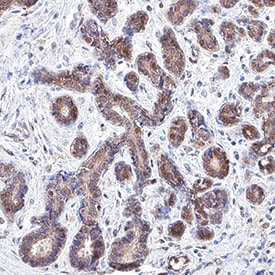
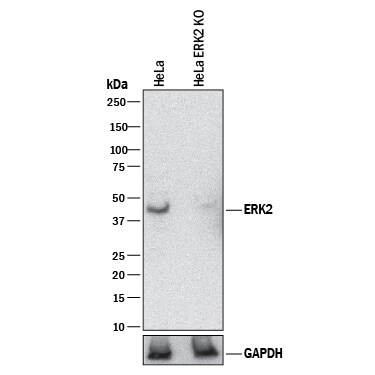
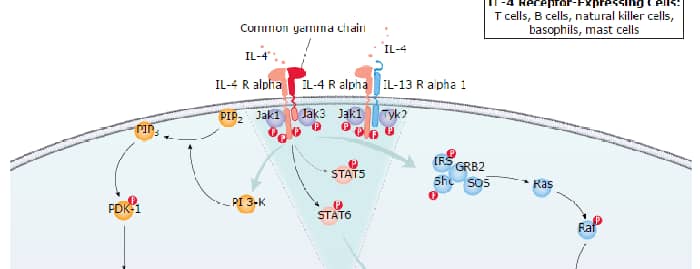
ERK1 and ERK2 (also known as MAPK3 and MAPK1) are 44 and 42 kDa Ser/Thr kinases, respectively. They are part of the Ras-Raf-ERK signal transduction cascade often found downstream of growth factor receptor activation. ERK1 and ERK2 were initially isolated and cloned as kinases activated in response to insulin and NGF. They are expressed in most, if not all, mammalian tissues. Dual threonine and tyrosine phosphorylation activate both ERKs, at Thr202/Tyr204 for human ERK1 and Thr185/Tyr187 for human ERK2.
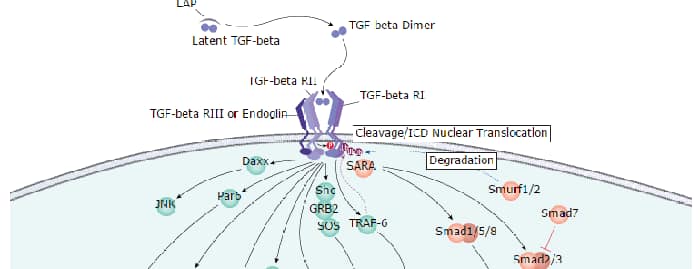
ERK5, also known as Big Mitogen-activated Protein Kinase 1 (BMK1) and MAPK7, is activated by several mechanisms, including receptor tyrosine kinases, G protein-coupled receptors, and osmotic stress. Like ERK1 and ERK2, ERK5 contains the conserved Thr-Glu-Tyr activation motif in its activation loop. Unlike these ERKs, however, ERK5 contains a unique C-terminal domain that regulates its activation and nuclear translocation.