin expression in Hep3B and Huh‐7 cells by fluorescent intensity. (C‐E) Co‐IP of SPP1 after CAF‐CM treatment, followed by Western blotting for SPP1, ITGA5, ITGAV, ITGB1, and ITGB5 in HCC cells (C), HCC cells silenced for ITGB1 and/or ITGB5 (D), and HCC cells incubated with/without SPP1‐BP (E).Abbreviations: ITGA4, integrin subunit alpha 4; ITGA5, integrin subunit alpha 5; ITGA8, integrin subunit alpha 8; ITGA9, integrin subunit alpha 9; ITGAV, integrin subunit alpha V; ITGB1, integrin subunit beta 1; ITGB3, integrin subunit beta 3; ITGB5, integrin subunit beta ; GAPDH, glyceraldehyde 3‐phosphate dehydrogenase; RFU, relative fluorescence units; IgG, immunoglobulin G; SPP1, secreted phosphoprotein 1; CAF, cancer‐associated fibroblast; CM, culture medium; IB, immunoblotting; siCtrl, negative control; SPP1‐BP, SPP1‐blocking peptide. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/36919193), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Osteopontin/OPN by Immunohistochemistry
Identification of the CAF‐derived molecules that induce resistance to sorafenib or lenvatinib in patients with HCC. (A) Heat map of 790 genes overexpressed in CAFs. (B) GSEA of the genes overexpressed in CAFs. (C) Process of candidate gene selection. (D) Expression levels of the 9 candidate genes according to sorafenib response in the GSE109211 dataset. (E) Comparison of SPP1 expression between sorafenib responders and non‐responders in the GSE109211 and GSE143233 datasets. (F) Immunofluorescence staining of alpha ‐SMA and SPP1 in tumor tissues from patients with HCC. (G) Comparison of SPP1 expression between CAFs and their paired para‐cancer fibroblasts in the WTS data from the 9 pairs of CAF and para‐cancer fibroblasts. (H) Representative H&E and SPP1 IHC images of the tumor (T), tumor stroma (S) (left), and non‐tumor fibrous tissue (middle) in an SPP1‐high patient. Scale bar, 50 μm. Comparison of SPP1 IHC staining intensity according to tumor location in the SPP1‐high group (right). (I) Representative H&E and SPP1 IHC images of tumor (T), tumor stroma (S) (left), and non‐tumor fibrous tissue (middle) in an SPP1‐low patient. Comparison of SPP1 IHC staining intensity according to tumor location in the SPP1‐low group (right). (Mean ± SEM; unpaired Welch's t‐test; *P < 0.05; **P < 0.01; ***P < 0.001).Abbreviations: HCC, hepatocellular carcinoma; NF, normal fibroblast; CAFs, cancer‐associated fibroblasts; ELISA, enzyme linked immunosorbent assay; H&E, hematoxylin and eosin; alpha ‐SMA, alpha smooth muscle actin; SPP1, secreted phosphoprotein 1; DAPI, 4′, 6‐diamidino‐2‐phenylindole; PAF, para‐cancer fibroblast; S, tumor stroma; T, tumor; IHC, immunohistochemistry. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/36919193), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Osteopontin/OPN by Immunohistochemistry
Identification of the CAF‐derived molecules that induce resistance to sorafenib or lenvatinib in patients with HCC. (A) Heat map of 790 genes overexpressed in CAFs. (B) GSEA of the genes overexpressed in CAFs. (C) Process of candidate gene selection. (D) Expression levels of the 9 candidate genes according to sorafenib response in the GSE109211 dataset. (E) Comparison of SPP1 expression between sorafenib responders and non‐responders in the GSE109211 and GSE143233 datasets. (F) Immunofluorescence staining of alpha ‐SMA and SPP1 in tumor tissues from patients with HCC. (G) Comparison of SPP1 expression between CAFs and their paired para‐cancer fibroblasts in the WTS data from the 9 pairs of CAF and para‐cancer fibroblasts. (H) Representative H&E and SPP1 IHC images of the tumor (T), tumor stroma (S) (left), and non‐tumor fibrous tissue (middle) in an SPP1‐high patient. Scale bar, 50 μm. Comparison of SPP1 IHC staining intensity according to tumor location in the SPP1‐high group (right). (I) Representative H&E and SPP1 IHC images of tumor (T), tumor stroma (S) (left), and non‐tumor fibrous tissue (middle) in an SPP1‐low patient. Comparison of SPP1 IHC staining intensity according to tumor location in the SPP1‐low group (right). (Mean ± SEM; unpaired Welch's t‐test; *P < 0.05; **P < 0.01; ***P < 0.001).Abbreviations: HCC, hepatocellular carcinoma; NF, normal fibroblast; CAFs, cancer‐associated fibroblasts; ELISA, enzyme linked immunosorbent assay; H&E, hematoxylin and eosin; alpha ‐SMA, alpha smooth muscle actin; SPP1, secreted phosphoprotein 1; DAPI, 4′, 6‐diamidino‐2‐phenylindole; PAF, para‐cancer fibroblast; S, tumor stroma; T, tumor; IHC, immunohistochemistry. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/36919193), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Osteopontin/OPN by Immunohistochemistry
Visualization of SPP1, NCAM1, and CD271 expression in bone marrow vascular regions by confocal microscopy.(A) Confocal scan of vascular region in BM biopsies with 3D orthographic cross-section view, co-stained with rabbit anti-NCAM1, mouse anti-CD271, goat anti-SPP1, and DAPI. (B) Intensity profile for all channels in A across a cell of interest in a representative z plane. (C) Single channel data for the florescent markers in A. Scale bars represent 50 μm. Cyan dashed lines indicate vessel surface. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/36876630), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Osteopontin/OPN by Immunohistochemistry
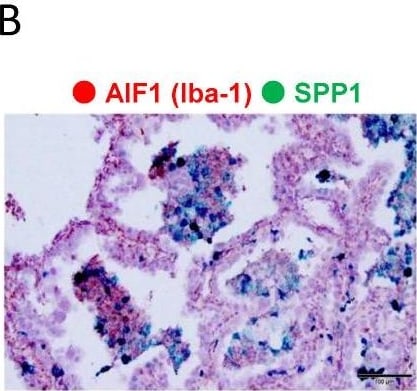
Evaluation of SPP1 expression on cancer cells and macrophages. (A,B). Representative dual ISH (green: SPP1; red: Iba-1) in adenocarcinoma. (C). Representative single-IHC of SPP1 in adenocarcinoma and SCC. (D). Double-IHC of SPP1 and Iba-1 to examine the SPP1 expression level in cancer cells (Iba-1-negative). Double-IHC of SPP1 and PU.1 to examine the SPP1 expression level on TAMs (PU.1-positive in the nucleus). (E). SPP1’s expression level was scored according to the proportion of stained cells as follows: less than 1% staining, score 0; 1% to 49% staining, score 1; more than 50% staining, score 2. The proportions of samples with each score among cancer cells and TAMs in adenocarcinoma and SCC are shown in the pie charts. Image collected and cropped by CiteAb from the following open publication (https://www.mdpi.com/2072-6694/14/18/4374), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Osteopontin/OPN by Western Blot
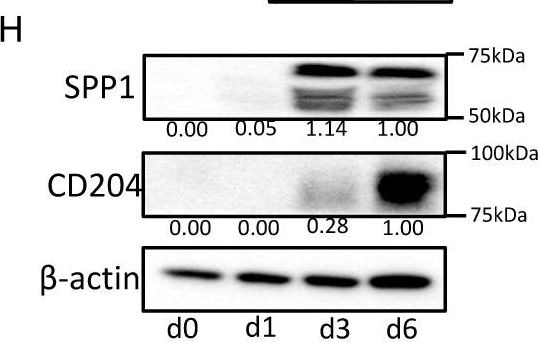
SPP1 expression in macrophages. (A) SPP1 expression in macrophages, THP-1 cells, U937 cells, and 10 lung cancer cell lines was examined by qRT-PCR. (B) Macrophages (blue) and THP-1 cells (green) were stimulated with the CM of cancer cell lines; then, the SPP1 mRNA expression level was evaluated. Control (CT) refers to macrophages or THP-1 without CM of cancer cell lines added. (C) Monocytes were cultured with M-CSF and GM-CSF as described in the Materials and Methods section, and SPP1 mRNA and CD204 mRNA levels were evaluated at different stages of monocyte/macrophage culture (days 0, 3, and 5). (D) Monocytes were cultured with M-CSF or GM-CSF for 5 days, and SPP1 and CD204 expression levels were examined. (E) SPP1′s concentration (ng/mL) in the CM of macrophages cultured with M-CSF or GM-CSF was examined by ELISA at days 1 to 3 (n = 3). (F) Immunocytostaining of SPP1 in the macrophages cultured with M-CSF or GM-CSF at days 0 and 5. (G) SPP1 and CD204 expression levels in THP-1 cells differentiated for 0, 1, 3, or 6 days were examined by qRT-PCR (G) and Western blot analysis (H). *: statistically significant, p value < 0.05. Image collected and cropped by CiteAb from the following open publication (https://www.mdpi.com/2072-6694/14/18/4374), licensed under a CC-BY license. Not internally tested by R&D Systems.Applications for Human Osteopontin/OPN Antibody
Dual RNAscope ISH-IHC Compatible
Sample: Immersion fixed paraffin-embedded sections of human kidney
ELISA
This antibody functions as an ELISA detection antibody when paired with Mouse Anti-Human Osteopontin/OPN Monoclonal Antibody (Catalog # MAB14332R).
This product is intended for assay development on various assay platforms requiring antibody pairs. We recommend the Human Osteopontin (OPN) DuoSet ELISA Kit (Catalog # DY1433) for convenient development of a sandwich ELISA or the Human Osteopontin (OPN) Quantikine ELISA Kit (Catalog # DOST00) for a complete optimized ELISA.
Immunocytochemistry
Sample: Immersion fixed HepG2 human hepatocellular carcinoma cell line
Immunohistochemistry
Sample: Immersion fixed paraffin-embedded sections of human breast cancer tissue
Neutralization
Formulation, Preparation, and Storage
Purification
Reconstitution
Reconstitute at 0.2 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Formulation
*Small pack size (-SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Stability & Storage
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: Osteopontin/OPN
References
- Ann. N.Y. Acad. Sci. (1995) 760, Apr. 21.
- Senger, D.R. et al. (1996) Biochim. Biophys. Acta. 1314:13.
- Agnihotri, R. et al. (2001) J. Biol. Chem. 276:28261.
Long Name
Alternate Names
Gene Symbol
Additional Osteopontin/OPN Products
Product Documents for Human Osteopontin/OPN Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human Osteopontin/OPN Antibody
For research use only
Citations for Human Osteopontin/OPN Antibody
Customer Reviews for Human Osteopontin/OPN Antibody
There are currently no reviews for this product. Be the first to review Human Osteopontin/OPN Antibody and earn rewards!
Have you used Human Osteopontin/OPN Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- ELISA Sample Preparation & Collection Guide
- ELISA Troubleshooting Guide
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- How to Run an R&D Systems DuoSet ELISA
- How to Run an R&D Systems Quantikine ELISA
- How to Run an R&D Systems Quantikine™ QuicKit™ ELISA
- ICC Cell Smear Protocol for Suspension Cells
- ICC Immunocytochemistry Protocol Videos
- ICC for Adherent Cells
- IHC Sample Preparation (Frozen sections vs Paraffin)
- ISH-IHC Protocol for Chromogenic Detection on Formalin Fixed Paraffin Embedded (FFPE) Tissue
- Immunocytochemistry (ICC) Protocol
- Immunocytochemistry Troubleshooting
- Immunofluorescence of Organoids Embedded in Cultrex Basement Membrane Extract
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Fluorescent ICC Staining of Cell Smears - Graphic
- Protocol for the Fluorescent ICC Staining of Cultured Cells on Coverslips - Graphic
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent ICC Staining of Cells on Coverslips
- Protocol for the Preparation and Fluorescent ICC Staining of Non-adherent Cells
- Protocol for the Preparation and Fluorescent ICC Staining of Stem Cells on Coverslips
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- Protocol for the Preparation of a Cell Smear for Non-adherent Cell ICC - Graphic
- Quantikine HS ELISA Kit Assay Principle, Alkaline Phosphatase
- Quantikine HS ELISA Kit Principle, Streptavidin-HRP Polymer
- Sandwich ELISA (Colorimetric) – Biotin/Streptavidin Detection Protocol
- Sandwich ELISA (Colorimetric) – Direct Detection Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: ELISA
- Troubleshooting Guide: Immunohistochemistry
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Associated Pathways