IL-15 Signaling Pathways
Click on the “Effects” button shown in the Explore Pathways box below to reveal the primary biological effects of IL-15 signaling in different immune cell types. Click on one of the other cytokines shown in the Explore Pathways box below for information on a different common cytokine receptor gamma-chain family member.
gamma
chain
gamma
chain
IL-15 Receptor-Expressing Cells:
T cells, natural killer cells,
natural killer T cells,
dendritic cells, monocytes
IL-15 Receptor-Expressing Cells:
T cells, natural killer cells,
natural killer T cells,
dendritic cells, monocytes
Use our Product Suggestion form to enter a request.
You will be notified once it becomes available.
Use our Product Suggestion form to enter a request.
You will be notified once it becomes available.
Anti-Apoptotic
Anti-Apoptotic
Mitogenic
Mitogenic
Featured Literature
Common gamma-Chain Family Cytokines Play a Pivotal Role in Regulating Immune System Functions Poster
View PosterOverview of IL-15 Signaling Pathways
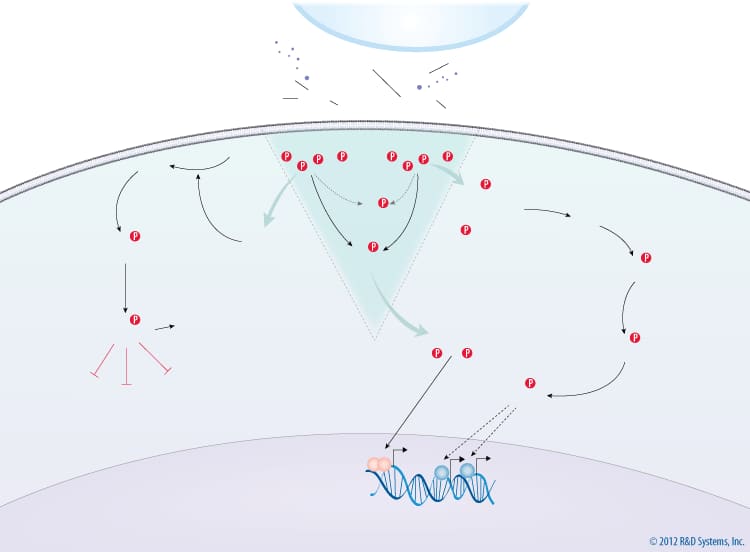
Interleukin-15 (IL-15) is a four alpha-helix bundle cytokine that is structurally and functionally related to IL-2. It is produced primarily by dendritic cells, monocytes, and epithelial cells. The heterotrimeric IL-15 receptor complex consists of a unique IL-15 R alpha subunit, IL-2/IL-15 R beta, and the common gamma-chain/IL-2 R gamma subunit. Unlike IL-2, IL-15 binds with high affinity to IL-15 R alpha, which then associates with a complex composed of the IL-2/IL-15 R beta and common gamma-chain/IL-2 R gamma subunits, expressed either on the same cell (cis-presentation) or on a different cell (trans-presentation). IL-15 signaling is essential for normal immune system functions. It stimulates T cell proliferation and inhibits IL-2-mediated activation-induced cell death. In addition, IL-15 is required for the development, survival, and activation of natural killer (NK) cells, homeostasis of natural killer T (NKT) cells and intraepithelial lymphocytes, and maintenance of naïve and memory CD8+ T cells. Both IL-15- and IL-15 R alpha-deficient mice lack NK cells and have severely reduced numbers of NKT cells, memory CD8+ T cells, and specific subsets of intestinal intraepithelial lymphocytes.
To learn more, please visit our Common gamma Chain Receptor Family Research Area.
Get Print Copy of this Pathway


