IL-33, also known as NF-HEV and DVS 27, is a 30 kDa proinflammatory protein that may also regulate gene transcription (1-3). DVS 27 was identifed as a gene that is upregulated in vasospastic cerebral arteries (1). NF-HEV was described as a nuclear factor that is preferentially expressed in the endothelial cells of high endothelial venules relative to endothelial cells from other tissues (2). IL-33 was identified based on sequence and structural homology with IL-1 family cytokines (3). DVS 27, NF-HEV, and IL-33 share 100% amino acid sequence identity. IL-33 is constitutively expressed in smooth muscle and airway epithelia. It is upregulated in arterial smooth muscle, dermal fibroblasts, and keratinocytes following IL-1 alpha or IL-1 beta stimulation (1, 3). Similar to IL-1, IL-33 can be cleaved in vitro by caspase-1, generating an N-terminal fragment that is slightly shorter than the C-terminal fragment (3, 4). The N-terminal portion of full length IL-33 contains a predicted bipartite nuclear localization sequence and a homeodomain-like helix-turn-helix DNA binding domain. By immunofluorescence, full length IL-33 localizes to the nucleus in HUVECs and transfectants (2). The C-terminal fragment, corresponding to mature IL-33, binds and triggers signaling through mast cell IL-1 R4/ST2L, a longtime orphan receptor involved in the augmentation of Th2 cell responses (3, 5-7). A ternary signaling complex is formed by the subsequent association of IL-33 and ST2L with IL-1R AcP (8). Stimulation of Th2 polarized lymphocytes with mature IL-33 in vitro induces IL-5 and IL-13 secretion (3). In vivo administration of mature IL-33 promotes increased production of IL-5, IL-13, IgE, and IgA, as well as splenomegaly and inflammatory infiltration of mucosal tissues (3). Full length and mature mouse IL-33 share approximately 55% and 90% aa sequence identity with human and rat IL-33, respectively. Mouse IL-33 shares less than 25% aa sequence identity with other IL-1 family proteins.
Mouse IL‑33 Antibody
R&D Systems | Catalog # AF3626

Key Product Details
Validated by
Biological Validation
Species Reactivity
Validated:
Mouse
Cited:
Human, Mouse, Rat, Transgenic Mouse
Applications
Validated:
Immunohistochemistry, Western Blot, ELISA Capture (Matched Antibody Pair), Neutralization, Intracellular Staining by Flow Cytometry, Immunocytochemistry, CyTOF-ready
Cited:
Immunohistochemistry, Immunohistochemistry-Paraffin, Immunohistochemistry-Frozen, Western Blot, Neutralization, Flow Cytometry, Immunofluorescence, Immunocytochemistry, Immunoprecipitation, Chromatin Immunoprecipitation (ChIP), ELISA Development, ELISA Development (Capture), ELISpot Development, In vivo assay, Neutralization Control
Label
Unconjugated
Antibody Source
Polyclonal Goat IgG
Loading...
Product Specifications
Immunogen
E. coli-derived recombinant mouse IL-33
Ser109-Ile266
Accession # Q8BVZ5
Ser109-Ile266
Accession # Q8BVZ5
Specificity
Detects mouse IL-33 in ELISAs and Western blots. In sandwich immunoassays, less than 2% cross-reactivity with recombinant human IL‑33 is observed.
Clonality
Polyclonal
Host
Goat
Isotype
IgG
Endotoxin Level
<0.10 EU per 1 μg of the antibody by the LAL method.
Scientific Data Images for Mouse IL‑33 Antibody
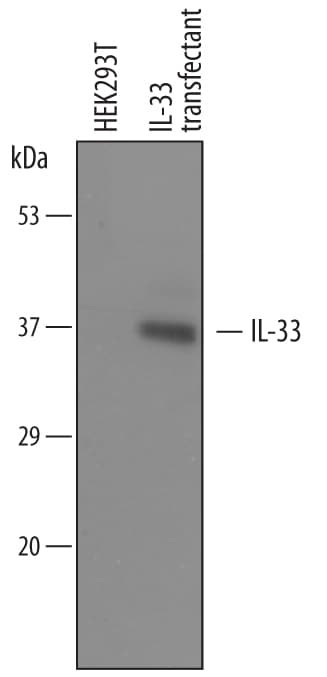
Detection of Mouse IL‑33 by Western Blot.
Western blot shows lysates of HEK293T human embryonic kidney cell line either mock transfected or transfected with full length mouse IL-33. PVDF membrane was probed with 0.4 µg/mL of Goat Anti-Mouse IL-33 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF3626) followed by HRP-conjugated Anti-Goat IgG Secondary Antibody (Catalog # HAF109). A specific band was detected for IL-33 at approximately 37 kDa (as indicated). This experiment was conducted under reducing conditions and using Immunoblot Buffer Group 1.IL‑33 in Mouse Spleen.
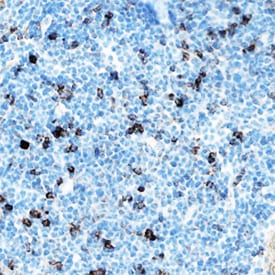
IL-33 was detected in immersion fixed frozen sections of mouse spleen using Goat Anti-Mouse IL-33 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF3626) at 5 µg/mL overnight at 4 °C. Tissue was stained using the Anti-Goat HRP-DAB Cell & Tissue Staining Kit (brown; Catalog # CTS008) and counterstained with hematoxylin (blue). Specific staining was localized to cytoplasm in lymphocytes. View our protocol for Chromogenic IHC Staining of Frozen Tissue Sections.IL‑33 in bEnd.3 Mouse Cell Line.
IL-33 was detected in immersion fixed bEnd.3 mouse endothelioma cell line using Goat Anti-Mouse IL-33 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF3626) at 10 µg/mL for 3 hours at room temperature. Cells were stained using the NorthernLights™ 557-conjugated Anti-Goat IgG Secondary Antibody (red, upper panel; Catalog # NL001) and counterstained with DAPI (blue, lower panel). View our protocol for Fluorescent ICC Staining of Cells on Coverslips.Detection of IL‑33 in bEND.3 cells by Flow Cytometry
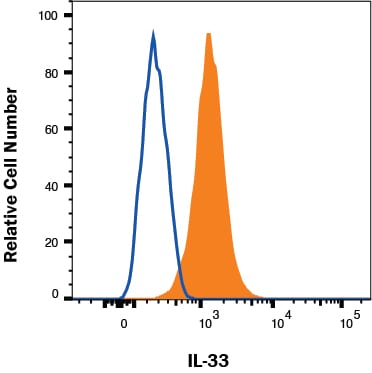
bEND.3 cells were stained with Goat Anti-Mouse IL‑33 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF3626, filled histogram) or isotype control antibody (Catalog # AB-108-C, open histogram) followed by Phycoerythrin-conjugated Anti-Goat IgG Secondary Antibody (Catalog # F0107). To facilitate intracellular staining, cells were fixed with Flow Cytometry Fixation Buffer (Catalog # FC004) and permeabilized with Flow Cytometry Permeabilization/Wash Buffer I (Catalog # FC005). View our protocol for Staining Intracellular Molecules.Detection of IL‑33 in Mouse Splenocytes by Flow Cytometry.
Mouse splenocytes were treated for 5 hours with 50 ng/mL PMA and 1 µg/mL Ca2+ionomycin then stained with Goat Anti-Mouse IL-33 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF3626, filled histogram) or isotype control antibody (Catalog # AB-108-C, open histogram), followed by Phycoerythrin-conjugated Anti-Goat IgG Secondary Antibody (Catalog # F0107). To facilitate intracellular staining, cells were fixed with paraformaldehyde and permeabilized with saponin.Proliferation Induced by IL‑33 and Neutralization by Human IL‑33 Antibody.
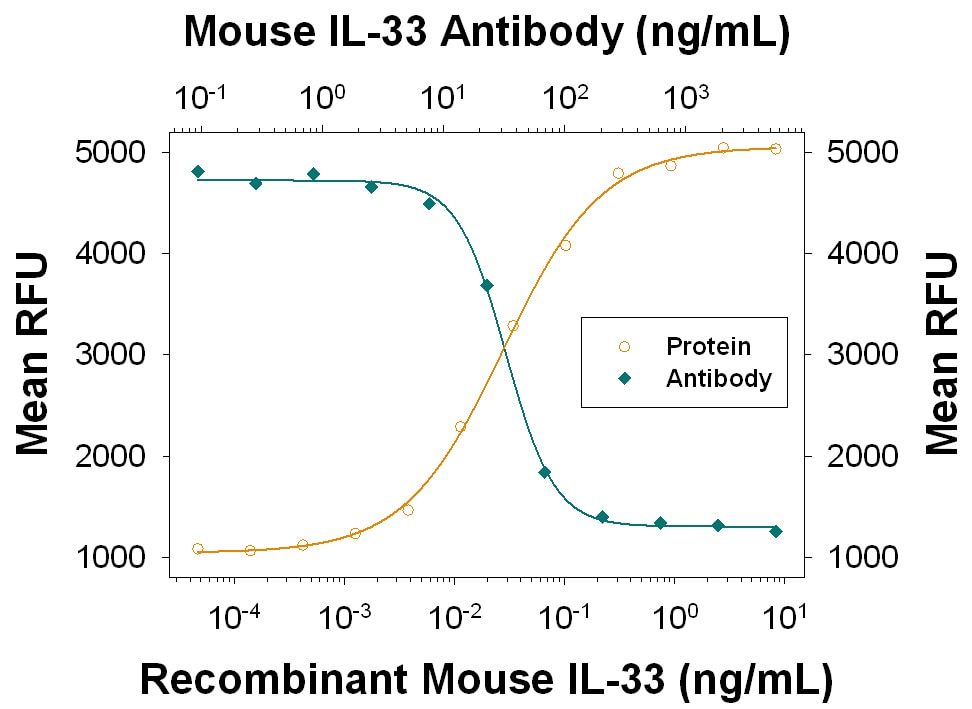
Recombinant Mouse IL-33 (Catalog # 3626-ML) stimulates proliferation in the D10.G4.1 mouse helper T cell line in a dose-dependent manner (orange line), as measured by Resazurin (Catalog # AR002). Proliferation elicited by Recombinant Mouse IL-33 (0.25 ng/mL) is neutralized (green line) by increasing concentrations of Goat Anti-Mouse IL-33 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF3626). The ND50 is typically 10-50 ng/mL.Detection of Mouse IL-33 by Immunohistochemistry
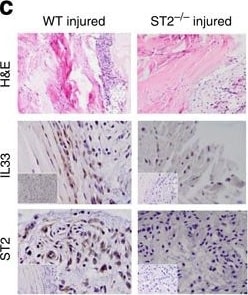
IL-33/ST2 axis in tendon healing in vivo.Kinetics of (a) Il33 and (b) soluble St2 gene expression post injury. Data are mean fold change±s.d. showing relative expression to 18s housekeeping gene (pooled data from four mice per group performed on four sequential occasions, n=16 per condition). *P<0.05, **P<0.01 versus control (WT) mice (ANOVA) +P<0.05,++P<0.01, NS not significant; control (WT) injured versus St2−/− injured. (c) Immunohistochemistry showing IL-33 and ST2 expression in tendon biopsies of WT and St2−/− mice day 1 post injury. IgG control shown in bottom left of pictures. Black horizontal line indicates 50 μm. (d) Col3 mRNA and (e) Collagen 3 protein levels in tendon biopsies of WT and St2−/− mice post injury. (f) Col1 mRNA and (g) Collagen 1 protein levels in tendon biopsies of WT and St2−/− mice post injury. For (d–g) data are mean±s.d. of duplicate samples, representative of four mice per condition (n=16). *P<0.05, **P<0.01 versus control (WT) mice. +P<0.05, ++P<0.01 control (WT) injured versus St2−/− injured mice (ANOVA). mRNA graphs show relative expression to 18s housekeeping gene. (h) Percentage change in tendon strength for WT and St2−/− mice post injury. Data are mean±s.d., representative of four mice per condition (n=16). *P<0.05, **P<0.01 versus control mice. #P<0.05 St2−/− injured versus WT injured mice (Mann–Whitney U-test). Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/25857925), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse IL-33 by Western Blot
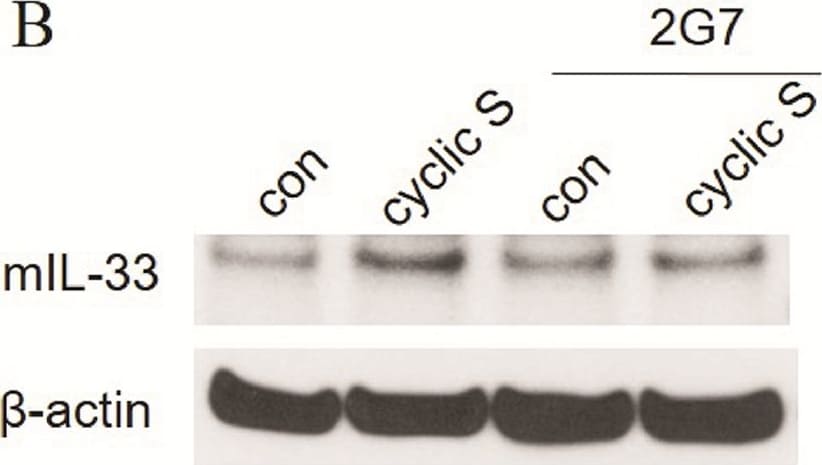
HMGB1 increases IL-33 expression and acts as an autocrine factor in enhancing IL-33 expression in a partial TLR-4 dependent fashion during cyclic stretch.(A) MLE-12 cells were treated with 3 μg/ml HMGB1 or control solution before 6h cyclic stretch and IL-33 production was measured by ELISA. (B) MLE-12 cells were treated with 10 μg/ml HMGB1 neutralizing antibody (2G7) or control solution before 6h cyclic stretch and IL-33 production was detected by Western blot. beta -actin served as loading control. (C) MLE-12 cells transfecting with non-specific control siRNA or TLR-4-specific siRNA were treated with 3 μg/ml HMGB1 or control solution before stretch. IL-33 production in each group was measured with ELISA. **P<0.01, ***P<0.001 when compared between groups denoted by horizontal lines. Each stretching group collected at least from three wells and represented a single experiment, the bar graphs illustrate data representative of three independent experiments. Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/28898270), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human IL-33 by Immunohistochemistry
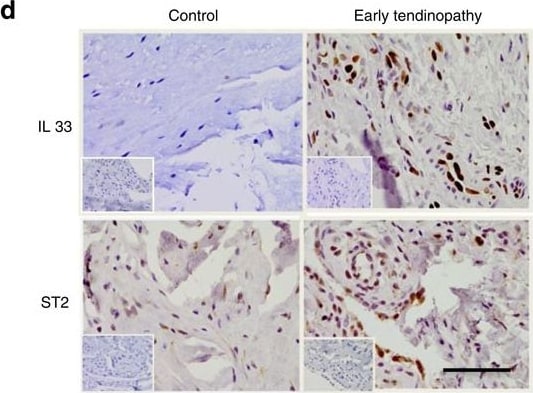
IL-33/ST2 expression in human tendon.(a) IL-33, (b) soluble ST2 (sST2) and (c) mST2 mRNA expression in human tendon samples. Fold change in gene expression of IL-33, sST2 and mST2 in control (semi-membranosus tendon, n=10), torn supraspinatus tendon (established pathology) and matched subscapularis human tendon samples (early pathology; n=17). Data are mean±s.d. relative to the housekeeping gene18S (mean of duplicate analysis). *P<0.05, **P<0.01, ***P<0.001 versus control (Student's t-test). (d) Immunostaining of IL-33 and ST2 in control (n=10), torn tendon (n=17) and early tendinopathy (n=17). Graphs illustrate modified Bonar scoring based on 10 high-power fields. Data are mean±s.d. *P<0.05, **P<0.01 versus control (Student's t-test). Scale bar, 65 μm. (e) Fold change in gene expression of IL33 and ST2 24 h post incubation with tumour necrosis factor (TNF-alpha ), IL-1 beta or in combination depicting relative expression to media alone utilizing housekeeping gene GAPDH. Data are mean±s.d. of triplicate samples, representative of three individual patient samples. *P<0.05, **P<0.01 versus control (media) (Student's t-test). (f) IL-33 immunostaining of human tendon explants cultured for 24 h with medium (control), 100 ng ml−1 TNF alpha or 100 ng ml−1 TNF alpha +100 ng ml−1 IL-1 beta. (g) Fold change in COL1 and COL3 mRNA expression in human tendon explants cultured for 24 h with rhIL-33, relative to housekeeping gene GAPDH. (h) Time course of COL1 and COL3 mRNA expression following incubation with rhIL-33, relative to housekeeping gene GAPDH. (i) Collagen 1 and 3 protein expression in human tendon explants 24 h post incubation with rhIL-33. For (g–i) data are mean±s.d. of triplicate samples, representative of three individual patients. *P<0.05, **P<0.01 versus control (Student's t-test). Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/25857925), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Mouse Mouse IL-33 Antibody by Immunohistochemistry
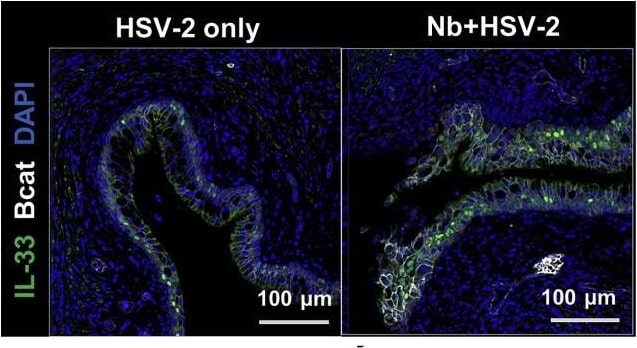
Prior Nb exposure results in earlier and exacerbated HSV-2 pathology. (I and J) (I) Vaginal IL-33 measured by ELISA (day 2) and (J) IF staining of vaginal tissue (day 3; n = 4). Data are representative of two independent experiments with 4–6 mice per group (mean ± SEM). Statistical significance was calculated by two-way analysis of variance (ANOVA) with Bonferroni correction for multiple comparisons and Mann-Whitney t test. ∗p ≤ 0.05, ∗∗p ≤ 0.01, ∗∗p ≤ 0.01, ∗∗∗p ≤ 0.001, ns, not significant. Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/33857419), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of IL-33 by Immunocytochemistry/ Immunofluorescence
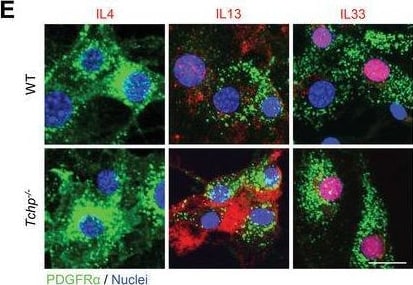
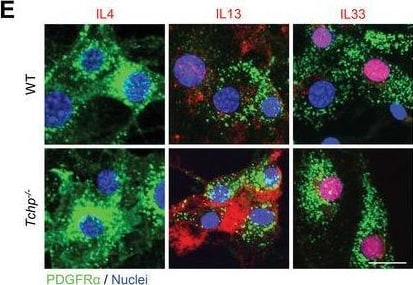
IL13 expression in FAPs is upregulated in Tchp−/− mice. A) qRT‐PCR analysis of RNA isolated from whole TA muscle of WT and Tchp−/− mice (n = 3 or 4) at 3 dpi. B) Immunofluorescence for IL13 (red), hematopoietic cells (CD45, green), and nuclei (Hoechst33342, blue) in TA muscle cross‐sections of WT and Tchp−/− mice at 3 dpi. Lower magnification images are shown in Figure S6A, Supporting Information. Scale bars, 10 µm. C) Quantification of the IL4‐, IL13‐, or IL33‐stained area per 40× view in (B) and Figure S6A,B (Supporting Information) (n = 6 or 7 fields each). D) Quantification of the IL13‐stained area per 40× view excluding the CD45‐stained area in (B) (n = 7 fields each). E) Immunofluorescence for IL4 (red), IL13 (red), and IL33 (red), PDGFR alpha + (green), and nuclei (Hoechst33342, blue) in PDGFR alpha + FAPs isolated from hindlimbs of WT and Tchp−/‐ mice. Scale bars: 20 µm. All data are the mean ± S.D from 3 to 5 mice. *p < 0.05, **p < 0.01, N.S., not significant; two‐tailed unpaired Student's t‐tests. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/36373718), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of IL-33 by Immunocytochemistry/ Immunofluorescence
IL13 expression in FAPs is upregulated in Tchp−/− mice. A) qRT‐PCR analysis of RNA isolated from whole TA muscle of WT and Tchp−/− mice (n = 3 or 4) at 3 dpi. B) Immunofluorescence for IL13 (red), hematopoietic cells (CD45, green), and nuclei (Hoechst33342, blue) in TA muscle cross‐sections of WT and Tchp−/− mice at 3 dpi. Lower magnification images are shown in Figure S6A, Supporting Information. Scale bars, 10 µm. C) Quantification of the IL4‐, IL13‐, or IL33‐stained area per 40× view in (B) and Figure S6A,B (Supporting Information) (n = 6 or 7 fields each). D) Quantification of the IL13‐stained area per 40× view excluding the CD45‐stained area in (B) (n = 7 fields each). E) Immunofluorescence for IL4 (red), IL13 (red), and IL33 (red), PDGFR alpha + (green), and nuclei (Hoechst33342, blue) in PDGFR alpha + FAPs isolated from hindlimbs of WT and Tchp−/‐ mice. Scale bars: 20 µm. All data are the mean ± S.D from 3 to 5 mice. *p < 0.05, **p < 0.01, N.S., not significant; two‐tailed unpaired Student's t‐tests. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/36373718), licensed under a CC-BY license. Not internally tested by R&D Systems.Applications for Mouse IL‑33 Antibody
Application
Recommended Usage
CyTOF-ready
Ready to be labeled using established conjugation methods. No BSA or other carrier proteins that could interfere with conjugation.
Immunocytochemistry
5-15 µg/mL
Sample: Immersion fixed bEnd.3 mouse endothelioma cell line
Sample: Immersion fixed bEnd.3 mouse endothelioma cell line
Immunohistochemistry
5-15 µg/mL
Sample: Immersion fixed frozen sections of mouse spleen
Sample: Immersion fixed frozen sections of mouse spleen
Intracellular Staining by Flow Cytometry
0.25 µg/106 cells
Sample: bEND.3 mouse endothelioma cell line or Mouse splenocytes treated with PMA and Ca2+ ionomycin, fixed with paraformaldehyde, and permeabilized with saponin
Sample: bEND.3 mouse endothelioma cell line or Mouse splenocytes treated with PMA and Ca2+ ionomycin, fixed with paraformaldehyde, and permeabilized with saponin
Western Blot
0.4 µg/mL
Sample: HEK293T human embryonic kidney cell line transfected with full length mouse IL-33
Sample: HEK293T human embryonic kidney cell line transfected with full length mouse IL-33
Neutralization
Measured by its ability to neutralize IL‑33-induced proliferation in the D10.G4.1 mouse helper T cell line. Schmitz, J. et al. (2005) Immunity 23:479. The Neutralization Dose (ND50) is typically 10-50 ng/mL in the presence of 0.25 ng/mL Recombinant Mouse IL‑33.
Mouse IL-33 Sandwich Immunoassay
Please Note: Optimal dilutions of this antibody should be experimentally determined.
Reviewed Applications
Read 7 reviews rated 4.6 using AF3626 in the following applications:
Flow Cytometry Panel Builder
Bio-Techne Knows Flow Cytometry
Save time and reduce costly mistakes by quickly finding compatible reagents using the Panel Builder Tool.
Advanced Features
- Spectra Viewer - Custom analysis of spectra from multiple fluorochromes
- Spillover Popups - Visualize the spectra of individual fluorochromes
- Antigen Density Selector - Match fluorochrome brightness with antigen density
Formulation, Preparation, and Storage
Purification
Antigen Affinity-purified
Reconstitution
Reconstitute at 0.2 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. See Certificate of Analysis for details.
*Small pack size (-SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
*Small pack size (-SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: IL-33
References
- Onda, H. et al. (1999) J. Cereb. Blood Flow Metab. 19:1279.
- Baekkevold, E.S. et al. (2003) Am. J. Pathol. 163:69.
- Schmitz, J. et al. (2005) Immunity 23:479.
- Black, R.A. et al. (1989) J. Biol. Chem. 264:5323.
- Xu, D. et al. (1998) J. Exp. Med. 187:787.
- Lohning, M. et al. (1998) Proc. Natl. Acad. Sci. USA 95:6930.
- Dinarello, C.A. (2005) Immunity 23:461.
- Chackerian, A.A. et al. (2007) J. Immunol. 179:2551.
Long Name
Interleukin 33
Alternate Names
C9orf26, DVS27, IL33, NF-HEV
Gene Symbol
IL33
UniProt
Additional IL-33 Products
Product Documents for Mouse IL‑33 Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Mouse IL‑33 Antibody
For research use only
Related Research Areas
Citations for Mouse IL‑33 Antibody
Customer Reviews for Mouse IL‑33 Antibody (7)
4.6 out of 5
7 Customer Ratings
Have you used Mouse IL‑33 Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
Showing
1
-
5 of
7 reviews
Showing All
Filter By:
-
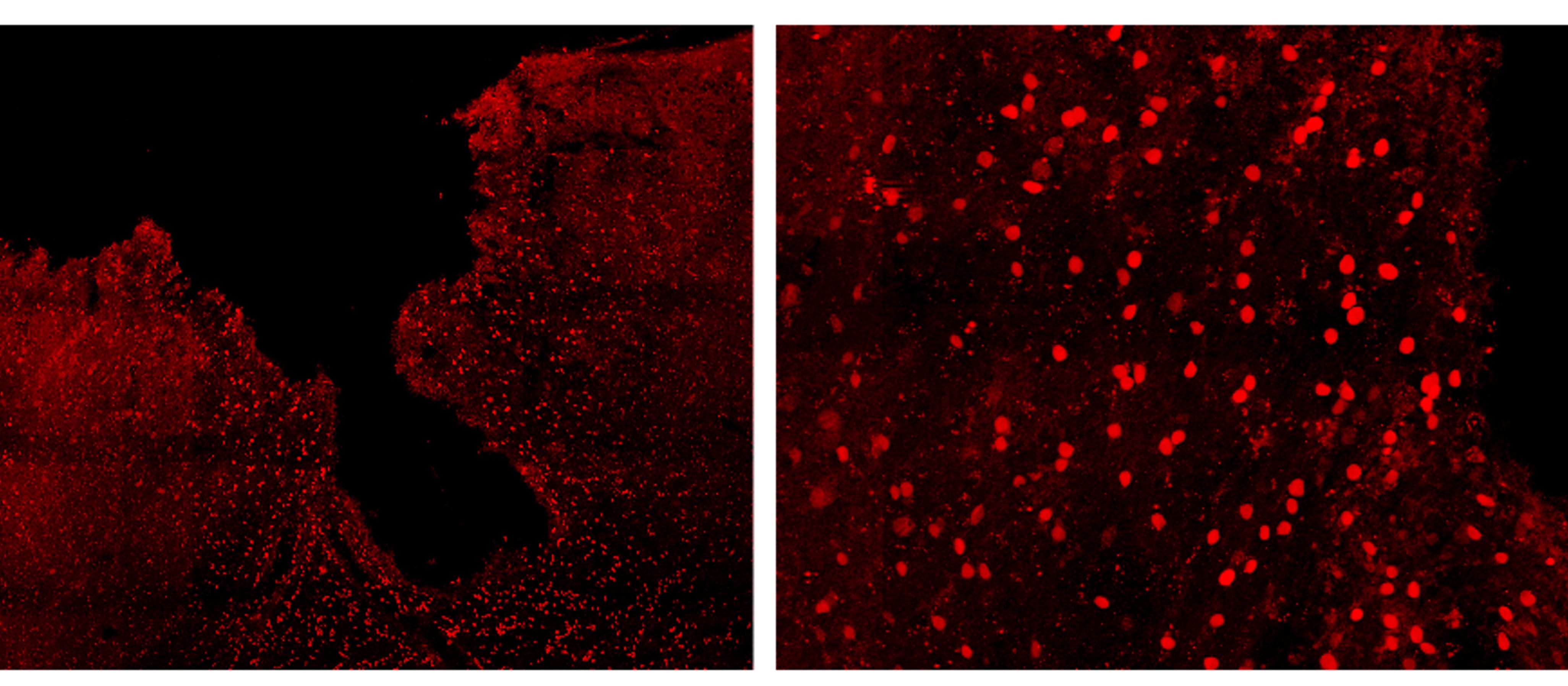
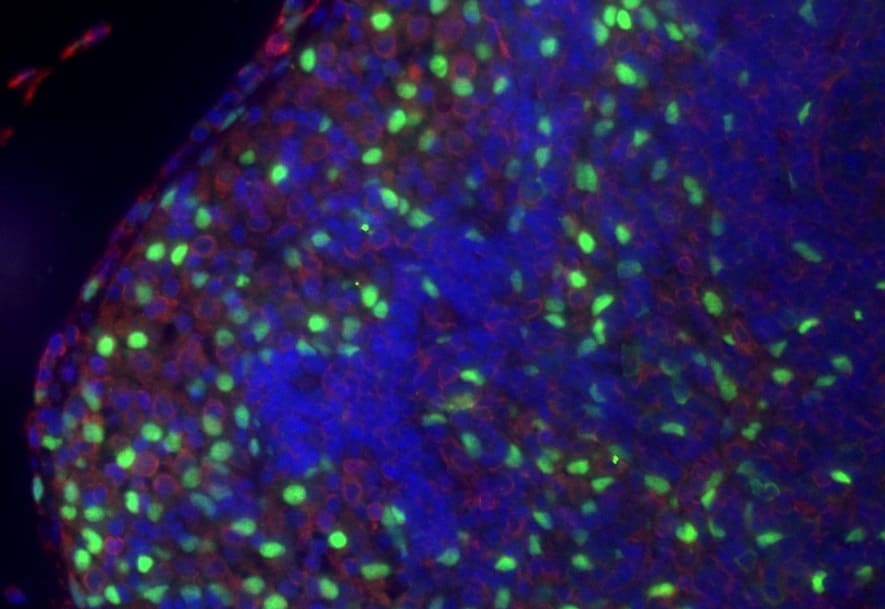
Application: Immunocytochemistry/ImmunofluorescenceSample Tested: Adult brainSpecies: MouseVerified Customer | Posted 10/13/2023Expression of IL-33 (in red) in the mouse cortex, 7 days after traumatic brain injury
-
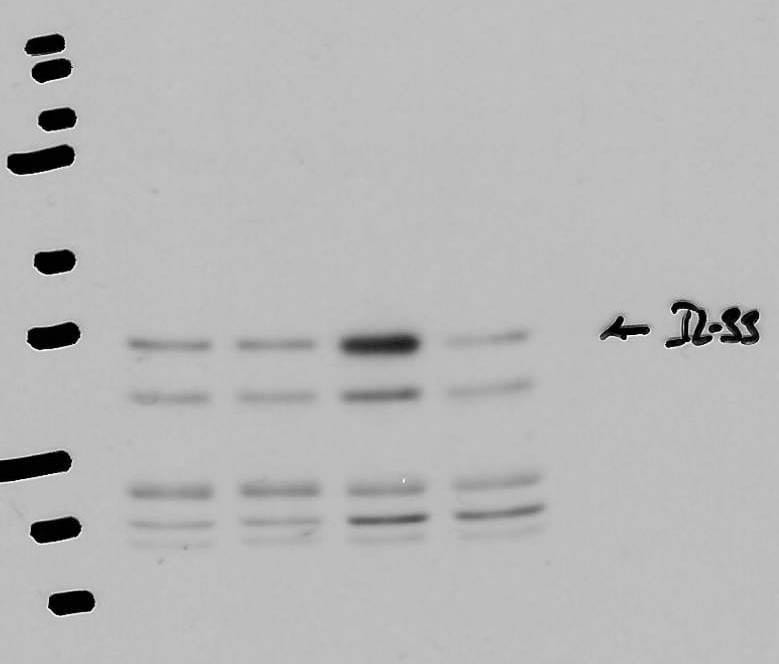
Application: Western BlotSample Tested: Aorta tissueSpecies: MouseVerified Customer | Posted 04/06/2021
-
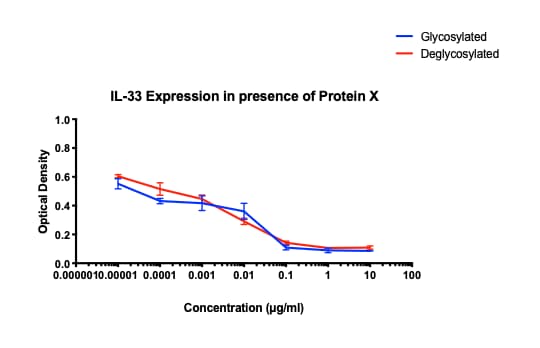
Application: ELISASample Tested: Mouse lymphocytesSpecies: MouseVerified Customer | Posted 01/28/2020Incubated with Protein X suspected of blocking activity of mIL-33
-
Application: Immunocytochemistry/ImmunofluorescenceSample Tested: Spleen tissueSpecies: MouseVerified Customer | Posted 10/06/2019green: IL-33, red: vimentin
-
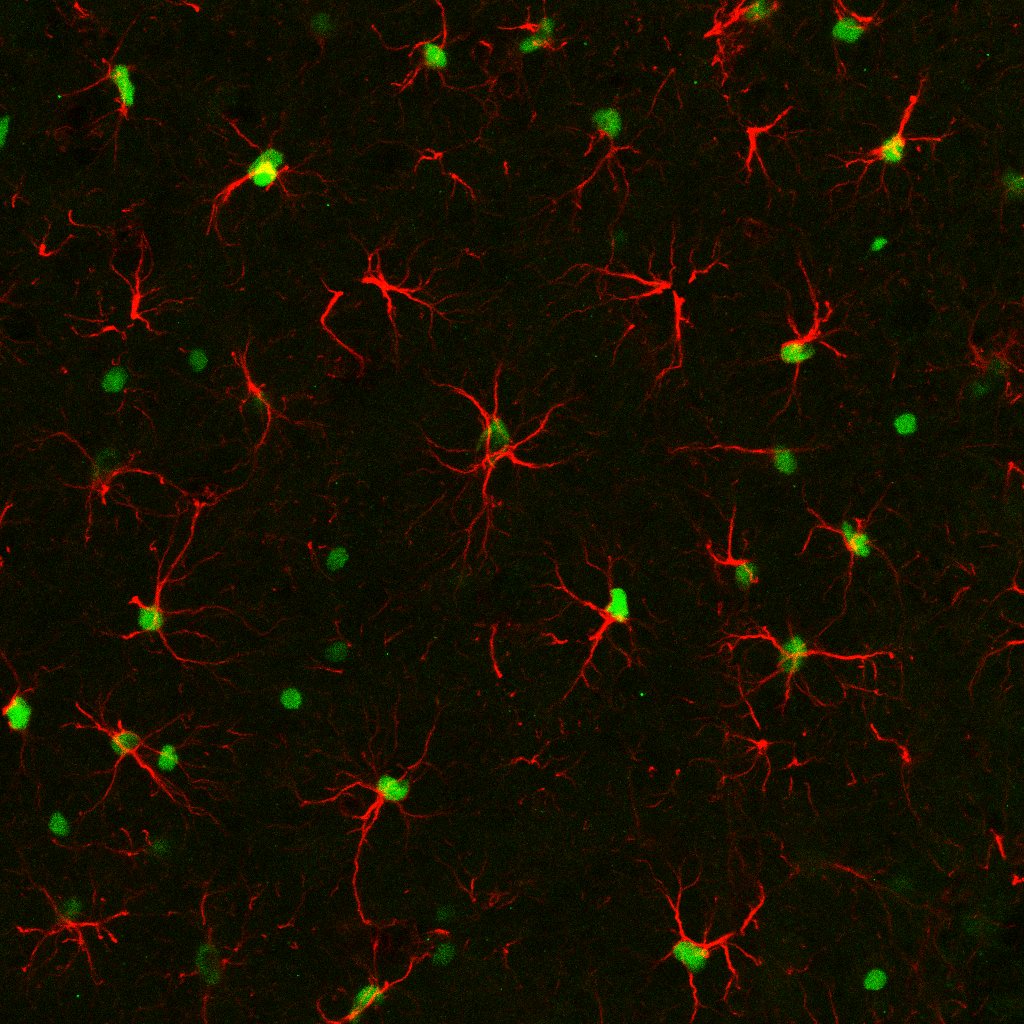
Application: Immunohistochemistry-FrozenSample Tested: 4% PFA-PBS fixed frozen sections from murine brainSpecies: MouseVerified Customer | Posted 03/14/2018PFA-fixed mouse brain sections were stained with Anti-IL-33(1:100) and GFAP antibodies. This image shows IL-33 (Green) and GFAP (Red) colocalization in the mouse brain.
-
Application: ELISASample Tested: Lung tissueSpecies: MouseVerified Customer | Posted 09/07/2017
-
Application: ImmunocytochemistrySample Tested: See PMID 23172891Species: MouseVerified Customer | Posted 01/08/2015
There are no reviews that match your criteria.
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- 7-Amino Actinomycin D (7-AAD) Cell Viability Flow Cytometry Protocol
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Extracellular Membrane Flow Cytometry Protocol
- Flow Cytometry Protocol for Cell Surface Markers
- Flow Cytometry Protocol for Staining Membrane Associated Proteins
- Flow Cytometry Staining Protocols
- Flow Cytometry Troubleshooting Guide
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- ICC Cell Smear Protocol for Suspension Cells
- ICC Immunocytochemistry Protocol Videos
- ICC for Adherent Cells
- IHC Sample Preparation (Frozen sections vs Paraffin)
- Immunocytochemistry (ICC) Protocol
- Immunocytochemistry Troubleshooting
- Immunofluorescence of Organoids Embedded in Cultrex Basement Membrane Extract
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Intracellular Flow Cytometry Protocol Using Alcohol (Methanol)
- Intracellular Flow Cytometry Protocol Using Detergents
- Intracellular Nuclear Staining Flow Cytometry Protocol Using Detergents
- Intracellular Staining Flow Cytometry Protocol Using Alcohol Permeabilization
- Intracellular Staining Flow Cytometry Protocol Using Detergents to Permeabilize Cells
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Propidium Iodide Cell Viability Flow Cytometry Protocol
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Liperfluo
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Characterization of Human Th22 Cells
- Protocol for the Characterization of Human Th9 Cells
- Protocol for the Fluorescent ICC Staining of Cell Smears - Graphic
- Protocol for the Fluorescent ICC Staining of Cultured Cells on Coverslips - Graphic
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent ICC Staining of Cells on Coverslips
- Protocol for the Preparation and Fluorescent ICC Staining of Non-adherent Cells
- Protocol for the Preparation and Fluorescent ICC Staining of Stem Cells on Coverslips
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- Protocol for the Preparation of a Cell Smear for Non-adherent Cell ICC - Graphic
- Protocol: Annexin V and PI Staining by Flow Cytometry
- Protocol: Annexin V and PI Staining for Apoptosis by Flow Cytometry
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Fluorokine Flow Cytometry Kits
- Troubleshooting Guide: Immunohistochemistry
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...
Associated Pathways
 Innate Lymphoid Cell Differentiation Pathways
Innate Lymphoid Cell Differentiation Pathways
 Th2 Differentiation Pathway
Th2 Differentiation Pathway