Granzyme B is a member of the granzyme family of the serine proteases found specifically in the cytotoxic granules of cytotoxic T lymphocytes (CTL) and natural killer (NK) cells (1, 2). Granzyme B plays an essential role in granule-mediated apoptosis and may have additional roles in rheumatoid arthritis and in bacterial and viral infections (3). It activates various caspases and cleaves proteins such as aggrecan (3). Human Granzyme B is synthesized as a precursor (247 residues) with a signal peptide (residues 1-18), a pro peptide (residues 19-20), and a mature chain (residues 21-247) (4-6). The recombinant human (rh) Granzyme B consisting of residues 19-247 was expressed and purified. After being activated by active cathepsin C, rhGranzyme B cleaves a thioester substrate described previously (3).
Human Granzyme B Antibody
R&D Systems | Catalog # MAB2906

Key Product Details
Species Reactivity
Validated:
Cited:
Applications
Validated:
Cited:
Label
Antibody Source
Product Specifications
Immunogen
Gly19-Tyr247
Accession # P10144
Specificity
Clonality
Host
Isotype
Scientific Data Images for Human Granzyme B Antibody
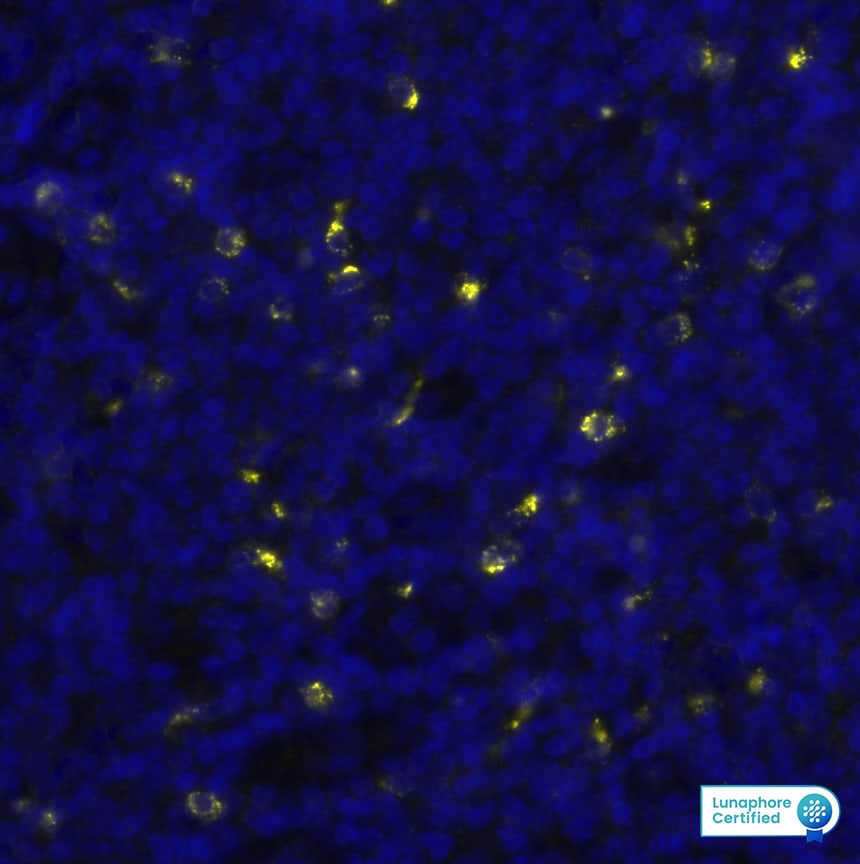
Granzyme B Antibody in Human Tonsil via seqIF™ staining on COMET™
Granzyme B was detected in immersion fixed paraffin-embedded sections of human Tonsil using Mouse Anti-Human Granzyme B, Monoclonal Antibody (Catalog #MAB2906) at a 25ug/mL concentration at 37 ° Celsius for 4 minutes. Before incubation with the primary antibody, tissue underwent an all-in-one dewaxing and antigen retrieval preprocessing using PreTreatment Module (PT Module) and Dewax and HIER Buffer H (pH 9; Epredia Catalog # TA-999-DHBH). Tissue was stained using the Alexa Fluor™ 647 Goat anti-Mouse IgG Secondary Antibody at 1:200 at 37 ° Celsius for 2 minutes. (Yellow; Lunaphore Catalog # DR647MS) and counterstained with DAPI (blue; Lunaphore Catalog # DR100). Specific staining was localized to the cytoplasm. Protocol available in COMET™ Panel Builder.Detection of Human Granzyme B by Western Blot.
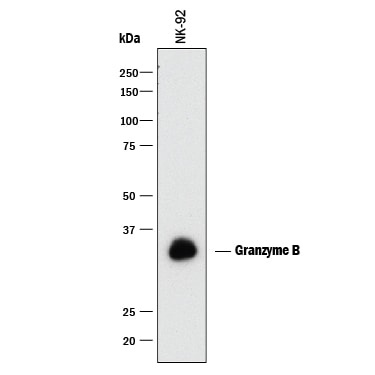
Western blot shows lysate of NK-92 human natural killer lymphoma cell line. PVDF membrane was probed with 0.5 µg/mL of Mouse Anti-Human Granzyme B Monoclonal Antibody (Catalog # MAB2906) followed by HRP-conjugated Anti-Mouse IgG Secondary Antibody (Catalog # HAF018). A specific band was detected for Granzyme B at approximately 32-35 kDa (as indicated). This experiment was conducted under reducing conditions and using Immunoblot Buffer Group 1.Granzyme B in Human PBMCs.
Granzyme B was detected in immersion fixed human peripheral blood mononuclear cells (PBMCs) using Mouse Anti-Human Granzyme B Monoclonal Antibody (Catalog # MAB2906) at 8 µg/mL for 3 hours at room temperature. Cells were stained using the NorthernLights™ 557-conjugated Anti-Mouse IgG Secondary Antibody (red; Catalog # NL007) and counterstained with DAPI (blue). Specific staining was localized to cytoplasm. View our protocol for Fluorescent ICC Staining of Non-adherent Cells.Detection of Human Granzyme B by Simple WesternTM.
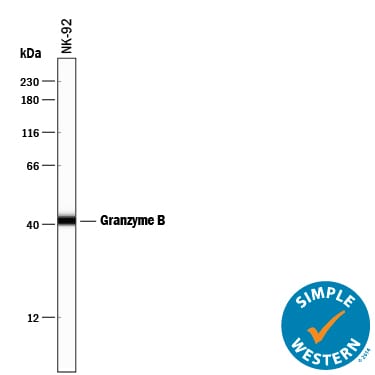
Simple Western lane view shows lysates of NK‑92 human natural killer lymphoma cell line, loaded at 0.2 mg/mL. A specific band was detected for Granzyme B at approximately 42 kDa (as indicated) using 5 µg/mL of Mouse Anti-Human Granzyme B Monoclonal Antibody (Catalog # MAB2906). This experiment was conducted under reducing conditions and using the 12-230 kDa separation system.Detection of Granzyme B in human tonsil.
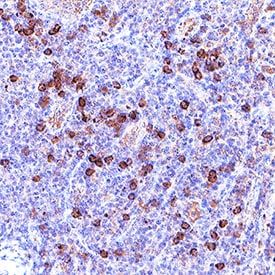
Granzyme B was detected in immersion fixed paraffin-embedded sections of human tonsil using Mouse Anti-Human Granzyme B Monoclonal Antibody (Catalog # MAB2906) at 5 µg/mL for 1 hour at room temperature followed by incubation with the Anti-Mouse IgG VisUCyte™ HRP Polymer Antibody (Catalog # VC001). Before incubation with the primary antibody, tissue was subjected to heat-induced epitope retrieval using VisUCyte Antigen Retrieval Reagent-Basic (Catalog # VCTS021). Tissue was stained using DAB (brown) and counterstained with hematoxylin (blue). Specific staining was localized to cytoplasm in lymphocytes. View our protocol for IHC Staining with VisUCyte HRP Polymer Detection Reagents.Detection of Human Granzyme B by Simple Western
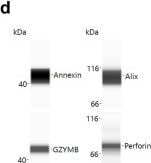
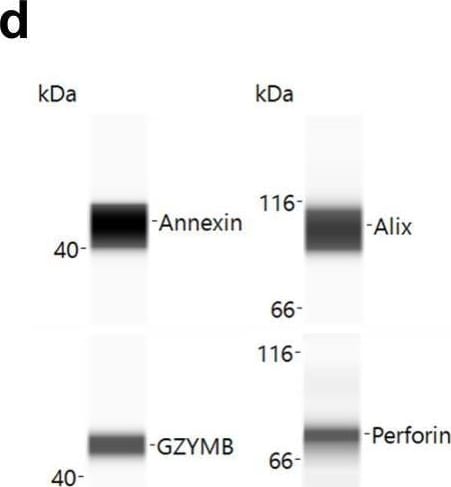
Patient derived NK cells can be expanded in vitro and NKEVs contain protein and nucleic acid cargo reflecting the cells of origin. NK cells were isolated from patient PBMCs via negative selection and EVs were isolated with size exclusion chromatography from the NK cell culture medium (n=20, 10 �pre� and 10 �post) (a). Representative image of cytokine profiling (n=12) of the secretome showed the presence of classic chemokines (CCL1, -5) and cytokines (IFNy, GM-CSF) (b). NKEVs were 100�200 nm in diameter post SEC (n=20) (c) and express canonical EV markers and cytotoxic NK proteins (d). Whole transcriptome sequencing (n=21; 20 patient NKEV and 1 control pool NKEV) found the majority of NKEV RNA cargo is protein coding and long noncoding transcripts (e). Differential gene expression (DE) analysis of LUAD/LUSC NKEVs identified a small number of significantly DE transcripts between LUAD and LUSC, such as ERAP2, which is upregulated in LUAD NKEVs (f). Mass spectrometry proteomics analysis (n=21; 20 patient NKEV and 1 control pool NKEV) of NKEV cargo identified over 4000 proteins (g), and modestly differentially expressed proteins between LUAD and LUSC groups (h). (b) is one representative dot blot, PT007-post and (d) is one representative Western blot, PT007-post. (c) shows NTA analysis for all individual samples in grey, with the mean distribution in red and SEM error bars. Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/40821787/), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human Granzyme B by Simple Western
Patient derived NK cells can be expanded in vitro and NKEVs contain protein and nucleic acid cargo reflecting the cells of origin. NK cells were isolated from patient PBMCs via negative selection and EVs were isolated with size exclusion chromatography from the NK cell culture medium (n=20, 10 “pre” and 10 “post) (a). Representative image of cytokine profiling (n=12) of the secretome showed the presence of classic chemokines (CCL1, -5) and cytokines (IFNy, GM-CSF) (b). NKEVs were 100–200 nm in diameter post SEC (n=20) (c) and express canonical EV markers and cytotoxic NK proteins (d). Whole transcriptome sequencing (n=21; 20 patient NKEV and 1 control pool NKEV) found the majority of NKEV RNA cargo is protein coding and long noncoding transcripts (e). Differential gene expression (DE) analysis of LUAD/LUSC NKEVs identified a small number of significantly DE transcripts between LUAD and LUSC, such as ERAP2, which is upregulated in LUAD NKEVs (f). Mass spectrometry proteomics analysis (n=21; 20 patient NKEV and 1 control pool NKEV) of NKEV cargo identified over 4000 proteins (g), and modestly differentially expressed proteins between LUAD and LUSC groups (h). (b) is one representative dot blot, PT007-post and (d) is one representative Western blot, PT007-post. (c) shows NTA analysis for all individual samples in grey, with the mean distribution in red and SEM error bars. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/40821787), licensed under a CC-BY license. Not internally tested by R&D Systems.Applications for Human Granzyme B Antibody
COMET
CyTOF-ready
Immunocytochemistry
Sample: Immersion fixed human peripheral blood mononuclear cells
Immunohistochemistry
Sample: Immersion fixed paraffin-embedded sections of human tonsil
Intracellular Staining by Flow Cytometry
Sample: NK‑92 human natural killer lymphoma cell line fixed with paraformaldehyde and permeabilized with saponin
Multiplex Immunofluorescence
Sample: Immersion fixed paraffin-embedded sections of human tonsil
Simple Western
Sample: NK‑92 human natural killer lymphoma cell line
Western Blot
Sample: NK‑92 human natural killer lymphoma cell line
Flow Cytometry Panel Builder
Bio-Techne Knows Flow Cytometry
Save time and reduce costly mistakes by quickly finding compatible reagents using the Panel Builder Tool.
Advanced Features
- Spectra Viewer - Custom analysis of spectra from multiple fluorochromes
- Spillover Popups - Visualize the spectra of individual fluorochromes
- Antigen Density Selector - Match fluorochrome brightness with antigen density
Formulation, Preparation, and Storage
Purification
Reconstitution
Reconstitute at 0.5 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Formulation
*Small pack size (-SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Stability & Storage
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: Granzyme B
References
- Kam, C-M. et al. (2000) Biochim. Biophys. Acta 1477:307.
- Smyth, M.J. et al. (1996) J. Leukoc. Biol. 60:555.
- Froelich, C.J. (2004) in Handbook of Proteolytic Enzymes, Barrett, A.J. et al. eds. pp. 1549.
- Schmid, J. and C. Weissman (1987) J. Immunol. 139:250.
- Caputo, A. et al. (1988) J. Biol. Chem. 263:6363.
- Trapani, J.A. et al. (1988) Proc. Natl. Acad. Sci. USA 85:6924.
Alternate Names
Gene Symbol
UniProt
Additional Granzyme B Products
Product Documents for Human Granzyme B Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human Granzyme B Antibody
For research use only
Citations for Human Granzyme B Antibody
Customer Reviews for Human Granzyme B Antibody
There are currently no reviews for this product. Be the first to review Human Granzyme B Antibody and earn rewards!
Have you used Human Granzyme B Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- 7-Amino Actinomycin D (7-AAD) Cell Viability Flow Cytometry Protocol
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Extracellular Membrane Flow Cytometry Protocol
- Flow Cytometry Protocol for Cell Surface Markers
- Flow Cytometry Protocol for Staining Membrane Associated Proteins
- Flow Cytometry Staining Protocols
- Flow Cytometry Troubleshooting Guide
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- ICC Cell Smear Protocol for Suspension Cells
- ICC Immunocytochemistry Protocol Videos
- ICC for Adherent Cells
- IHC Sample Preparation (Frozen sections vs Paraffin)
- Immunocytochemistry (ICC) Protocol
- Immunocytochemistry Troubleshooting
- Immunofluorescence of Organoids Embedded in Cultrex Basement Membrane Extract
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Intracellular Flow Cytometry Protocol Using Alcohol (Methanol)
- Intracellular Flow Cytometry Protocol Using Detergents
- Intracellular Nuclear Staining Flow Cytometry Protocol Using Detergents
- Intracellular Staining Flow Cytometry Protocol Using Alcohol Permeabilization
- Intracellular Staining Flow Cytometry Protocol Using Detergents to Permeabilize Cells
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Propidium Iodide Cell Viability Flow Cytometry Protocol
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Liperfluo
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Characterization of Human Th22 Cells
- Protocol for the Characterization of Human Th9 Cells
- Protocol for the Fluorescent ICC Staining of Cell Smears - Graphic
- Protocol for the Fluorescent ICC Staining of Cultured Cells on Coverslips - Graphic
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent ICC Staining of Cells on Coverslips
- Protocol for the Preparation and Fluorescent ICC Staining of Non-adherent Cells
- Protocol for the Preparation and Fluorescent ICC Staining of Stem Cells on Coverslips
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- Protocol for the Preparation of a Cell Smear for Non-adherent Cell ICC - Graphic
- Protocol: Annexin V and PI Staining by Flow Cytometry
- Protocol: Annexin V and PI Staining for Apoptosis by Flow Cytometry
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Fluorokine Flow Cytometry Kits
- Troubleshooting Guide: Immunohistochemistry
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars