Key Product Details
Validated by
Biological Validation
Species Reactivity
Validated:
Human
Cited:
Human, Mouse, Porcine
Applications
Validated:
Immunohistochemistry, Western Blot, ELISA Capture (Matched Antibody Pair), Neutralization, Dual RNAscope ISH-IHC Compatible
Cited:
Immunohistochemistry, Immunohistochemistry-Paraffin, Western Blot, ELISA, Neutralization, Immunocytochemistry, Antibody Array Development, Bioassay, ELISA Capture, ELISA Development, ELISA Development (Capture), Functional Assay, Luminex Development
Label
Unconjugated
Antibody Source
Monoclonal Mouse IgG2B Clone # 23007
Loading...
Product Specifications
Immunogen
E. coli-derived recombinant human CCL2/JE/MCP-1
Specificity
Detects human CCL2/JE/MCP-1 in ELISAs and Western blots. In ELISAs, this antibody does not cross-react with recombinant mouse (rm) CCL2, 3, 4, rhCCL3, 4, 5, 7, or 8.
Clonality
Monoclonal
Host
Mouse
Isotype
IgG2B
Endotoxin Level
<0.10 EU per 1 μg of the antibody by the LAL method.
Scientific Data Images for Human CCL2/JE/MCP‑1 Antibody
CCL2/JE/MCP‑1 in Human Crohn's Disease Intestine.
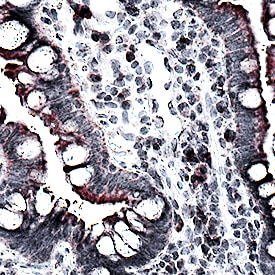
CCL2/JE/MCP-1 was detected in immersion fixed paraffin-embedded sections of human Crohn's disease intestine using Mouse Anti-Human CCL2/JE/MCP-1 Monoclonal Antibody (Catalog # MAB679) at 15 µg/mL for 1 hour at room temperature followed by incubation with the Anti-Mouse IgG VisUCyte™ HRP Polymer Antibody (VC001). Before incubation with the primary antibody, tissue was subjected to heat-induced epitope retrieval using Antigen Retrieval Reagent-Basic (CTS013). Tissue was stained using DAB (brown) and counterstained with hematoxylin (blue). Specific staining was localized to cell membranes and cytoplasm. View our protocol for IHC Staining with VisUCyte HRP Polymer Detection Reagents.Chemotaxis Induced by CCL2/MCP-1 and Neutralization by Human CCL2/MCP‑1 Antibody.
Recombinant Human CCL2/MCP-1 (279-MC) chemoattracts the BaF3 mouse pro-B cell line transfected with human CCR2A in a dose-dependent manner (orange line). The amount of cells that migrated through to the lower chemotaxis chamber was measured by Resazurin (AR002). Chemotaxis elicited by Recombinant Human CCL2/MCP-1 (75 ng/mL) is neutralized (green line) by increasing concentrations of Human CCL2/MCP-1 Monoclonal Antibody (Catalog # MAB679). The ND50 is typically 0.5-2.0 µg/mL.Detection of Human CCL2/JE/MCP-1 by Immunohistochemistry
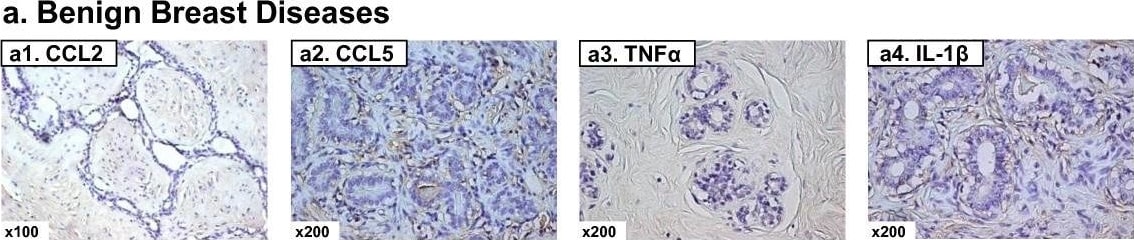
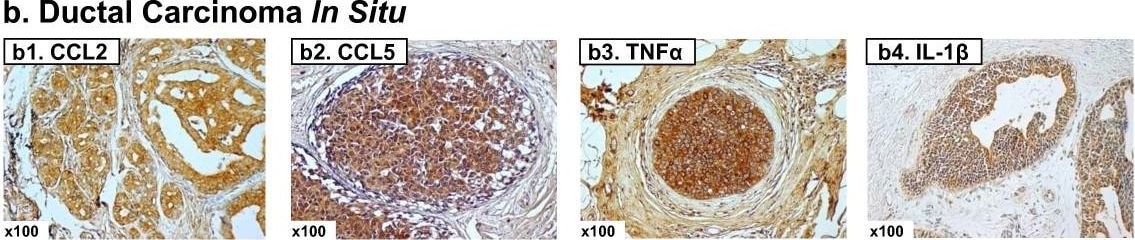
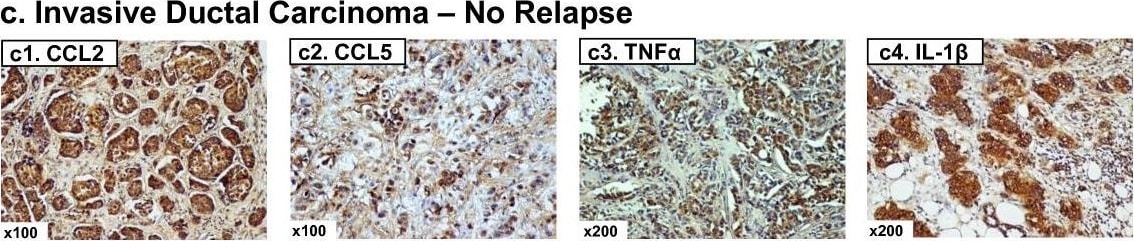
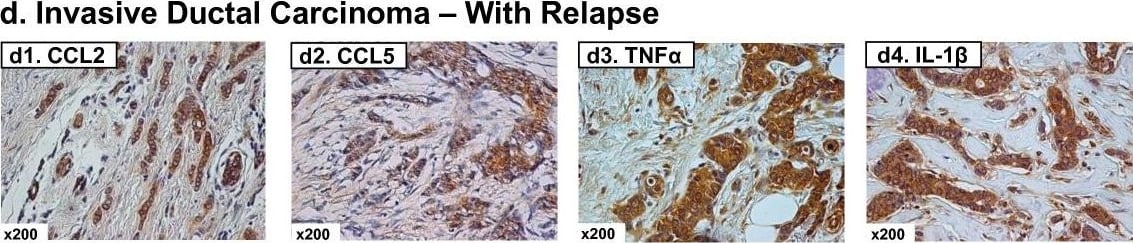
Expression patterns of CCL2, CCL5, TNF alpha and IL-1 beta in healthy individuals and breast cancer patients. Representative examples of the expression of CCL2, CCL5, TNF alpha and IL-1 beta in the different groups of patients included in the study, in biopsies obtained at the time of diagnosis. (a1-a4) Patients diagnosed with benign breast disorders. The pictures demonstrate the lack of staining of the four factors in the normal breast epithelial cells, as denoted in the majority of patients included in this group. (b1-b4) DCIS patients. The pictures demonstrate positive staining of the four factors in the malignant lesions, as denoted in the majority of patients included in this group. (c1-c4) IDC-no-relapse patients. The pictures demonstrate positive staining of the four factors in the tumor cells, as denoted in the majority of patients included in this group. (d1-d4) IDC-with-relapse patients. The pictures demonstrate positive staining of the four factors in the tumor cells, as denoted in the majority of patients included in this group. (a1, b1, c1, d1) CCL2 staining; (a2, b2, c2, d2) CCL5 staining; (a3, b3, c3, d3) TNF alpha staining; (a4, b4, c4, d4) IL-1 beta staining. The expression of the proteins was determined by IHC using specific antibodies, whose specificity in IHC was verified. The values of photo magnification are indicated in the left bottom corner of each of the pictures. Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/21486440), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human CCL2/JE/MCP-1 by Immunohistochemistry
Expression patterns of CCL2, CCL5, TNF alpha and IL-1 beta in healthy individuals and breast cancer patients. Representative examples of the expression of CCL2, CCL5, TNF alpha and IL-1 beta in the different groups of patients included in the study, in biopsies obtained at the time of diagnosis. (a1-a4) Patients diagnosed with benign breast disorders. The pictures demonstrate the lack of staining of the four factors in the normal breast epithelial cells, as denoted in the majority of patients included in this group. (b1-b4) DCIS patients. The pictures demonstrate positive staining of the four factors in the malignant lesions, as denoted in the majority of patients included in this group. (c1-c4) IDC-no-relapse patients. The pictures demonstrate positive staining of the four factors in the tumor cells, as denoted in the majority of patients included in this group. (d1-d4) IDC-with-relapse patients. The pictures demonstrate positive staining of the four factors in the tumor cells, as denoted in the majority of patients included in this group. (a1, b1, c1, d1) CCL2 staining; (a2, b2, c2, d2) CCL5 staining; (a3, b3, c3, d3) TNF alpha staining; (a4, b4, c4, d4) IL-1 beta staining. The expression of the proteins was determined by IHC using specific antibodies, whose specificity in IHC was verified. The values of photo magnification are indicated in the left bottom corner of each of the pictures. Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/21486440), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human CCL2/JE/MCP-1 by Immunohistochemistry
Expression patterns of CCL2, CCL5, TNF alpha and IL-1 beta in healthy individuals and breast cancer patients. Representative examples of the expression of CCL2, CCL5, TNF alpha and IL-1 beta in the different groups of patients included in the study, in biopsies obtained at the time of diagnosis. (a1-a4) Patients diagnosed with benign breast disorders. The pictures demonstrate the lack of staining of the four factors in the normal breast epithelial cells, as denoted in the majority of patients included in this group. (b1-b4) DCIS patients. The pictures demonstrate positive staining of the four factors in the malignant lesions, as denoted in the majority of patients included in this group. (c1-c4) IDC-no-relapse patients. The pictures demonstrate positive staining of the four factors in the tumor cells, as denoted in the majority of patients included in this group. (d1-d4) IDC-with-relapse patients. The pictures demonstrate positive staining of the four factors in the tumor cells, as denoted in the majority of patients included in this group. (a1, b1, c1, d1) CCL2 staining; (a2, b2, c2, d2) CCL5 staining; (a3, b3, c3, d3) TNF alpha staining; (a4, b4, c4, d4) IL-1 beta staining. The expression of the proteins was determined by IHC using specific antibodies, whose specificity in IHC was verified. The values of photo magnification are indicated in the left bottom corner of each of the pictures. Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/21486440), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human CCL2/JE/MCP-1 by Immunohistochemistry
Expression patterns of CCL2, CCL5, TNF alpha and IL-1 beta in healthy individuals and breast cancer patients. Representative examples of the expression of CCL2, CCL5, TNF alpha and IL-1 beta in the different groups of patients included in the study, in biopsies obtained at the time of diagnosis. (a1-a4) Patients diagnosed with benign breast disorders. The pictures demonstrate the lack of staining of the four factors in the normal breast epithelial cells, as denoted in the majority of patients included in this group. (b1-b4) DCIS patients. The pictures demonstrate positive staining of the four factors in the malignant lesions, as denoted in the majority of patients included in this group. (c1-c4) IDC-no-relapse patients. The pictures demonstrate positive staining of the four factors in the tumor cells, as denoted in the majority of patients included in this group. (d1-d4) IDC-with-relapse patients. The pictures demonstrate positive staining of the four factors in the tumor cells, as denoted in the majority of patients included in this group. (a1, b1, c1, d1) CCL2 staining; (a2, b2, c2, d2) CCL5 staining; (a3, b3, c3, d3) TNF alpha staining; (a4, b4, c4, d4) IL-1 beta staining. The expression of the proteins was determined by IHC using specific antibodies, whose specificity in IHC was verified. The values of photo magnification are indicated in the left bottom corner of each of the pictures. Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/21486440), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human CCL2/JE/MCP-1 by ELISA
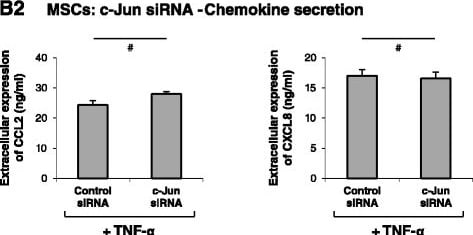
Induction of CCL2 and CLXL8 in TNF alpha -stimulated MSCs is not mediated via the AP-1 pathway. (A) Human BM-derived MSCs were stimulated by TNF-alpha (50 ng/ml) for 5 and 10 minutes. Control cells were treated by the vehicle of TNF-alpha. c-Jun levels and phosphorylation were determined by western blot (WB) analyses. Glyceraldehyde 3-phosphate dehydrogenase (GAPDH) was used as loading control. (B) Human BM-derived MSCs were transiently transfected by small interfering RNA (siRNA) to c-Jun or by control siRNA. (B1) c-Jun expression was determined by WB analyses. beta -Tubulin was used as loading control. (B2) Following siRNA transfection, the cells were stimulated by TNF-alpha (25 ng/ml; in this part of the study we used a suboptimal concentration of TNF-alpha in order to facilitate detection of inhibitory effects) for 24 hours. Expression levels of CCL2 and CXCL8 in the supernatants of the cells were determined by ELISA, in the linear range of absorbance. #siRNA to c-Jun has yielded minor increases or reductions in CCL2 and CXCL8 secretion in different experiments (see Results and discussion), and thus overall there was no significant effect on CCL2 and CXCL8 secretion. In all panels, the findings are representatives of n = 3 independent experiments that have shown similar results. Image collected and cropped by CiteAb from the following publication (https://stemcellres.biomedcentral.com/articles/10.1186/s13287-015-0080-7), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human CCL2/JE/MCP-1 by Western Blot
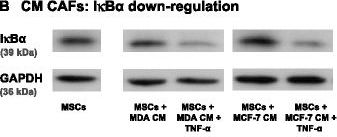
NF-kappa B is essential in mediating TNF-alpha -induced release of chemokines by MSCs. (A) Human BM-derived MSCs were stimulated by TNF-alpha (50 ng/ml) for 15 minutes. The levels of I kappa B alpha (the negative regulator of the NF-kappa B pathway) were determined by WB analyses. GAPDH was used as a loading control throughout. (B) CAFs were generated by culturing MSCs with Tumor CM from MDA-MB-231 (MDA) or MCF-7 breast tumor cells over a prolonged period of time (~30 days). TNF-alpha (50 ng/ml) was added for the last 24 hours to MSCs + Tumor CM cells and I kappa B alpha levels were determined by WB analyses. (C) CAF #1 cells were stimulated for 48 hours by TNF-alpha (50 ng/ml). I kappa B alpha levels were determined by WB analyses. (D) Human BM-derived MSCs were stimulated with TNF-alpha (50 ng/ml) for 10 minutes. p65 phosphorylation was determined by WB analyses. (E) Human BM-derived MSCs were transiently transfected by siRNA to p65 or by control siRNA. (E1) p65 expression was determined by WB analyses. (E2) Following siRNA transfection, the cells were stimulated by TNF-alpha (25 ng/ml; a suboptimal concentration of TNF-alpha in order to facilitate detection of inhibitory effects) for 48 hours. Expression of CCL2 and CXCL8 in the supernatants of the cells was determined by ELISA, in the linear range of absorbance. In all panels, the findings are representatives of n = 3 independent experiments that have shown similar results. Image collected and cropped by CiteAb from the following publication (https://stemcellres.biomedcentral.com/articles/10.1186/s13287-015-0080-7), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human CCL2/JE/MCP-1 by ELISA
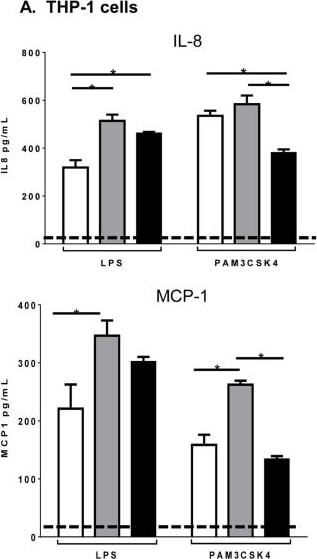
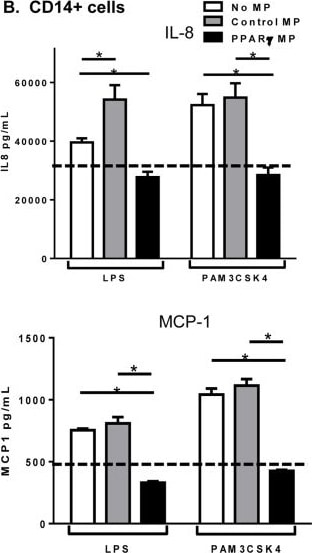
Microparticle composition influences inflammatory cytokine production from activated monocytes.THP-1 cells (A) or CD14 sorted blood monocytes (B) were treated with control microparticles (grey bars) or PPAR gamma -overexpressing microparticles (black bars) or no microparticles (white bars) for 4 hours before activation with LPS or PAM3CSK4 for 24 hours. Supernatants were collected and pro-inflammatory cytokines IL-8 (top), MCP-1 (bottom) were measured by ELISA. Dotted line represents baseline cytokine production from unactivated cells with no microparticle exposure. Mean values with standard errors represent one of at least 3 experiments. Two-way ANOVA with Tukey's multiple comparison post test was performed to determine statistical significance. * indicates (p<0.05). Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/25426628), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human CCL2/JE/MCP-1 by ELISA
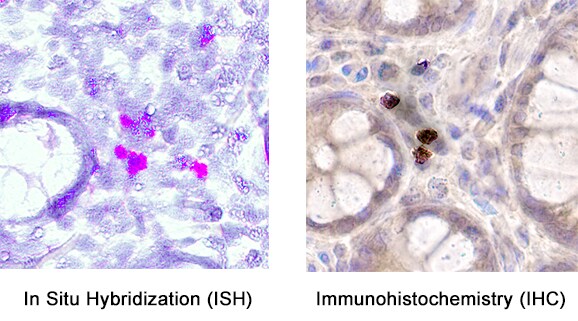
Microparticle composition influences inflammatory cytokine production from activated monocytes.THP-1 cells (A) or CD14 sorted blood monocytes (B) were treated with control microparticles (grey bars) or PPAR gamma -overexpressing microparticles (black bars) or no microparticles (white bars) for 4 hours before activation with LPS or PAM3CSK4 for 24 hours. Supernatants were collected and pro-inflammatory cytokines IL-8 (top), MCP-1 (bottom) were measured by ELISA. Dotted line represents baseline cytokine production from unactivated cells with no microparticle exposure. Mean values with standard errors represent one of at least 3 experiments. Two-way ANOVA with Tukey's multiple comparison post test was performed to determine statistical significance. * indicates (p<0.05). Image collected and cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/25426628), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of CCL2/JE/MCP‑1 in human Crohn's disease.
Formalin-fixed paraffin-embedded tissue sections of human Crohn’s Disease were probed for CCL2 mRNA (ACD RNAScope Probe, catalog # 423811; Fast Red chromogen, ACD catalog # 322360). Adjacent tissue section was processed for immunohistochemistry using mouse anti-human CCL2 monoclonal antibody (R&D Systems MAB679) at 20ug/mL with 1 hour incubation at room temperature followed by incubation with anti-mouse IgG VisUCyte HRP Polymer Antibody (VC001) and DAB chromogen (yellow-brown). Tissue was counterstained with hematoxylin (blue). Specific staining was localized to cytoplasm and membrane.Applications for Human CCL2/JE/MCP‑1 Antibody
Application
Recommended Usage
Dual RNAscope ISH-IHC Compatible
5-15 µg/mL
Sample: Immersion fixed paraffin-embedded sections of human Crohn's disease
Sample: Immersion fixed paraffin-embedded sections of human Crohn's disease
Immunohistochemistry
5-15 µg/mL
Sample: Immersion fixed paraffin-embedded sections of human Crohn's disease intestine
Sample: Immersion fixed paraffin-embedded sections of human Crohn's disease intestine
Western Blot
1 µg/mL
Sample: Recombinant Human CCL2/JE/MCP‑1 (Catalog # 279-MC)
Sample: Recombinant Human CCL2/JE/MCP‑1 (Catalog # 279-MC)
Neutralization
Measured by its ability to neutralize CCL2/JE/MCP‑1-induced chemotaxis in the BaF3 mouse pro‑B cell line transfected with human CCR2A. The Neutralization Dose (ND50) is typically 0.5-2.0 µg/mL in the presence of 75 ng/mL Recombinant Human CCL2/JE/MCP‑1.
Human CCL2/JE/MCP-1 Sandwich Immunoassay
Please Note: Optimal dilutions of this antibody should be experimentally determined.
Reviewed Applications
Read 3 reviews rated 5 using MAB679 in the following applications:
Formulation, Preparation, and Storage
Purification
Protein A or G purified from ascites
Reconstitution
Reconstitute at 0.5 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. See Certificate of Analysis for details.
*Small pack size (-SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
*Small pack size (-SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: CCL2/JE/MCP-1
Alternate Names
GDCF-2, HC11, HSMCR30, MCAF, MCP-1, SMC-CF
Gene Symbol
CCL2
Additional CCL2/JE/MCP-1 Products
Product Documents for Human CCL2/JE/MCP‑1 Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human CCL2/JE/MCP‑1 Antibody
For research use only
Citations for Human CCL2/JE/MCP‑1 Antibody
Customer Reviews for Human CCL2/JE/MCP‑1 Antibody (3)
5 out of 5
3 Customer Ratings
Have you used Human CCL2/JE/MCP‑1 Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
Showing
1
-
3 of
3 reviews
Showing All
Filter By:
-
Application: Western BlotSample Tested: Hepatocellular carcinoma cellsSpecies: HumanVerified Customer | Posted 08/17/2021
-
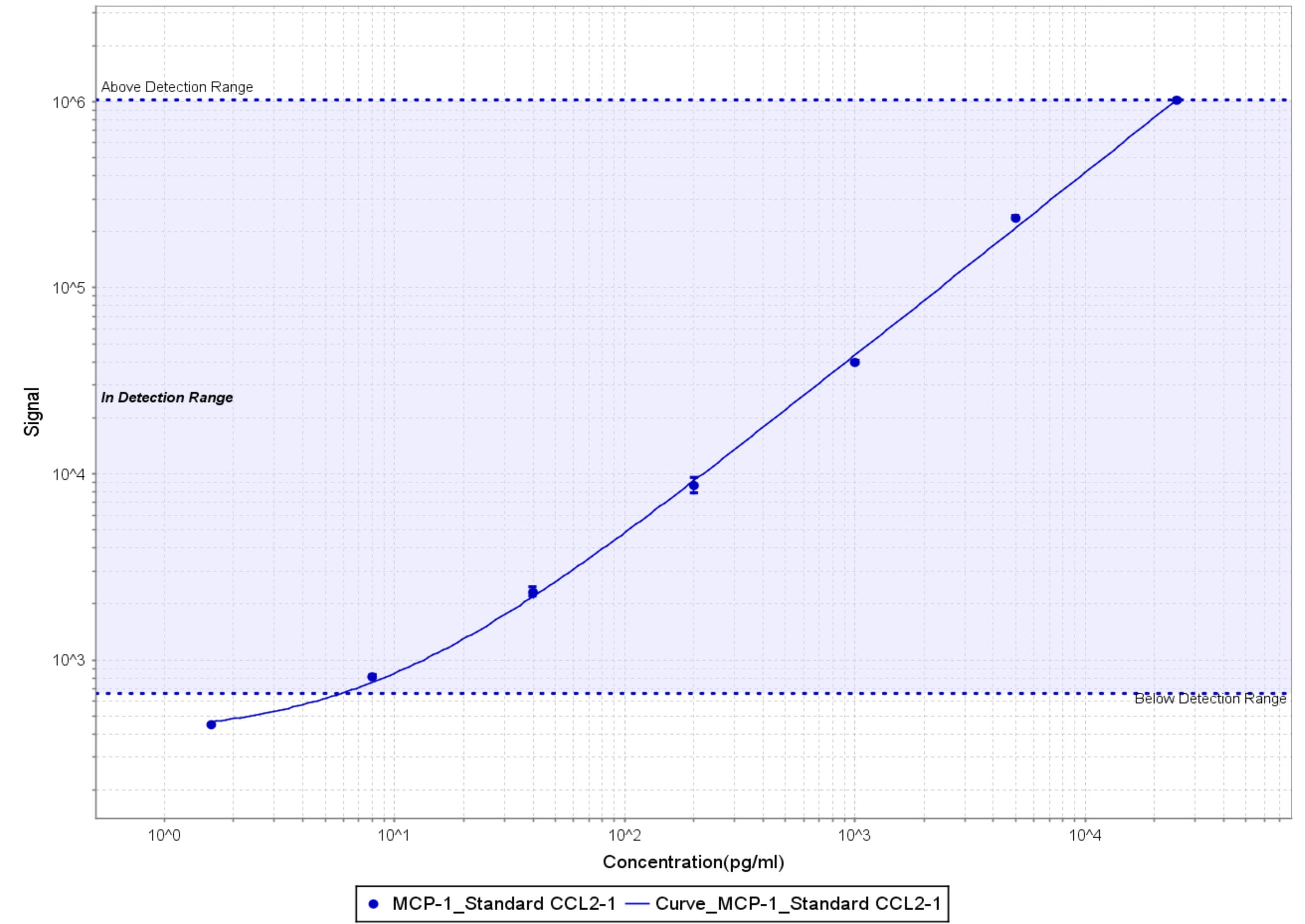
Application: MSD assaySample Tested: Vitreous humorSpecies: Cynomolgus MonkeyVerified Customer | Posted 11/20/2018After biotinylation, used as a capture reagent according to the manufacturer’s protocol (Meso Scale Diagnostics LLC). Paired with SulfoTag-modified AF-279-NA as a detection antibody. Calibration curves with Recombinant Human CCL2 (279-MC-010/CF) is shown (dynamic range 6-25,000 pg/ml)
-
Application: ELISASample Tested: CHO Chinese hamster ovary cell lineSpecies: HamsterVerified Customer | Posted 12/13/2017
There are no reviews that match your criteria.
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- IHC Sample Preparation (Frozen sections vs Paraffin)
- ISH-IHC Protocol for Chromogenic Detection on Formalin Fixed Paraffin Embedded (FFPE) Tissue
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Immunohistochemistry
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars