Immune Checkpoint
Immune Checkpoint Products | Co-inhibitory Targets | Co-stimulatory Targets | Emerging Targets | Read More About Immune Checkpoint Pathways | Resources | Related Research Areas
The immune checkpoint refers to a range of immunoregulatory pathways that enhance or suppress immune responses. Crucial factors in some of these pathways are co-stimulatory and co-inhibitory molecules, which act as key modulators of T cell activity.
- Co-stimulatory molecules: deliver activating signals that boost T cell proliferation, survival, and function
- Co-inhibitory molecules: act as checkpoints that suppress T cell activation to maintain immune balance and prevent autoimmunity
The delicate balance between these activating and inhibitory signals ensures that immune responses are appropriately regulated. Dysregulation of this balance is frequently exploited by cancer cells to evade immune surveillance. As a result, both co-stimulatory and co-inhibitory pathways have emerged as promising therapeutic targets in cancer immunotherapy, offering opportunities to either amplify anti-tumor immunity or relieve immunosuppression within the tumor microenvironment. R&D Systems offers a wide range of reagents and kits designed to identify and study immune checkpoint targets, including blocking antibodies, proteins, immunoassays, and more.
Immune Checkpoint Blocking Antibodies
Discover our extensive range of blocking antibodies directed against key immune checkpoint targets. Leveraging our in-house manufactured proteins, target-specific bioassays are employed to thoroughly validate the efficacy and specificity of each antibody.
Antibodies for Immune Cell Profiling
Choose from thousands of primary antibodies conjugated to 30+ fluorochromes for immune cell profiling in the TME. Our selection makes it easy to find the exact antibody-fluorochrome combinations you need and provides flexibility for multicolor panel design.
Avi-tag and Amine-Labeled Biotinylated Proteins
Biotinylated proteins can be powerful tools for assessing protein-protein interactions and screening antibody or small molecule libraries for potential therapeutics. With R&D Systems proteins, you'll get the performance and consistency needed to successfully advance your drug discovery research.
Key Product Categories
Co-inhibitory Immune Checkpoint Targets
Co-inhibitory immune checkpoint proteins play a critical role in maintaining immune homeostasis by regulating the amplitude and duration of immune responses to prevent autoimmunity. However, many tumors exploit these checkpoint pathways to evade immune surveillance by suppressing T cell activation and function within the tumor microenvironment. While targeting T cell co-inhibitory receptors such as CTLA-4 and PD-1 has led to significant advances in cancer immunotherapy, there are still some key challenges including:
- Limited efficacy of current monoclonal antibodies targeting CTLA-4, PD-1, or its ligand PD-L1
- Development of resistance or relapse in some patients that initially respond to treatment
- Activation of alternative immune checkpoint pathways that undermine treatment
As a result, there is an ongoing need to identify additional immune checkpoint targets and develop new therapies to improve treatment outcomes, either alone or in combination with existing immune checkpoint inhibitors.
Binding of HVEM to BTLA or CD160 Inhibits T Cell Activation
B and T lymphocyte attenuator (BTLA) is an inhibitory receptor within the CD28 family that shares structural features with CTLA-4 and PD-1.1 The extracellular portion of BTLA consists of a single Ig-like domain, followed by a transmembrane segment and an intracellular tail containing an immunoreceptor tyrosine-based inhibitory motif (ITIM) and an immunoreceptor tyrosine-based switch motif (ITSM), which are critical for transmitting inhibitory signals.2-4 BTLA is expressed on a variety of immune cell types, including T cells, natural killer (NK) cells, B cells, dendritic cells (DCs), and macrophages.5 While it is expressed at relatively low levels on naïve CD4+ and CD8+ T cells, BTLA is rapidly upregulated after T cell activation.6,7
Unlike other CD28 family receptors, BTLA does not bind to a B7 family ligand, but rather binds to herpesvirus entry mediator (HVEM), a member of the TNF receptor superfamily.8 HVEM is widely present on numerous immune populations, such as dendritic cells, naïve T and B cells, NK cells, monocytes, and neutrophils.6 The interaction between BTLA and HVEM inhibits T and B cell activation, proliferation, and cytokine production, and functions as a regulatory checkpoint controlling dendritic cell accumulation in lymphoid tissues.8-12 While naïve effector T cells down-regulate HVEM expression following activation, regulatory T cells up-regulate HVEM expression following stimulation, and its interaction with BTLA on effector T cells enhances their suppressive capacity.7
Beyond BTLA, HVEM also interacts with the CD160 receptor and the TNF superfamily ligands, lymphotoxin alpha and LIGHT.13-15 While HVEM acts as a ligand by binding to BTLA or CD160 to convey inhibitory signals, it also functions as a receptor by interacting with LIGHT or lymphotoxin-alpha to promote immune cell activation.6,13-15 Thus, HVEM acts as a versatile molecular switch that can mediate both stimulatory and inhibitory pathways.
In cancer settings, BTLA is present on CD8+ T cells isolated from melanoma patients’ peripheral blood, and its engagement with HVEM on melanoma cells suppresses T cell proliferation and IFN-gamma production.16 Elevated BTLA expression on tumor-specific CD8+ T cells also contributes to their functional impairment, while blockade of BTLA partially rescues their proliferative and cytokine-producing abilities.17 Beyond the effects of blocking either BTLA or PD-1 alone, simultaneous inhibition of these receptors significantly improves the expansion and function of tumor antigen-specific CD8+ T cells.17 Addition of TIM-3 blockade further enhances T cell responses, indicating that BTLA, PD-1, and TIM-3 collectively contribute to the dysfunctional state of CD8+ T cells in melanoma.17 These insights have positioned BTLA as a potential next-generation immunotherapy target.
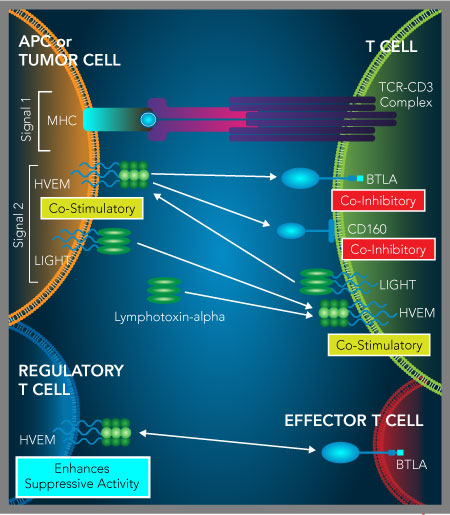
HVEM serves as a molecular switch regulating both T cell co-stimulatory and co-inhibitory signaling. HVEM is a TNF receptor superfamily protein that interacts with both BTLA and CD160 as a ligand, and LIGHT and Lymphotoxin-alpha as a receptor. While binding of HVEM to BTLA or CD160 produces a T cell co-inhibitory signal, HVEM binding to LIGHT or Lymphotoxin-alpha delivers a T cell co-stimulatory signal. The HVEM-BTLA interaction can also inhibit T cell functions by HVEM on regulatory T cells binding to T cell-expressed BTLA, which increases the suppressive activity of regulatory T cells.
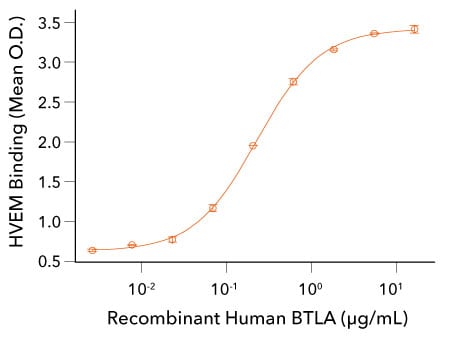
Recombinant Human BTLA Binds to HVEM. Recombinant Mouse HVEM/TNFRSF14 Fc Chimera (R&D Systems, Catalog # 2516-HV) was coated on a plate at 0.5 µg/mL and the indicated concentrations of Recombinant Human BTLA His tag (R&D Systems, Catalog # 9235-BT) were added. Recombinant Human BTLA bound with an ED50 of 0.1-0.5 µg/mL.
References
- Watanabe, N. et al. (2003) BTLA is a lymphocyte inhibitory receptor with similarities to CTLA-4 and PD-1. Nat. Immunol. 4:670. PMID: 12796776.
- Compaan, D.M. et al. (2005) Attenuating lymphocyte activity: the crystal structure of the BTLA-HVEM complex. J. Biol. Chem. 280:39553. PMID: 16169851.
- Gavrieli, M. et al. (2003) Characterization of phosphotyrosine binding motifs in the cytoplasmic domain of B and T lymphocyte attenuator required for association with protein tyrosine phosphatases SHP-1 and SHP-2. Biochem. Biophys. Res. Commun. 312:1236. PMID: 14652006.
- Chemnitz, J.M. et al. (2006) B and T lymphocyte attenuator-mediated signal transduction provides a potent inhibitory signal to primary human CD4 T cells that can be initiated by multiple phosphotyrosine motifs. J. Immunol. 176:6603. PMID: 16709818.
- Hurchla, M.A. et al. (2005) B and T lymphocyte attenuator exhibits structural and expression polymorphisms and is highly induced in anergic CD4+ T cells. J. Immunol. 174:3377. PMID: 15749870.
- Murphy, K.M. et al. (2006) Balancing co-stimulation and inhibition with BTLA and HVEM. Nat. Rev. Immunol. 6:671. PMID: 16932752.
- Tao, R. et al. (2008) Regulatory T cell expression of herpesvirus entry mediator suppresses the function of B and T lymphocyte attenuator-positive effector T cells. J. Immunol. 180:6649. PMID: 18453584.
- Sedy, J.R. et al. (2005) B and T lymphocyte attenuator regulates T cell activation through interaction with herpesvirus entry mediator. Nat. Immunol. 6:90. PMID: 15568026.
- Gonzalez, L.C. et al. (2005) A coreceptor interaction between the CD28 and TNF receptor family members B and T lymphocyte attenuator and herpesvirus entry mediator. Proc. Natl. Acad. Sci. USA 102:1116. PMID: 15647361.
- Vendel, A.C. et al. (2009) B and T lymphocyte attenuator regulates B cell receptor signaling by targeting Syk and BLNK. J. Immunol. 182:1509. PMID: 19155498.
- Yu, X. et al. (2019) BTLA/HVEM signaling: milestones in research and role in chronic hepatitis B virus infection. Front. Immunol. 10:617. PMID: 30984188.
- de Trez, C. et al. (2008) The inhibitory HVEM-BTLA pathway counter regulates lymphotoxin receptor signaling to achieve homeostasis of dendritic cells. J. Immunol. 180:238. PMID: 18097025.
- Cai, G. et al. (2008) CD160 inhibits activation of human CD4+ T cells through interaction with herpesvirus entry mediator. Nat. Immunol. 9:176. PMID: 18193050.
- Harrop, J.A. et al. (1998) Herpesvirus entry mediator ligand (HVEM-L), a novel ligand for HVEM/TR2, stimulates proliferation of T cells and inhibits HT29 cell growth. J. Biol. Chem. 273:27548. PMID: 9765287.
- Mauri, D.N. et al. (1998) LIGHT, a new member of the TNF superfamily, and lymphotoxin alpha are ligands for herpesvirus entry mediator. Immunity 8:21. PMID: 9462508.
- Derre, L. et al. (2010) BTLA mediates inhibition of human tumor-specific CD8+ T cells that can be partially reversed by vaccination. J. Clin. Invest. 120:157. PMID: 20038811.
- Fourcade, J. et al. (2012) CD8(+) T cells specific for tumor antigens can be rendered dysfunctional by the tumor microenvironment through upregulation of the inhibitory receptors BTLA and PD-1. Cancer Res. 72:887. PMID: 22205715.
Binding of B7-1 or B7-2 to CTLA-4 Down-Regulates T Cell Activity
CTLA-4 and CD28 are receptors expressed on the surface of T cells that specifically bind to the B7 family proteins, B7-1 (CD80) and B7-2 (CD86), which are primarily expressed by antigen-presenting cells (APCs). CD28 is constitutively expressed on naïve T cells and delivers a co-stimulatory signal upon binding to these ligands. In contrast, CTLA-4 expression is induced after T cell activation and transmits an inhibitory signal upon ligand engagement. CTLA-4 binds to both B7-1/CD80 and B7-2/CD86 with significantly higher affinity than CD28, resulting in suppression of the T cell response when CTLA-4 is upregulated.1
Moreover, CTLA-4 is constitutively expressed on regulatory T cells (Tregs) and can down-modulate the levels of B7-1/CD80 and B7-2/CD86 on APCs through trans-endocytosis, providing an additional means to suppress T cell activity.2,3 Activation of CTLA-4 also triggers reverse signaling in dendritic cells, inducing the production of indoleamine 2,3-dioxygenase (IDO), an enzyme that breaks down tryptophan and leads to the generation of immunosuppressive metabolites such as kynurenine.4,5 The depletion of tryptophan and accumulation of kynurenine can inhibit T cell activation and proliferation, suggesting CTLA-4 may indirectly control T cell responses by affecting dendritic cell functions.
Chronic and sustained expression of CTLA-4, along with other inhibitory receptors, is associated with T cell exhaustion in cancer.6-8 This exhausted phenotype is characterized by impaired T cell proliferation, reduced cytokine production, and diminished cytotoxic function, all contributing to weakened anti-tumor immune responses. Consequently, blocking CTLA-4 can enhance anti-cancer immunity by preventing CTLA-4-mediated inhibitory signaling, limiting regulatory T cell suppression, and partially restoring the functionality of exhausted T cells. As a result, CTLA-4 is one of the primary targets for cancer immunotherapy.8
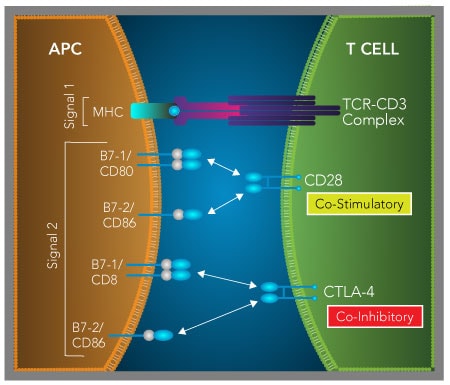
B7-1/CD80 and B7-2/CD86 mediate T cell co-stimulatory and co-inhibitory effects by binding to either CD28 or CTLA-4. Following recognition of the antigenic peptide/major histocompatibility complex (MHC) by the T cell receptor (TCR), binding of B7-1/CD80 or B7-2/CD86 to CD28 provides a co-stimulatory signal to promote T cell activation. Following T cell activation, the co-inhibitory receptor, CTLA-4, is up-regulated and binds with higher affinity to B7-1/CD80 and B7-2/CD86 than CD28, resulting in a down-regulation of T cell activity. In cancer, tumor-associated macrophages express B7-1/CD80 and B7-2/CD86, which allows them to suppress the functions of activated T cells through CTLA-4. Up-regulated expression of CTLA-4 and other inhibitory receptors is also associated with tumor-specific T cell exhaustion. This dysfunctional T cell state is characterized by a decrease in proliferative capacity and reduced T cell effector functions that prevent effective tumor elimination.
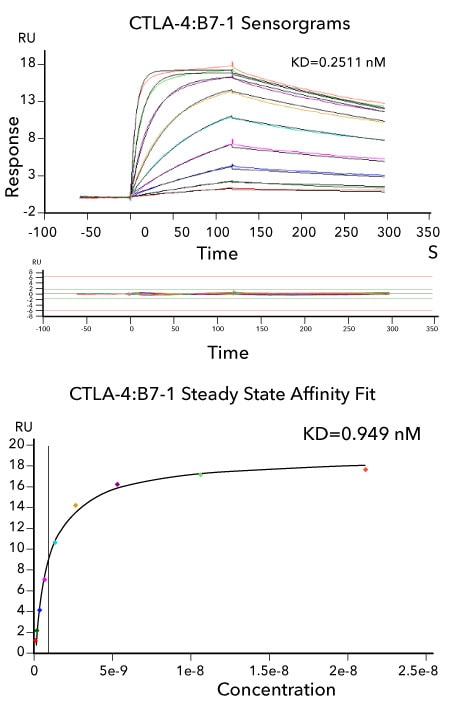
Affinity Measurements and Binding Kinetics of the CTLA-4:B7-1 Interaction by Surface Plasmon Resonance. Sensorgram data of captured Avi-tag Biotinylated Recombinant Human CTLA-4 Fc Chimera (R&D Systems, Catalog # AVI7268) binding to Recombinant Human B7-1 Fc Chimera (R&D Systems, Catalog # 10133-B1). Briefly, Avi-tag Biotinylated Recombinant Human CTLA-4 Fc Chimera was captured at a low coupling density to the active flow cell via the Avi-tag biotin. Recombinant Human B7-1 Fc Chimera at a concentration range between 82 pM and 21 nM was flowed over both active and uncoupled reference flow cells at each concentration. Kinetic sensorgrams were fit to a 1:1 binding model to determine the binding kinetics and affinity, with an interaction affinity of KD=0.2511 nM. The corresponding overlaid kinetic fits with the residual plot shown below. The corresponding steady state affinity fit is shown at the bottom. The experiment was performed on a Biacore T200, GE Healthcare.
References
- Sansom, D.M. (2000) CD28, CTLA-4 and their ligands: who does what and to whom? Immunology 101:169. PMID: 11012769.
- Qureshi, O.S. et al. (2011) Trans-endocytosis of CD80 and CD86: a molecular basis for the cell-extrinsic function of CTLA-4. Science 332:600. PMID: 21474713.
- Ovcinnikovs, V. et al. (2019) CTLA-4-mediated transendocytosis of costimulatory molecules primarily targets migratory dendritic cells. Sci. Immunol. PMID: 31152091.
- Grohmann, U. et al. (2002) CTLA-4-Ig regulates tryptophan catabolism in vivo. Nat. Immunol. 3:1097. PMID: 12368911.
- Munn, D.H. et al. (2004) Ligation of B7-1/B7-2 by human CD4+ T cells triggers indoleamine 2,3-dioxygenase activity in dendritic cells. J. Immunol. 172:4100. PMID: 15034022.
- Wherry, E.J. & M. Kurachi (2015) Molecular and cellular insights into T cell exhaustion. Nat. Rev. Immunol. 15:486. PMID: 26205583.
- Zhao, Y. et al. (2020) Exhaustion and senescence: two crucial dysfunctional states of T cells in the tumor microenvironment. Cell. Mol. Immunol. 17:27. PMID: 31853000.
- Seidel, J.A. et al. (2018) Anti-PD-1 and anti-CTLA-4 therapies in cancer: mechanisms of action, efficacy, and limitations. Front. Oncol. 8:86. PMID: 296444214.
LAG-3 Interacts with Multiple Ligands and Inhibits T cell Responses
Lymphocyte Activation Gene 3 (LAG-3), or CD223, is a type I transmembrane protein belonging to the immunoglobulin superfamily (IgSF). It is expressed on the surface of activated CD4+ and CD8+ T cells, natural killer (NK) cells, regulatory T cells, Tr1 cells, and plasmacytoid dendritic cells (pDCs) as either a dimer or a larger oligomeric structure.1-3 Structurally, LAG-3 is similar to CD4, featuring four extracellular Ig-like domains, but it also contains an additional loop on its membrane-distal Ig-like domain, which is absent in CD4. This unique loop gives LAG-3 the ability to bind to MHC class II molecules on antigen-presenting cells (APCs) or tumor cells with much higher affinity than CD4, thereby dampening T cell receptor (TCR) signaling.4,5
In addition to MHC class II, LAG-3 also interacts with Galectin-3, LSECtin, and Fibrinogen-like protein 1 (FGL1).2,6-8 These molecules are often expressed on various tumor cell types and contribute to T cell inhibition, although it remains unclear which ligand is chiefly responsible for the immunosuppressive effects associated with LAG-3.6,8 While the molecular details of LAG-3 signaling have not been fully determined, studies demonstrate that it suppresses T cell activation, proliferation, and cytokine secretion, restricts plasmacytoid dendritic cell activation, and boosts the suppressive capacity of regulatory T cells.9-15 Additionally, LAG-3 has been reported to limit cytokine production from mature natural killer cells.16 On regulatory T cells, LAG-3 engages with MHC class II molecules on dendritic cells (DCs), reducing their maturation and ability to stimulate immune responses.17 Conversely, a soluble form of LAG-3 can bind MHC class II on immature DCs, promoting their maturation and enhancing the cross-presentation of antigens to CD8+ T cells.18,19
Similar to PD-1, LAG-3 expression is commonly up-regulated on exhausted T cells in the tumor microenvironment.2,3 Although LAG-3 blockade alone only modestly reduces tumor growth in some mouse models, combining anti-LAG-3 with anti-PD-1 treatment yields a synergistic boost in antitumor immunity and prolongs survival beyond that seen with anti-PD-1 alone.20,21 This combined approach increases the number and infiltration of CD8+ and sometimes CD4+ T cells into tumors, along with heightened IFN-gamma production. These encouraging results have led to clinical trials testing antagonistic anti-LAG-3 antibodies both as a monotherapy and in combination with anti-PD-1.22,23 Furthermore, due to the immune-activating properties of soluble LAG-3, a recombinant soluble variant known as IMP321 is currently undergoing early-phase clinical evaluation in combination with anti-PD-1 therapy.
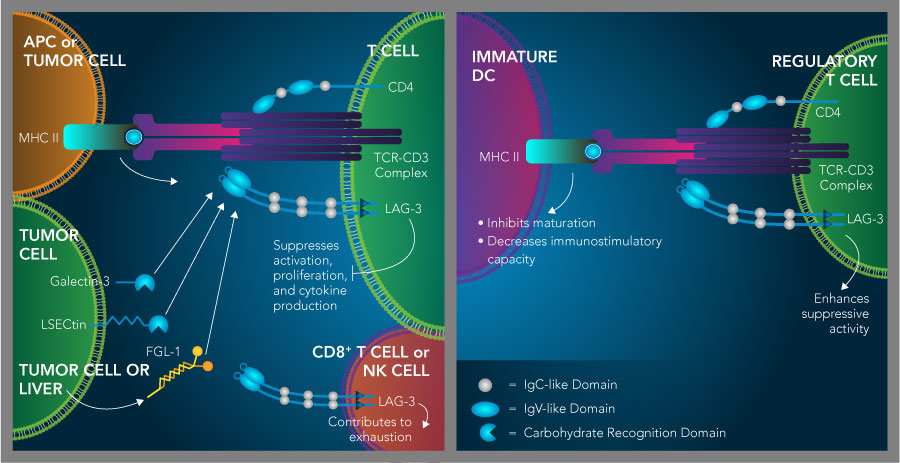
LAG-3 is an immunosuppressive receptor that inhibits T cell activity and promotes the suppressive activity of regulatory T cells. LAG-3 binds to multiple ligands including MHC class II, Galectin-3, LSECtin, and Fibrinogen-like protein 1 (FGL1), which have all been reported to have T cell inhibitory effects. Similar to PD-1, TIM-3, TIGIT, and BTLA, LAG-3 is also up-regulated on exhausted T cells and natural killer cells in cancer and is thought to contribute to their dysfunction (left). On regulatory T cells, LAG-3 expression enhances their suppressive function and can inhibit dendritic cell maturation and immunostimulatory capacity through its interaction with MHC class II (right).

LAG-3 Induces TNF-alpha Secretion by Mouse Immature Dendritic Cells. JAWSII mouse immature dendritic cells were treated with the indicated concentrations of Recombinant Cynomolgus Monkey LAG-3 (R&D Systems, Catalog # 9992-L3). TNF-alpha secretion was measured in cell culture supernatants using the Mouse TNF-alpha Quantikine™ ELISA Kit (R&D Systems, Catalog # MTA00B). The ED50for this effect is 0.15-0.9 ug/mL in the presence of a cross-linking antibody, Mouse Anti-His Tag Monoclonal Antibody (R&D Systems, Catalog # MAB050R).
References
- He, Y. et al. (2016) Lymphocyte-activation gene-3, an important immune checkpoint in cancer. Cancer Sci. 107:1193. PMID: 27297395.
- Anderson, A.C. et al. (2016) Lag-3, Tim-3, and TIGIT: Co-inhibitory receptors with specialized functions in immune regulation. Immunity 44:989. PMID: 27192565.
- Wherry, E.J. (2001) T cell exhaustion. Nat. Immunol. 12:492. PMID: 21739672.
- Huard, B. et al. (1994) Lymphocyte-activation gene 3/major histocompatibility complex class II interaction modulates the antigenic response of CD4+ T lymphocytes. Eur. J. Immunol. 24:3216. PMID: 7805750.
- Huard, B. et al. (1995) CD4/major histocompatibility complex class II interaction analyzed with CD4- and lymphocyte activation gene-3 (LAG-3)-Ig fusion proteins. Eur. J. Immunol. 25:2718. PMID: 7589152.
- Xu, F. et al. (2014) LSECtin expressed on melanoma cells promotes tumor progression by inhibiting antitumor T-cell responses. Cancer Res. 74:3418. PMID: 24769443.
- Kouo, T. et al. (2015) Galectin-3 shapes antitumor immune responses by suppressing CD8+ T cells via LAG-3 and inhibiting expansion of plasmacytoid dendritic cells. Cancer Immunol. Res. 3:412. PMID: 25691328.
- Wang, J. et al. (2019) Fibrinogen-like protein 1 is a major immune inhibitory ligand of LAG-3. Cell 176:334. PMID: 30580966.
- Workman, C.J. et al. (2009) LAG-3 regulates plasmacytoid dendritic cell homeostasis J. Immunol. 182:1885. PMID: 19201841.
- Huard, B. et al. (1996) T cell major histocompatibility complex class II molecules down-regulate CD4+ T cell clone responses following LAG-3 binding. Eur. J. Immunol. 26:1180. PMID: 8647185.
- Baixeras, E. et al. (1992) Characterization of the lymphocyte activation gene 3-encoded protein. A new ligand for human leukocyte antigen class II antigens. J. Exp, Med. 176:327. PMID: 1380059.
- Hannier, S. et al. (1998) CD3/TCR complex-associated lymphocyte activation gene-3 molecules inhibit CD3/TCR signaling. J. Immunol. 161:4058. PMID: 9780176.
- Huang, C.T. et al. (2004) Role of LAG-3 in regulatory T cells. Immunity 21:503. PMID: 15485628.
- Workman, C.J. & D.A. Vignali (2003) The CD4-related molecule, LAG-3 (CD223), regulates the expansion of activated T cells. Eur. J. Immunol. 33:970. PMID: 12672063.
- Workman, C.J. et al. (2004) Lymphocyte activation gene-3 (CD223) regulates the size of the expanding T cell population following antigen activation in vivo. J. Immunol. 172:5450. PMID: 15100286.
- Narayanan, S. et al. (2020) LAG-3 is a central regulator of NK cell cytokine production.
- Liang, B. et al. (2008) Regulatory T cells inhibit dendritic cells by lymphocyte activation gene-3 engagement of MHC class II. J. Immunol. 180:5916. PMID: 18424711.
- Triebel, F. (2003) LAG-3: a regulator of T-cell and DC responses and its use in therapeutic vaccination. Trends Immunol. 24:619. PMID: 14644131.
- Casati, C. et al. (2006) Soluble human LAG-3 molecule amplifies the in vitro generation of type 1 tumor-specific immunity. Cancer Res. 66:4450. PMID: 16618772.
- Woo, S.R. et al. (2012) Immune inhibitory molecules LAG-3 and PD-1 synergistically regulate T-cell function to promote tumoral immune escape. Cancer Res. 72:917. PMID: 22186141.
- Andrews, L.P. et al. (2017) LAG3 (CD223) as a cancer immunotherapy target. Immunol. Rev. 276:80. PMID: 28258692.
- Dempke, W.C.M. et al. (2017) Second- and third-generation drugs for immuno-oncology treatment - the more the better? Eur. J. Cancer 74:55. PMID: 28335888.
- Marin-Acevedo, J.A. et al. (2018) Next generation of immune checkpoint therapy in cancer: new developments and challenges. J. Hematol. Oncol. 11:39. PMID: 29544515.
Binding of PD-L1 or PD-L2 to PD-1 Inhibits T Cell Activation
PD-1 is a co-inhibitory receptor from the CD28 family that becomes upregulated on T cells following T cell receptor (TCR) activation or in response to certain cytokines. It engages with two ligands, PD-L1 (also called B7-H1) and PD-L2 (B7-DC), both of which are expressed on antigen-presenting cells and whose expression is increased upon IFN-gamma stimulation.1 Interaction between PD-1 and its ligands disrupts early signaling through the TCR and CD28 pathways, inhibits IL-2 production and T cell proliferation, T cell effector functions, and T cell survival.2-4 Tumor cells often take advantage of this by overexpressing PD-L1 to inhibit anti-tumor T cell responses and promote immune evasion.2,4-6 Similar to CTLA-4, elevated PD-1 expression is associated with T cell exhaustion in cancer. Blocking PD-1 or PD-L1 with specific monoclonal antibodies has been shown to enhance anti-tumor immunity in a broad range of cancers.4,7,8
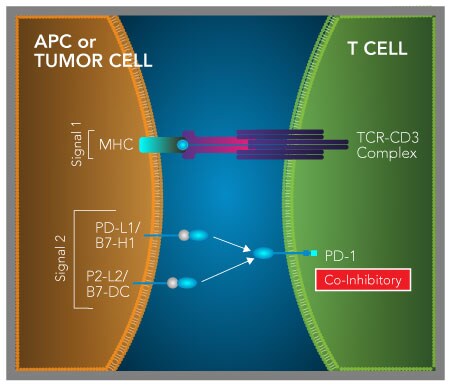
Binding of PD-L1 or PD-L2 to PD-1 delivers a T cell co-inhibitory signal. Following TCR activation, expression of the co-inhibitory receptor, PD-1, is induced on naïve T cells. Ligation of PD-1 by either of its ligands, PD-L1/B7-H1 or PD-L2/B7-DC, expressed on antigen-presenting cells (APCs) inhibits TCR-mediated T cell proliferation and cytokine production. In cancer, PD-L1 is frequently expressed by tumor cells and other cells present in the tumor microenvironment including tumor-associated macrophages (TAMs) and myeloid-derived suppressor cells (MDSCs). PD-L1 expressed on tumor cells, TAMs, or MDSCs binds to T cell-expressed PD-1 and suppresses T cell-mediated anti-tumor immune responses. Up- regulated expression of PD-1 is also found on exhausted T cells in cancer and is thought to contribute to T cell dysfunction.
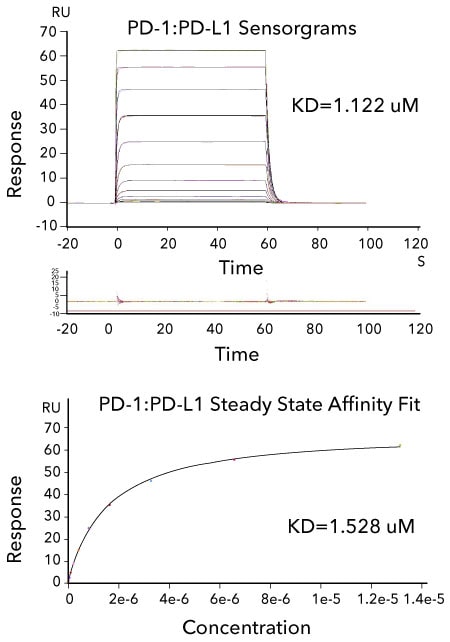
Affinity Measurements and Binding Kinetics of the PD-1:PD-L1 Interaction by Surface Plasmon Resonance. Sensorgram data of captured Avi-tag Biotinylated Recombinant Human PD-L1 His tag (R&D Systems, Catalog # AVI9049) binding to Recombinant Human PD-1 His tag (R&D Systems, Catalog # 8986-PD). The corresponding overlaid kinetic fits with the residual plot shown below. The concentration of Recombinant Human PD-1 His-tag ranged from 3.2 nM to 13.2 μM. The corresponding steady state affinity fit is shown below. The experiment was performed on a BiacoreT200, GE Healthcare.
References
- Brown, J.A. et al. (2003) Blockade of programmed death-1 ligands on dendritic cells enhances T cell activation and cytokine production. J. Immunol. 170:1257. PMID: 12538684.
- Wang, Y. et al. (2018) Regulation of PD-L1: Emerging routes for targeting tumor immune evasion. Front. Pharmacol. 9:536. PMID: 29910728.
- Schildberg, F.A. (2016) Coinhibitory pathways in the B7-CD28 ligand-receptor family. Immunity 44:955. PMID: 27192563.
- Seidel, J.A. et al. (2018) Anti-PD-1 and anti-CTLA-4 therapies in cancer: mechanisms of action, efficacy, and limitations. Front. Oncol. 8:86. PMID: 29644214.
- Topalian, S.L. et al. (2015) Immune checkpoint blockade: a common denominator approach to cancer therapy. Cancer Cell 27:450. PMID: 25858804.
- Zheng, Y. et al. (2019) PD-L1 expression levels on tumor cells affect their immunosuppressive activity. Oncol. Lett. 18:5399. PMID: 31612048.
- Wherry, E.J. & M. Kurachi (2015) Molecular and cellular insights into T cell exhaustion. Nat. Rev. Immunol. 15:486. PMID: 26205583.
- Zhao, Y. et al. (2020) Exhaustion and senescence: two crucial dysfunctional states of T cell in the tumor microenvironment. Cell. Mol. Immunol. 17:27. PMID: 31853000.
TIGIT Binds to CD112 or CD155 and Inhibits T Cell and Natural Killer Cell Activity
T cell immunoreceptor with immunoglobulin and ITIM domains (TIGIT), also known as VSTM3, VSIG9, and WUCAM, is an inhibitory receptor belonging to the immunoglobulin superfamily (IgSF). TIGIT has a gene structure that is similar to members of the CD28 family, featuring a single IgV-like domain in its extracellular region that mediates ligand binding, followed by a single-pass transmembrane segment, and a cytoplasmic tail.1 The cytoplasmic tail of TIGIT contains both an immunoreceptor tyrosine-based inhibitory motif (ITIM) and an immunoglobulin tail tyrosine-like (ITT) motif, which play key roles in transmitting inhibitory signals. TIGIT is expressed on activated and memory T cells, natural killer (NK) cells, follicular helper T (Tfh) cells, and regulatory T cells.1-5 Similar to PD-1 and TIM-3, TIGIT is also highly expressed on exhausted T cells and exhausted NK cells in chronic infections and cancer.6,7
TIGIT recognizes and binds three different Nectin family ligands: CD155 (also known as PVR), CD112 (Nectin-2), and CD113 (Nectin-3), with the strongest affinity for CD155/PVR.3,4,8 These ligands are mainly expressed on dendritic cells (DCs), T cells, and various non-hematopoietic cells such as epithelial, endothelial, and tumor cells.1-4 Initially, TIGIT binding to CD155 was found to inhibit T cell proliferation and cytokine production indirectly through induction of tolerogenic DCs, but more recent studies have demonstrated that TIGIT can directly suppress TCR-driven T cell activation, proliferation, and cytokine release.1,4,5,9 On regulatory T cells, high levels of TIGIT are linked to lineage stability, stronger suppressive function, and increased IL-10 secretion.10-12 Importantly, TIGIT engagement on regulatory T cells promotes secretion of Fgl2, a factor that inhibits differentiation of Th1 and Th17 cells but does not affect the Th2 pathway, thereby shifting immune responses away from pro-inflammatory Th1/Th17 responses toward a Th2-dominant profile.9,13 On NK cells, TIGIT interaction with CD155 or CD112 suppresses their cytotoxic activity.3,14 Consequently, TIGIT plays a role in dampening immune responses through its effects on various immune populations.
In the context of cancer, CD155 and CD112 are frequently overexpressed across many tumor types, and TIGIT is prominently expressed on tumor-infiltrating lymphocytes (TILs) derived from both mouse models and human cancers.6,11,15-20 Expression of TIGIT on CD8+ TILs is typically accompanied by elevated PD-1 and TIM-3 expression and correlates with an exhausted, dysfunctional T cell state.11,20 Within the tumor microenvironment, TIGIT+ regulatory T cells exhibit enhanced suppressive ability and produce more IL-10.11 Studies using TIGIT knockout mice bearing tumors such as B16F10 melanoma or MC38 colon carcinoma demonstrate that the loss of TIGIT slows tumor growth and enhances CD8+ T cell activity.11 Similar results have been reported in other TIGIT deficient tumor models and with TIGIT blockade, although the delay in tumor growth has also been attributed to heightened NK cell function in some studies.7,21-23 While blocking either TIGIT or PD-L1 alone produces minimal impact on tumor progression and survival in a colorectal cancer model, combined inhibition of TIGIT and PD-L1 or TIGIT and PD-1 significantly enhances anti-tumor immune responses.6 Parallel findings emerged from studies using dual TIGIT/PD-1 blockade in mouse glioblastoma models.24 Moreover, TIGIT-deficient tumor-bearing mice treated with anti-TIM-3 antibodies exhibit reduced tumor growth and metastasis, along with improved survival compared to controls, implying that co-targeting TIGIT and TIM-3 can synergistically augment anti-tumor immunity.11 Based on this accumulating evidence, antagonistic monoclonal antibodies targeting TIGIT are currently being tested in clinical trials for patients with advanced and metastatic cancers.25
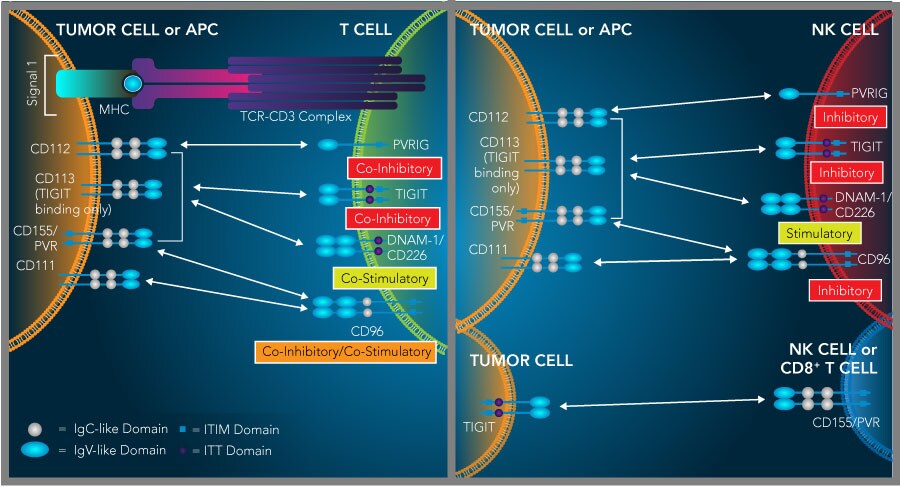
The TIGIT, PVRIG, CD96, and DNAM-1/CD226 proteins bind to CD155/PVR and/or CD112 and have opposing effects on lymphocyte functions. TIGIT, PVRIG, CD96, and DNAM-1/CD226 are immunoglobulin superfamily receptors that mediate opposing effects on lymphocyte functions following ligand binding. All of these receptors bind to one or more of the following nectin or nectin-like molecules, CD111/Nectin-1, CD112/Nectin-2, CD113/Nectin-3, and CD155/PVR, which are frequently overexpressed on a broad range of tumors. The TIGIT protein binds to CD155/PVR, CD112 and CD113 and inhibits both T cell and NK cell activity. Conversely, DNAM-1/CD226 binds to CD155/PVR and CD112 and promotes CD8+ T cell and NK cell activity. PVRIG and CD96/TACTILE are additional T cell and NK cell inhibitory receptors that compete with DNAM-1/CD226 for binding to CD112 or CD155/PVR, respectively. Although the interaction between CD155/PVR and CD96/TACTILE has been shown to inhibit CD8+ T cell and NK cell functions in mice, whether this interaction mediates co-inhibitory or co-stimulatory effects in humans is still being investigated. In addition to its expression on T cells and NK cells, TIGIT may also be expressed on tumor cells and interact with T cell- or NK cell-expressed CD155/PVR, leading to a down-regulation of T cell and NK cell activity.
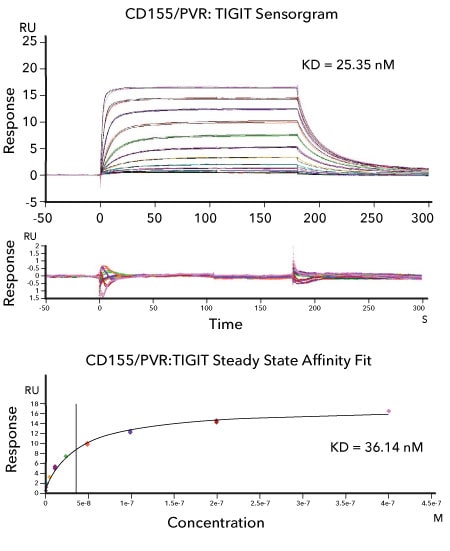
Affinity Measurements and Binding Kinetics of the CD155/PVR:TIGIT Protein Interaction by Surface Plasmon Resonance. Sensorgram data of captured Avi-tag Biotinylated Recombinant Human CD155/PVR Fc Chimera (R&D Systems, Catalog # AVI9174) binding to Recombinant Human TIGIT His-tag (R&D Systems, Catalog # 9525-TG). The corresponding overlaid kinetic fits with the residual plot shown below. The concentration of Recombinant Human TIGIT His-tag ranged from 0.2 nM to 400 nM. The corresponding steady state affinity fit is shown at the bottom. The experiment was performed on a Biacore T200, GE Healthcare.
References
- Levin, S.D. et al. (2011) Vstm3 is a member of the CD28 family and an important modulator of T-cell function. Eur. J. Immunol. 41:902. PMID: 21416464.
- Boles, K.S. et al. (2009) A novel molecular interaction for the adhesion of follicular CD4 T cells to follicular DC. Eur. J. Immunol. 39:695. PMID: 19197944.
- Stanietsky, N. et al. (2009) The interaction of TIGIT with PVR and PVRL2 inhibits human NK cell cytotoxicity. Proc. Natl. Acad. Sci. USA 106:17858. PMID: 19815499.
- Yu, X. et al. (2009) The surface protein TIGIT suppresses T cell activation by promoting the generation of mature immunoregulatory dendritic cells. Nat. Immunol. 10:48. PMID: 19011627.
- Joller, N. et al. (2011) Cutting edge: TIGIT has T cell-intrinsic inhibitory functions. J. Immunol. 186:1338. PMID: 21199897.
- Johnston, R.J. et al. (2014) The immunoreceptor TIGIT regulates antitumor and antiviral CD8(+) T cell effector function. Cancer Cell 26:923. PMID: 25465800.
- Zhang, Q. et al. (2018) Blockade of the checkpoint receptor TIGIT prevents NK cell exhaustion and elicits potent anti-tumor immunity. Nat. Immunol. 19:723. PMID: 29915296.
- Anderson, A.C. et al. (2016) Lag-3, Tim-3, and TIGIT: Co-inhibitory receptors with specialized functions in immune regulation. Immunity 44:989. PMID: 27192565.
- Lozano, E. et al. (2012) The TIGIT/CD226 axis regulates human T cell function. J. Immunol. 188:3869. PMID: 22427644.
- Fuhrman, C.A. et al. (2015) Divergent phenotypes of human regulatory T cells expressing the receptors TIGIT and CD226. J. Immunol. 195:145. PMID: 25994968.
- Kurtulus, S. et al. (2015) TIGIT predominantly regulates the immune response via regulatory T cells. J. Clin. Invest. 125:4053. PMID: 26413872.
- Burton, B.R. et al. (2014) Sequential transcriptional changes dictate safe and effective antigen-specific immunotherapy. Nat. Commun. 5:4741. PMID: 25182274.
- Joller, N. et al. (2014) Treg cells expressing the coinhibitory molecule TIGIT selectively inhibit proinflammatory Th1 and Th17 cell responses. Immunity 40:569. PMID: 24745333.
- Stanietsky, N. et al. (2013) Mouse TIGIT inhibits NK-cell cytotoxicity upon interaction with PVR. Eur. J. Immunol. 43:2138. PMID: 23677581.
- Masson, D. et al. (2001) Overexpression of the CD155 gene in human colorectal carcinoma. Gut 49:236. PMID: 11454801.
- Tahara-Hanaoka, S. et al. (2006) Tumor rejection by the poliovirus receptor family ligands of the DNAM-1 (CD226) receptor. Blood 107:1491. PMID: 16249389.
- Carlsten, M. et al. (2007) DNAX accessory molecule-1 mediated recognition of freshly isolated ovarian carcinoma by resting natural killer cells. Cancer Res. 67:1317. PMID: 17283169.
- Pende, D. et al. (2005) Analysis of the receptor-ligand interactions in the natural killer-mediated lysis of freshly isolated myeloid or lymphoblastic leukemias: evidence for the involvement of the Poliovirus receptor (CD155) and Nectin-2 (CD112). Blood 105:2066. PMID: 15536144.
- El-Sherbiny, Y.M. et al. (2007) The requirement for DNAM-1, NKG2D, and NKp46 in the natural killer cell-mediated killing of myeloma cells. Cancer Res. 67:8444. PMID: 17875681.
- Chauvin, J.M. et al. (2015) TIGIT and PD-1 impair tumor antigen-specific CD8+ T cells in melanoma patients. J. Clin. Invest. 125:2046. PMID: 25866972.
- He, W. et al. (2017) CD155/TIGIT signaling regulates CD8+ T-cell metabolism and promotes tumor progression in human gastric cancer. Cancer Res. 77:6375. PMID: 28883004.
- Guillerey, C. et al. (2018) TIGIT immune checkpoint blockade restores CD8+ T-cell immunity against multiple myeloma. Blood 132:1689. PMID: 29986909.
- Wu, L. et al. (2019) Blockade of TIGIT/CD155 signaling reverses T-cell exhaustion and enhances antitumor capability in head and neck squamous cell carcinoma. Cancer Immunol. Res. 7:1700. PMID: 31387897.
- Hung, A.L. et al. (2018) TIGIT and PD-1 dual checkpoint blockade enhances antitumor immunity and survival in GBM. Oncoimmunology 7:1466769. PMID: 30221069.
- Marin-Acevedo, J.A. et al. (2018) Next generation of immune checkpoint therapy in cancer: new developments and challenges. J. Hematol. Oncol. 11:39. PMID: 29544515.
TIM-3 is an Inhibitory Receptor that Binds Gal-9, CEACAM-1, and HMGB1
T cell immunoglobulin and mucin domain-containing protein 3 (TIM-3), also known as Hepatitis A virus cellular receptor 2 (HAVCR2), belongs to the TIM family, which also includes TIM-1 and TIM-4 in humans, and TIM-1 through TIM-8 in mice.1 Members of the TIM family are type I transmembrane glycoproteins with an extracellular region consisting of an N-terminal IgV-like domain, a mucin-like domain, and a stalk region, containing potential sites for O- and N-linked glycosylation, respectively. The extracellular domain is connected to a transmembrane segment and, except for TIM-4, a cytoplasmic tail harboring a conserved cluster of five tyrosine residues essential for downstream signaling pathways.2,3 TIM-3 is primarily expressed on IFN-gamma-producing CD8+ T cells and Th1 cells, with lower expression on Th17 cells, and it has been shown to negatively regulate T cell proliferation and cytokine secretion.1-6 TIM-3 is also found on certain populations of regulatory T cells, dendritic cells (DCs), macrophages, monocytes, and mast cells.7,8 Similar to PD-1, TIM-3 is markedly upregulated on exhausted T cells and natural killer cells in chronic infections and in tumors.9-12
The IgV domain of TIM-3 can engage several ligands, including galectin-9, phosphatidylserine (PtdSer), high mobility group box 1 protein (HMGB1), and CEACAM-1.13-18 Galectin-9, the first recognized TIM-3 ligand, binds to the carbohydrate motif within the IgV domain and triggers apoptosis in Th1 cells.13 Unlike galectin-9, which binds at a distinct site, the other ligands share overlapping binding regions located in the FG-CC’ loop of the IgV domain.3 PtdSer, which becomes exposed on apoptotic cells, binds not only to TIM-3 but also to TIM-1 and TIM-4, and facilitates uptake of apoptotic cells by macrophages and dendritic cells and antigen cross-presentation.19 HMGB1 is a damage-associated molecule released by stressed or dying cells that helps shuttle nucleic acids into endosomal compartments, which is critical for activating innate immune responses against tumors and infections. When TIM-3 binds to HMGB1, it hampers this process, thus suppressing innate immunity and anti-tumor defense.16 Lastly, TIM-3 interacts with CEACAM-1 in a cis manner that is essential for TIM-3 glycosylation, stability, and its ability to inhibit T cell activity.3,17 Additionally, these proteins can bind in trans to suppress T cell responses.3
Within multiple cancer types, TIM-3 is highly expressed on CD8+ tumor-infiltrating lymphocytes (TILs) and CD4+ regulatory T cells.3,7,11,20,21 All TIM-3+ CD8+ TILs also express PD-1, defining a population of particularly exhausted T cells found in both mouse tumor models and human cancers.22 Inhibition of TIM-3 alone has shown mixed results depending on the model, but combined blockade of TIM-3 and PD-1 synergistically reduces tumor growth.9,22-24 This combination therapy restores exhausted CD8+ TIL function, increasing tumor-specific T cell proliferation and cytokine production, while simultaneously dampening the suppressive effects of TIM-3+ regulatory T cells. Furthermore, dual inhibition of TIM-3 and PD-1 may restore the activity of exhausted natural killer cells, block galectin-9-driven expansion of immunosuppressive myeloid-derived suppressor cells (MDSCs), and prevent TIM-3 on intratumoral DCs from binding to HMGB1, thereby reactivating the innate immune response to tumor-derived nucleic acids.25,26 These findings have encouraged exploration of combination immunotherapies targeting TIM-3 alongside PD-1 or CTLA-4, and those incorporating TIM-3 blockade with agonists of T cell co-stimulatory receptors.
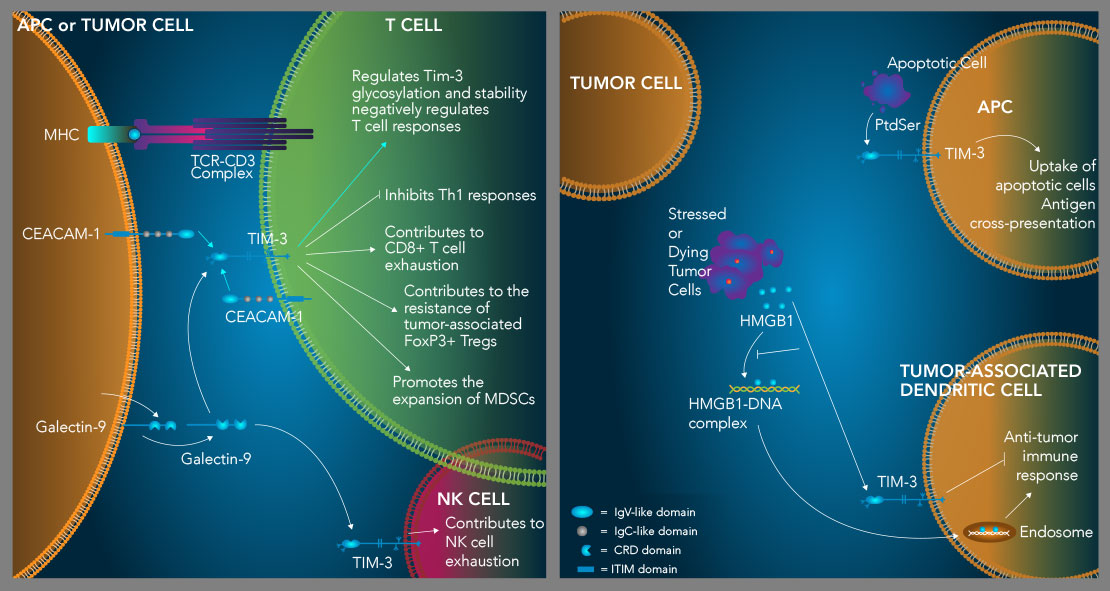
TIM-3 suppresses Th1 immune responses, contributes to T cell and NK cell exhaustion, and enhances the suppressive activity of Tregs. TIM-3 is an inhibitory receptor expressed on multiple hematopoietic cell types. High level expression of TIM-3 on CD8+ T cells and natural killer cells is associated with an exhausted phenotype, while TIM-3 expression on tumor-associated FoxP3+ regulatory T cells (Tregs) marks a subset of Tregs with enhanced suppressor functions and increased resiliency. TIM-3 binds to four different ligands including Galectin-9 (Gal-9), CEACAM-1, HMGB1, and phosphatidylserine (PtdSer). TIM-3 is expressed on Th1 cells and binding of galectin-9 to TIM-3 on these cells induces Th1 cell apoptosis. IFN-gamma produced by Th1 cells up-regulates the expression of galectin-9, which promotes the expansion of myeloid-derived suppressor cells (MDSCs). MDSCs produce increased levels of galectin-9, further driving Th1 cell apoptosis and CD8+ T cell exhaustion. TIM-3 also binds to CEACAM-1, which is required for TIM-3 glycosylation, stability, and its ability to negatively regulate T cell responses (left). Additionally, TIM-3 interacts with the damage-associated molecule, HMGB1, which inhibits the ability of HMGB1 to transport tumor-derived nucleic acids into endosomes in tumor-associated dendritic cells to initiate anti-tumor immune responses (right). Finally, TIM-3 binds to PtdSer on apoptotic cells and mediates apoptotic cell uptake and antigen cross-presentation by dendritic cells (right). How this interaction may affect TIM-3+ T cells is currently unknown.
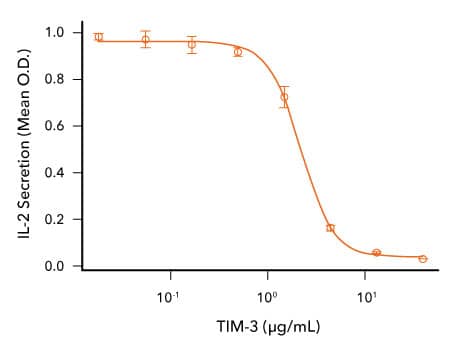
TIM-3 Inhibits Anti-CD3-induced IL-2 Secretion by Human T Cells. Human T cells were incubated with an immobilized Mouse Anti-Human CD3 epsilon Monoclonal Antibody (R&D Systems, Catalog # MAB100; 1μg/mL) and the indicated concentrations of Recombinant Cynomolgus Monkey TIM-3 (R&D Systems, Catalog # 7914-TM). IL-2 secretion was measured in cell culture supernatants using the Human IL-2 QuantikineTM ELISA Kit (R&D Systems, Catalog # D2050). The ED50 for this effect is typically 0.5-3 μg/mL.
References
- Freeman, G.J. et al. (2010) TIM genes: a family of cell surface phosphatidylserine receptors that regulate innate and adaptive immunity. Immunol. Rev. 235:172. PMID: 20536563.
- Monney, L. et al. (2002) Th1-specific cell surface protein Tim-3 regulates macrophage activation and severity of an autoimmune disease. Nature 415:536. PMID: 11823861.
- Das, M. et al. (2017) Tim-3 and its role in regulating anti-tumor immunity. Immunol. Rev. 276:97. PMID: 28258697.
- Sanchez-Fueyo, A. et al. (2003) Tim-3 inhibits T helper type 1-mediated auto- and alloimmune responses and promotes immunological tolerance. Nat. Immunol. 4:1093. PMID: 14556005.
- Hastings, W.D. et al. (2009) Tim-3 is expressed on activated human CD4+ T cells and regulates Th1 and Th17 cytokines. Eur. J. Immunol. 39:2492. PMID: 19676072.
- Anderson, A.C. et al. (2016) Lag-3, Tim-3, and TIGIT: Co-inhibitory receptors with specialized functions in immune regulation. Immunity 44:989. PMID: 27192565.
- Anderson, A.C. et al. (2007) Promotion of tissue inflammation by the immune receptor Tim-3 expressed on innate immune cells. Science 318:1141. PMID: 18006747.
- Zhao, H. et al. (2017) Tregs: where we are and what comes next? Front. Immunol. 8:1578. PMID: 29225597.
- Sakuishi, K. et al. (2013) Tim3+FoxP3+ regulatory T cells are tissue-specific promoters of T-cell dysfunction in cancer. Oncoimmunology 2:23849. PMID: 23734331.
- Jones, R.B. et al. (2008) Tim-3 expression defines a novel population of dysfunctional T cells with highly elevated frequencies in progressive HIV-1 infection. J. Exp. Med. 205:2763. PMID: 19001139.
- Sakuishi, K. et al. (2010) Targeting Tim-3 and PD-1 pathways to reverse T cell exhaustion and restore anti-tumor immunity. J. Exp. Med. 207:2187. PMID: 20819927.
- Gallois, A. et al. (2014) Reversal of natural killer cell exhaustion by TIM-3 blockade. Oncoimmunology 3:946365. PMID: 25964857.
- Zhu, C. et al. (2005) The Tim-3 ligand galectin-9 negatively regulates T helper type 1 immunity. Nat. Immunol. 6:1245. PMID: 16286920.
- Cao, E. et al. (2007) T cell immunoglobulin mucin-3 crystal structure reveals a galectin-9-independent ligand-binding surface. Immunity 26:311. PMID: 17363302.
- Bu, X. et al. (2010) T cell/transmembrane, Ig, and mucin-3 allelic variants differentially recognize phosphatidylserine and mediate phagocytosis of apoptotic cells. J. Immunol. 184:1918. PMID: 20083673.
- Chiba, S. et al. (2012) Tumor-infiltrating DCs suppress nucleic acid-mediated innate immune responses through interactions between the receptor TIM-3 and the alarmin HMGB1. Nat. Immunol. 13:832. PMID: 22842346.
- Huang, Y.H. et al. (2015) CEACAM1 regulates TIM-3-mediated tolerance and exhaustion. Nature 517:386. PMID: 25363763.
- Sabatos-Peyton, C.A. et al. (2017) Blockade of Tim-3 to phosphatidylserine and CEACAM1 is a shared feature of anti-Tim-3 antibodies that have functional efficacy. Oncoimmunology 7:1385690. PMID: 29308307.
- Nakayama, M. et al. (2009) Tim-3 mediates phagocytosis of apoptotic cells and cross-presentation. Blood 113:3821. PMID: 19224762.
- Gao, X. et al. (2012) TIM-3 expression characterizes regulatory T cells in tumor tissues and is associated with lung cancer progression. PLoS One 7:30676. PMID: 22363469.
- Sun, H. & C. Sun (2019) The rise of NK cell checkpoints as promising therapeutic targets in cancer immunotherapy. Front. Immunol. 10:2354. PMID: 31681269.
- Anderson, A.C. (2014) Tim-3: An emerging target in the cancer immunotherapy landscape. Cancer Immunol. Res. 2:393. PMID: 24795351.
- Ngiow, S.F. et al. (2011) Anti-TIM3 antibody promotes T cell IFN-gamma-mediated antitumor immunity and suppresses established tumors. Cancer Res. 71:3540. PMID: 21430066.
- Koyama, S. et al. (2016) Adaptive resistance to therapeutic PD-1 blockade is associated with upregulation of alternative immune checkpoints. Nat. Commun. 7:10501. PMID: 26883990.
- Du, W. et al. (2017) TIM-3 as a target for cancer immunotherapy and mechanisms of action. Int. J. Mol. Sci. 18:645. PMID: 28300768.
- Dardalhon, V. et al. (2010) Tim-3/galectin-9 pathway: regulation of Th1 immunity through promotion of CD11b+Ly-6G+ myeloid cells. J. Immunol. 185:1383. PMID: 20574007.
VISTA/B7-H5 is a T Cell Co-inhibitory Checkpoint Protein
VISTA, also known as B7-H5, is an inhibitory immune checkpoint protein primarily expressed on hematopoietic cells, with the highest levels detected in human myeloid cells, CD4+ T cells, and FoxP3+ regulatory T cells.1-3 Unlike other B7 family members that exhibit extracellular regions containing both IgV-like and IgC-like domains, the extracellular domain in VISTA uniquely consists of a single IgV-like domain, resembling that of the B7 family receptors, CD28, CTLA-4, and ICOS. VISTA is proposed to function dually as both a co-inhibitory ligand and a co-inhibitory receptor on T cells.1,2,4,5 When acting as a ligand, VISTA engages an as-yet unidentified receptor on T cells, directly suppressing their activity. Supporting this, experimental data demonstrate that a recombinant VISTA-Ig fusion protein inhibits proliferation and the production of IL-2 and IFN-gamma in CD4+ and CD8+ T cells, while also promoting differentiation of naïve T cells toward FoxP3+ regulatory T cells in vitro.1,2 Similarly, overexpressing VISTA on bone marrow-derived dendritic cells reduces T cell proliferation and cytokine secretion, and this effect can be neutralized with a VISTA-specific monoclonal antibody.2
As an inhibitory receptor, VISTA regulates T cell function independently of antigen-presenting cells (APCs). Mice lacking VISTA accumulate spontaneously activated T cells, elevated pro-inflammatory cytokines, and develop more severe experimental autoimmune encephalomyelitis than wild-type animals.4,6 Furthermore, stimulating CD4+ T cells with a VISTA-specific agonistic antibody inhibits their activation in vitro and in vivo, underscoring its role as an intrinsic inhibitory receptor on these cells.4 This inhibitory effect is thought to arise through VISTA interacting with itself or another receptor during T cell-T cell interactions.7 Additionally, the ligand for VISTA may also be expressed on tumor cells, which may exploit this axis to suppress T cell responses directly. R&D Systems in-house research has identified VSIG-3/IGSF11 as a VISTA ligand capable of inhibiting human T cell proliferation and cytokine production; however, the physiological relevance of this interaction remains to be established.8
Extensive studies suggest that VISTA plays a critical role in modulating anti-tumor immunity. Initial investigations revealed that overexpression of VISTA on tumor cells impairs protective anti-tumor immune responses.2 Subsequent mouse tumor model research showed elevated VISTA levels on CD11b+Gr1+ myeloid-derived suppressor cells and regulatory T cells within the tumor microenvironment, as well as on tumor cells subjected to hypoxic stress.9 Blocking VISTA with monoclonal antibodies increased tumor-specific T cell proliferation, infiltration, and effector activity, while reducing the number of myeloid-derived suppressor cells, inhibiting regulatory T cell development and function, and suppressing tumor growth. Clinically, heightened VISTA expression in human tumors generally correlates with disease progression and poorer patient outcomes.10 Recent data demonstrate that VISTA expression on tumor cells reduces CD8+ T cell infiltration in syngeneic mouse models, and anti-VISTA antibody treatment improves survival.11 Additional evidence suggests that VISTA expressed on tumor-associated macrophages, myeloid-derived suppressor cells, or tumor epithelial cells can interact with PSGL-1 on T cells within acidic tumor microenvironments, selectively dampening immune activation in these niches.12 Given these findings, targeting VISTA, either alone or combined with other immune checkpoint inhibitors, has become a promising area of research in cancer therapy, with strategies adapted to different types of cancer and microenvironmental conditions.13
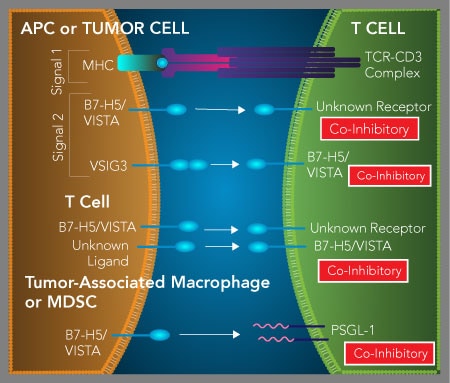
VISTA/B7-H5 functions as both a T cell inhibitory ligand and receptor. VISTA is expressed on both antigen-presenting cells (APCs) and T cells and has been shown to function as both a T cell co-inhibitory ligand and receptor. As a ligand, VISTA on APCs or tumor cells binds to an unidentified receptor expressed on activated T cells and inhibits T cell proliferation and cytokine production. Recently, it has been suggested that expression of VISTA on tumor cells or myeloid cells present in the tumor microenvironment may inhibit T cell functions by interacting with T cell-expressed PSGL-1 under acidic conditions. In addition to functioning as a T cell co-inhibitory ligand, T cell-expressed VISTA functions as a co-inhibitory receptor to suppress T cell functions. This interaction has been proposed to occur by VISTA interacting with either itself or a currently unidentified protein through T cell-T cell interactions, or through tumor cell-T cell interactions, if the ligand for VISTA is expressed on tumor cells.
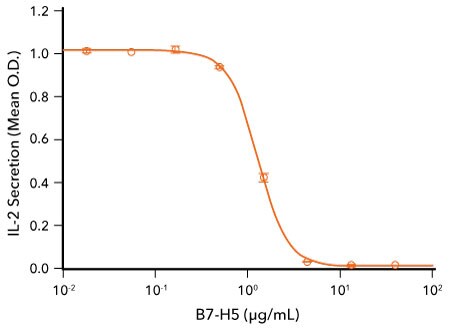
VISTA Inhibits Anti-CD3-Induced IL-2 Secretion by Human T Cells. Human T cells were incubated with an immobilized Mouse Anti-Human CD3 epsilon Monoclonal Antibody (R&D Systems, Catalog # MAB100) and the indicated concentrations of Recombinant Human VISTA/B7-H5 Fc Chimera (R&D Systems, Catalog # 7126-B7). IL-2 secretion was measured in cell culture supernatants using the Human IL-2 QuantikineTM ELISA Kit (R&D Systems, Catalog # D2050). The ED50 for this effect is typically 1-6 μg/mL.
References
- Lines, J.L. et al. (2014) VISTA is an immune checkpoint molecule for human T cells. Cancer Res. 74:1924. PMID: 24691993.
- Wang, L. et al. (2011) VISTA, a novel mouse Ig superfamily ligand that negatively regulates T cell responses. J. Exp. Med. 208:577. PMID: 21383057.
- Flies, D.B. et al. (2011) Cutting edge: a monoclonal antibody specific for the programmed death-1 homolog prevents graft-versus-host disease in mouse models. J. Immunol. 187:1537. PMID: 21768399.
- Flies, D.B. et al. (2014) Coinhibitory receptor PD-1H preferentially suppresses CD4+ T cell-mediated immunity. J. Clin. Invest. 124:1966. PMID: 24743150.
- Schildberg, F.A. et al. (2016) Coinhibitory pathways in the B7-CD28 ligand-receptor family. Immunity 44:955. PMID: 27192563.
- Wang, L. et al. (2014) Disruption of the immune-checkpoint VISTA gene imparts a proinflammatory phenotype with predisposition to the development of autoimmunity. Proc. Natl. Acad. Sci. USA 111:14846. PMID: 25267631.
- Xu, W. et al. (2018) The structure, expression, and multifaceted role of immune-checkpoint protein VISTA as a critical regulator of anti-tumor immunity, autoimmunity, and inflammation. Cell. Mol. Immunol. 15:438. PMID: 29375120.
- Wang, J. et al. (2019) VSIG-3 as a ligand of VISTA inhibits human T-cell function. Immunology 156:74. PMID: 30220083.
- Le Mercier, I. et al. (2014) VISTA regulates the development of protective anti-tumor immunity. Cancer Res. 74:1933. PMID: 24691994.
- EITanbouly, M.A. et al. (2019) VISTA: a novel immunotherapy target for normalizing innate and adaptive immunity. Semin. Immunol. 42:101308. PMID: 31604531.
- Mulati, K. et al. (2019) VISTA expressed in tumour cells regulates T cell function. Br. J. Cancer 120:115. PMID: 30382166.
- Johnston, R.J. et al. (2019) VISTA is an acidic pH-selective ligand for PSGL-1. Nature 574:565. PMID: 31645726.
- EITanbouly, M.A. et al. (2020) VISTA: Coming of age as a multi-lineage immune checkpoint. Clin. Exp. Immunol. 200:120. PMID: 31930484.
Co-Stimulatory Immune Checkpoint Targets
Alongside strategies that focus on blocking inhibitory immune checkpoint receptors to boost anti-tumor immunity, current research is also exploring the use of agonists targeting co-stimulatory immune checkpoint receptors to enhance immune cell functions. Most of these co-stimulatory receptors belong to the tumor necrosis factor receptor superfamily (TNFRSF) and are predominantly found on T cells, B cells, and natural killer (NK) cells.
Activation of these receptors, either through their natural ligands or specific agonistic antibodies, triggers signaling pathways that significantly enhance immune cell survival, proliferation, and effector functions. The therapeutic potential of stimulating these co-stimulatory receptors alone or in combination with blockade of inhibitory immune checkpoints such as CTLA-4 and PD-1 to improve cancer treatment outcomes is being actively investigated.
Activation of 4-1BB by 4-1BB L Delivers a CD28-Independent Co-Stimulatory Signal
4-1BB, also referred to as CD137 and TNFRSF9, is widely expressed across diverse immune cell populations, including activated CD4+ and CD8+ T cells, natural killer (NK) cells, NKT cells, dendritic cells, and regulatory T cells.1-5 Its ligand, 4-1BB Ligand/TNFSF9, which is present on activated macrophages, B cells, and dendritic cells, binds 4-1BB with high affinity.6-8 This binding initiates a co-stimulatory signal that drives the activation, expansion, and survival of CD4+ and CD8+ T cells independently of CD28-mediated co-stimulation.8-11 Engagement of 4-1BB on CD8+ T cells enhances T cell receptor (TCR) signaling and augments their cytotoxic activity.12 Likewise, triggering 4-1BB on NK cells drives the production of IFN-gamma and increases their cytolytic function.13 In human NK cells, 4-1BB expression is upregulated following Fc receptor engagement, and subsequent 4-1BB activation boosts NK cell-mediated killing through antibody-dependent cell-mediated cytotoxicity.14-16 During dendritic cell maturation, 4-1BB expression is significantly elevated, and its activation leads to increased expression of the co-stimulatory molecules B7-1/CD80 and B7-2/CD86, enhanced cytokine secretion, and improved survival.3,17
In mouse tumor models, administration of agonistic anti-4-1BB antibodies has resulted in powerful anti-tumor immune responses, which have been linked to heightened CD8+ T cell and NK cell activity, alongside a reduction in the suppressive function of regulatory T cells.18-20 Moreover, combining agonistic anti-4-1BB antibodies with anti-CTLA-4 antibodies in these models has shown a synergistic effect that further amplifies anti-tumor immunity.21-23 Based on these findings, the use of agonistic anti-4-1BB antibodies, either alone or in combination with agonistic anti-OX40 antibodies or antagonistic antibodies targeting inhibitory immune checkpoints, is currently under investigation for the treatment of multiple human cancers.24,25
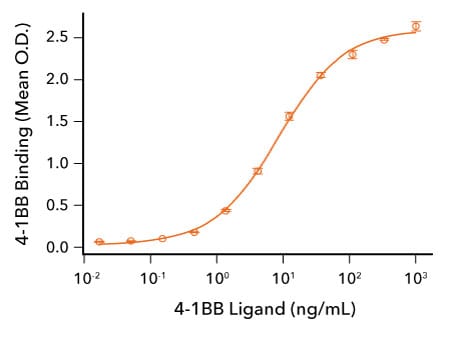
Human 4-1BB Ligand Binds 4-1BB. Recombinant Human 4-1BB/TNFRSF9 (R&D Systems, Catalog # 9220-4B) was immobilized at 50 ng/mL and the indicated concentrations of Recombinant Human 4-1BB Ligand (R&D Systems, Catalog # 2295-4L) were added. Recombinant Human 4-1BB Ligand bound with an ED50 of 2.5-15 ng/mL.
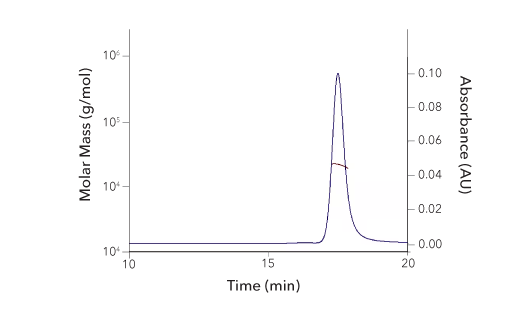
| SEC-MALS Data | Result |
| Retention Time | 17.3-17.9 min |
| MW-Predicted (Monomer) | 18.0 kDa |
| MW-MALS | 21.8 kDa |
| Polydispersity | 1.002 |
| System Suitability: BSA Monomer 66.4 ± 3.32 kDa | Pass |
Recombinant Human 4‑1BB/TNFRSF9/CD137 Protein SEC-MALS. Recombinant Human 4-1BB/TNFRSF9 (Catalog # 9220-4B) has a molecular weight (MW) of 21.8 kDa as analyzed by SEC-MALS, suggesting that this protein is a monomer. MW may differ from predicted MW due to post-translational modifications (PTMs) present (i.e. Glycosylation).
References
1. Wen, T.et al. (2002) 4-1BB ligand-mediated costimulation of human T cells induces CD4 and CD8 T cell expansion, cytokine production, and the development of cytolytic effector function. J. Immunol. 168:4897. PMID: 11994439
2. Zheng, G. et al (2004) The 4-1BB costimulation augments the proliferation of CD4+CD25+ regulatory T cells. J. Immunol. 173:2428. PMID: 15294956
3. Choi, B.K. et al (2009) 4-1BB functions as a survival factor in dendritic cells. J. Immunol. 182:4107. PMID: 19299708
4. Vinay, D.S. & B.S. Kwon:(2011) 4-1BB signaling beyond T cells. Cell. Mol. Immunol. 8:281. PMID: 21217771
5. Kim, D. et al (2008) 4-1BB engagement costimulates NKT cell activation and exacerbates NKT cell ligand-induced airway hyperresponsiveness and inflammation. J. Immunol. 180:2062. PMID: 18250411
6. Goodwin, R.G. et al (1993) Molecular cloning of a ligand for the inducible T cell gene 4-1BB: a member of an emerging family of cytokines with homology to tumor necrosis factor. Eur. J. Immunol. 23:2631. PMID: 8405064
7. Pollok, K.E. et al (1994) 4-1BB T-cell antigen binds to mature B cells and macrophages, and costimulates anti-mu-primed splenic B cells. Eur. J. Immunol. 24:367. PMID: 8299685
8. DeBenedette, M.A. et al (1997) Costimulation of CD28- T lymphocytes by 4-1BB ligand. J. Immunol. 158:551. PMID: 8992967
9. Chu, N.R. et al (1997) Role of IL-12 and 4-1BB ligand in cytokine production by CD28+ and CD28- T cells. J. Immunol. 158:3081. PMID: 9120260
10. Saoulli, K.S. et al (1998) CD28-independent, TRAF2-dependent costimulation of resting T cells by 4-1BB ligand. J. Exp. Med. 187:1849. PMID: 9607925
11. Cannons, J.L. et al (2001) 4-1BB ligand induces cell division, sustains survival, and enhances effector function of CD4 and CD8 T cells with similar efficacy. J. Immunol. 167:1313. PMID: 11466348
12. Shuford, W.W. et al (1997) 4-1BB costimulatory signals preferentially induce CD8+ T cell proliferation and lead to the amplification in vivo of cytotoxic T cell responses. J. Exp. Med. 186:47. PMID: 9206996
13. Melero, I. et al (1998) NK1.1 cells express 4-1BB (CDw137) costimulatory molecule and are required for tumor immunity elicited by anti-41BB monoclonal antibodies. Cell. Immunol. 190:167. PMID: 9878117
14. Lin, W. et al (2008) Fc-dependent expression of CD137 on human NK cells: insights into "agonistic" effects of anti-CD137 monoclonal antibodies. Blood 112:699. PMID: 18519814
15. Kohrt, H.E. et al (2011) CD137 stimulation enhances the antilymphoma activity of anti-CD20 antibodies. Blood 117:2423. PMID: 21193697
16. Wang, W. et al (2015) NK cell-mediated antibody-dependent cellular cytotoxicity in cancer immunotherapy. Front. Immunol. 6:368. PMID: 26284063
17. Kuang, Y. et al (2012) Effects of 4-1BB signaling on the biological function of murine dendritic cells. Oncol. Lett. 3:477. PMID: 22740935
18. Melero, I. et al (1997) Monoclonal antibodies against the 4-1BB T-cell activation molecule eradicate established tumors. Nat. Med. 3:682. PMID: 9176498
19. Xu, D. et al (2004) NK and CD8+ T cell-mediated eradication of poorly immunogenic B16-F10 melanoma by the combined action of IL-12 gene therapy and 4-1BB costimulation. Int. J. Cancer 109:499. PMID: 14991570
20. Buchan, S.L.et al. (2018) Antibodies to costimulatory receptor 4-1BB enhance anti-tumor immunity via T regulatory cell depletion and promotion of CD8 T cell effector function. Immunity 49:958. PMID: 30446386
21. Kocak, E. et al (2006) Combination therapy with anti-CTL antigen-4 and anti-4-1BB antibodies enhances cancer immunity and reduces autoimmunity. Cancer Res. 66:7276. PMID: 16849577
22. Li, B. et al (2007) Established B16 tumors are rejected following treatment with GM-CSF-secreting tumor cell immunotherapy in combination with anti-4-1BB mAb. Clin. Immunol. 125:76. PMID: 17706463
23. Curran, M. et al (2011) Combination CTLA-4 blockade and 4-1BB activation enhances tumor rejection by increasing T-cell infiltration, proliferation, and cytokine production. PLoS One 6:19499. PMID: 21559358
24. Dempke, W.C.M. et al (2017) Second- and third-generation drugs for immuno-oncology treatment-the more the better? Eur. J. Cancer 74:55. PMID: 28335888
25. Marin-Acevedo, J.A.:(2018) Next generation of immune checkpoint therapy in cancer: new developments and challenges. J. Hematol. Oncol. 11:39. PMID: 29544515
CD27 Binding to CD70 Promotes T Cell Expansion and Improves Anti-Tumor Immunity
CD27, also known as TNFRSF7, is mainly expressed on T cells, B cells, and natural killer (NK) cells. Although CD27 is present at low levels on naïve CD4+ and CD8+ T cells, its expression is upregulated following T cell activation.1,2 CD27 engages with its ligand, CD27 Ligand/CD70, which is expressed on activated dendritic cells, B cells, T cells, and NK cells, providing a co-stimulatory signal that complements CD28 signaling during T cell activation.3-9 Activation of CD27 on T cells facilitates antigen-specific clonal expansion of CD4+ and CD8+ cell populations, supports the differentiation of Th1 and CD8+ effector T cells, and promotes the survival of effector T cells.5,10-15 Additionally, a soluble variant of CD27 can be generated through proteolytic shedding from the surface of activated T cells.4 While absent on naïve B cells, CD27 expression increases on activated B cells, promoting their proliferation within germinal centers and aiding in the development of both effector and memory B cell subsets.3,4 CD27 is also present on NK cells, where its stimulation enhances NK cell proliferation and boosts IFN-gamma secretion.2
In mouse models bearing EL-4 lymphoma tumors, transgenic expression of CD70 was demonstrated to drive tumor regression through mechanisms dependent on CD8+ T cells and IFN-gamma.16 Moreover, an agonistic anti-CD27 monoclonal antibody showed anti-tumor activity in immunocompetent mice implanted with syngeneic T cell or BCL1 lymphoma, with comparable results observed in a melanoma model.17-19 The use of a fully human agonistic anti-CD27 antibody also yielded significant anti-tumor effects in syngeneic tumor models expressing human CD27.20 When combined with anti-CTLA-4 blocking antibodies, this human anti-CD27 antibody synergistically enhanced anti-tumor immunity.21 As a result, both agonistic anti-CD27 and anti-CD70 antibodies are being evaluated in clinical trials for their potential to treat hematologic cancers as well as solid tumors.22,23
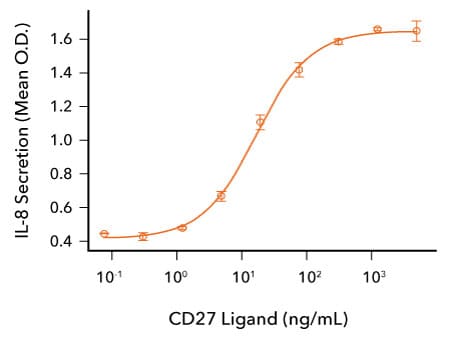
CD27 Ligand/CD70 Induces IL-8 Secretion by Human Fibrosarcoma Cells. HT1080 human fibrosarcoma cells transfected with human CD27 were treated with the indicated concentrations of Recombinant Human CD27 Ligand/TNFSF7 (R&D Systems, Catalog # 9328-CL). IL-8 secretion was measured in cell culture supernatants using the Human IL-8/CXCL8 QuantikineTM ELISA Kit (R&D Systems, Catalog # D8000C). The ED50 for this effect is 5-25 ng/mL.
References
1. Loenen, W.A.et al. (1992) The CD27 membrane receptor, a lymphocyte-specific member of the nerve growth factor receptor family, gives rise to a soluble form by protein processing that does not involve receptor endocytosis. Eur. J. Immunol. 22:447. PMID: 1311261
2. Takeda, K. et al (2000) CD27-mediated activation of murine NK cells. J. Immunol. 164:1741. PMID: 10657619
3. Lens, S.M. et al (1998) Control of lymphocyte function through CD27-CD70 interactions. Semin. Immunol. 19:491. PMID: 9826582
4. Borst, J. et al (2005) CD27 and CD70 in T cell and B cell activation. Curr. Opin. Immunol. 17:275. PMID: 15886117
5. Goodwin, R.G. et al (1993) Molecular and biological characterization of a ligand for CD27 defines a new family of cytokines with homology to tumor necrosis factor. Cell 73:447. PMID: 8387892
6. Bowman, M.R. et al (1994) The cloning of CD70 and its identification as the ligand for CD27. J. Immunol. 152:1756. PMID: 8120384
7. Hintzen, R.Q. et al (1994) CD70 represents the human ligand for CD27. Int. Immunol. 6:477. PMID: 8186199
8. Tesselaar, K. et al (2003) Expression of the murine CD27 ligand CD70 in vitro and in vivo. J. Immunol. 170:33. PMID: 12496380
9. Nolte, M.A. et al (2009) Timing and tuning of CD27-CD70 interactions: the impact of signal strength in setting the balance between adaptive responses and immunopathology. Immunol. Rev. 229:216. PMID: 19426224
10. Van Oosterwijk, M.F. et al (2007) CD27-CD70 interactions sensitise naive CD4+ T cells for IL-12-induced Th1 cell development. Int. Immunol. 19:713. PMID: 17548342
11. Xiao, Y. et al (2008) CD27 instructs CD4+ T cells to provide help for the memory CD8+ T cell response after protein immunization. J. Immunol. 181:1071. PMID: 18606659
12. Rowley, T.F. & A.A. Shamkhani:(2004) Stimulation by soluble CD70 promotes strong primary and secondary CD8+ cytotoxic T cell responses in vivo. J. Immunol. 172:6039. PMID: 15128787
13. Van Gisbergen, K. et al (2011) The costimulatory molecule CD27 maintains clonally diverse CD8(+) T cell responses of low antigen affinity to protect against viral variants. Immunity 35:97. PMID: 21763160
14. Peperzak, V. et al (2010) The Pim kinase pathway contributes to survival signaling in primed CD8+ T cells upon CD27 costimulation. J. Immunol. 185:6670. PMID: 21048108
15. Pen, J.J. et al (2013) Modulation of regulatory T cell function by monocyte-derived dendritic cells matured through electroporation with mRNA encoding CD40 ligand, constitutively active TLR4, and CD70. J. Immunol. 191:1976. PMID: 23842750
16. Arens, R. et al (2004) Tumor rejection induced by CD70-mediated quantitative and qualitative effects on effector CD8+ T cell formation. J. Exp. Med. 199:1595. PMID: 15184507
17. Sakanishi, T. & H. Yagita:(2010) Anti-tumor effects of depleting and non-depleting anti-CD27 monoclonal antibodies in immune-competent mice. Biochem. Biophys. Res. Commun. 393:829. PMID: 20171165
18. French, R.R. et al (2007) Eradication of lymphoma by CD8 T cells following anti-CD40 monoclonal antibody therapy is critically dependent on CD27 costimulation. Blood 109:4810. PMID: 17311995
19. Roberts, D.J. et al (2010) Control of established melanoma by CD27 stimulation is associated with enhanced effector function and persistence, and reduced PD-1 expression of tumor infiltrating CD8(+) T cells. J. Immunother. 33:769. PMID: 20842060
20. He, L-Z. et al (2013) Agonist anti-human CD27 monoclonal antibody induces T cell activation and tumor immunity in human CD27-transgenic mice. J. Immunol. 191:4174. PMID: 24026078
21. He, L-Z. et al (2013) Combination therapies augment the anti-tumor activity of agonist CD27 mAb in human CD27 transgenic mouse models. J. Immunother. Cancer 1:76. PMID:
22. Dempke, W.C.M. et al (2017) Second- and third-generation drugs for immuno-oncology treatment-the more the better? Eur. J. Cancer 74:55. PMID: 28335888
23. Marin-Acevedo, J.A.:(2018) Next generation of immune checkpoint therapy in cancer: new developments and challenges. J. Hematol. Oncol. 11:39. PMID: 29544515
CD40 Signaling Indirectly Promotes T Cell Priming, Activation, and Th1 Polarization
CD40, also referred to as TNFRSF5, is a type I transmembrane receptor within the TNF receptor superfamily. It is expressed on the surface of antigen-presenting cells (APCs) including dendritic cells, macrophages, monocytes, and B lymphocytes.1,2 Its ligand, CD40 Ligand, is found on activated T cells and B cells, and its expression can be triggered on natural killer cells, monocytes, basophils, and mast cells during inflammatory responses.3 The binding of CD40 to CD40 Ligand facilitates B cell activation and proliferation, and promotes T cell-dependent humoral immunity.3,4 Moreover, engagement of CD40 initiates signaling pathways that induce the production of cytokines, co-stimulatory molecules, adhesion factors, and enzymes that influence both cellular and humoral immune responses.5,6 By modulating co-stimulatory molecule expression and cytokine release, such as TNF-alpha and IL-12, CD40 signaling indirectly impacts T cell priming, activation processes, and Th1 differentiation.
CD40 expression has also been identified on a variety of tumor cells, where it can either promote or inhibit tumor progression depending on the biological context.6 While simultaneous expression of CD40 and CD40 Ligand on tumor cells can enhance their growth and motility, the CD40/CD40 Ligand interaction may also exert direct cytotoxic effects on cancer cells or suppress tumor growth by activating APCs. Significantly, agonistic anti-CD40 antibodies have been shown to trigger robust CD8+ T cell-mediated anti-tumor responses in syngeneic mouse lymphoma models.7-9 Positive anti-tumor effects have further been observed in early phase clinical trials employing recombinant human CD40 Ligand and agonistic anti-CD40 antibodies in patients with various advanced cancers.10-12 Many of these agonists continue to be assessed in clinical trials, supporting the hypothesis that CD40/CD40 L stimulation may serve as a promising approach for cancer treatment.13,14
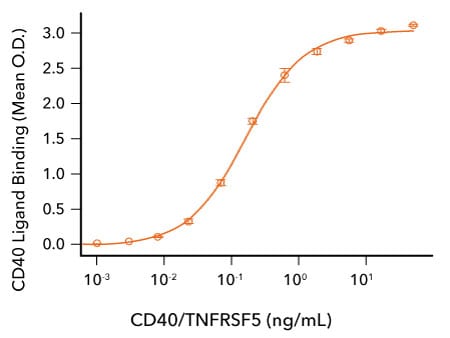
CD40/TNFRSF5 Binds to CD40 Ligand. Recombinant Human CD40 Ligand/TNFSF5 (R&D Systems, Catalog # 6420-CL; HEK293-expressed) was immobilized at 2 μg/mL, 100 μL/well and the indicated concentrations of Avi-tag Biotinylated Recombinant Human CD40/TNFRSF5 Fc Chimera (R&D Systems, Catalog # AVI10380) were added. The concentration of Avi-tag Biotinylated Recombinant Human CD40 that produces 50% optimal binding is 40-320 ng/mL.
References
1. van Kooten, C. & J. Banchereau:(1997) Functions of CD40 on B cells, dendritic cells and other cells. Curr. Opin. Immunol. 9:330. PMID: 9203418
2. Schonbeck, U. et al (1997) Ligation of CD40 activates interleukin 1beta-converting enzyme (caspase-1) activity in vascular smooth muscle and endothelial cells and promotes elaboration of active interleukin 1beta. J. Biol. Chem. 272:19569. PMID: 9235962
3. Elgueta, R. et al (2009) Molecular mechanism and function of CD40/CD40L engagement in the immune system. Immunol. Rev. 229:152. PMID: 19426221
4. Rickert, R.C. et al (2011) Signaling by the tumor necrosis factor receptor superfamily in B-cell biology and disease. Immunol. Rev. 244:115. PMID: 22017435
5. Schonbeck, U. et al (2001) The CD40/CD154 receptor/ligand dyad. Cell. Mol. Life Sci. 58:4. PMID: 11229815
6. Kawabe, T. et al (2011) CD40/CD40 ligand interactions in immune responses and pulmonary immunity. Nagoya J. Med. Sci. 73:69. PMID: 21928689
7. van Kooten, C. & J. Banchereau:(2000) CD40-CD40 ligand. J. Leukoc. Biol. 67:2. PMID: 10647992
8. French, R.R. et al (1999) CD40 antibody evokes a cytotoxic T-cell response that eradicates lymphoma and bypasses T-cell help. Nat. Med. 5:548. PMID: 10229232
9. Tutt, A.L. et al (2002) T cell immunity to lymphoma following treatment with anti-CD40 monoclonal antibody. J. Immunol. 168:2720. PMID: 11884438
10. Vonderheide, R.H. et al (2001) Phase I study of recombinant human CD40 ligand in cancer patients. J. Clin. Oncol. 19:3280. PMID: 11432896
11. Vonderheide, R.H. et al (2007) Prospect of targeting the CD40 pathway for cancer therapy. Clin. Cancer Res. 13:1083. PMID: 17317815
12. Vonderheide, R.H. et al (2013) Agonistic CD40 antibodies and cancer therapy. Clin. Cancer Res. 19:1035. PMID: 23460534
13. Dempke, W.C.M. et al (2017) Second- and third-generation drugs for immuno-oncology treatment-the more the better? Eur. J. Cancer 74:55. PMID: 28335888
14. Marin-Acevedo, J.A.:(2018) Next generation of immune checkpoint therapy in cancer: new developments and challenges. J. Hematol. Oncol. 11:39. PMID: 29544515
GITR:GITR L Binding Promotes T Cell Activation and Inhibits Regulatory T Cell Activity
Glucocorticoid-induced TNF receptor family-related protein (GITR), also known as TNFRSF18, is a co-stimulatory receptor belonging to the TNF receptor superfamily. GITR is a type I transmembrane protein that is expressed at low levels on resting CD4+ and CD8+ T cells, but is upregulated upon T cell activation.1-4 GITR is also expressed on natural killer (NK) cells and is highly abundant on CD4+CD25+ regulatory T cells, where its levels increase further following activation.5-7 GITR binds to GITR Ligand (TNFSF18), which is a type II transmembrane protein primarily expressed by antigen-presenting cells such as macrophages, B cells, and dendritic cells (DCs), with its expression notably elevated on DCs after activation.8-10 Stimulation of GITR by GITR Ligand enhances T cell proliferation triggered by anti-CD3, co-stimulates activation of both CD4+ and CD8+ T cells, and promotes NK cell activation, cytotoxicity, and production of IFN-gamma.1,3,4,7,8 Additionally, GITR activation on regulatory T cells leads to their proliferation, but diminishes their suppressive functions.4,6,11
GITR has emerged as a compelling target for cancer immunotherapy because agonists directed at this receptor activate CD4+ and CD8+ effector T cells, while concurrently suppressing regulatory T cell activity. In various syngeneic mouse tumor models, both recombinant GITR Ligand-Fc fusion proteins and agonistic anti-GITR antibodies elicited robust anti-tumor immune responses.12-16 The anti-tumor efficacy was associated with increased tumor infiltration by CD4+ and CD8+ T cells, expansion of IFN-gamma-secreting CD8+ T cells and NK cells, and reduction of regulatory T cells within the tumor microenvironment.12-16 Furthermore, combination therapies involving agonistic anti-GITR and anti-CTLA-4 blocking antibodies demonstrated synergistic enhancement of anti-tumor effects.12,17 As a result, several agonistic anti-GITR antibodies are now under evaluation in phase I clinical trials.18,19
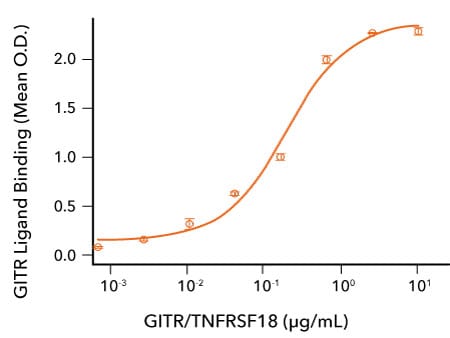
GITR Binds to GITR Ligand. Recombinant Human GITR Ligand/TNFSF18 (R&D Systems, Catalog # 6987-GL) was immobilized at 0.5 μg/mL, 100 μL/well and the indicated concentrations of Avi-tag Biotinylated Recombinant Human GITR/TNFRSF18 Fc Chimera (R&D Systems, Catalog # AVI689) were added. Avi-tag Biotinylated Recombinant Human GITR/TNFRSF18 bound with an ED50 of 0.05-0.5 μg/mL.
References
1. Nocentini, G.et al. (1997) A new member of the tumor necrosis factor/nerve growth factor receptor family inhibits T cell receptor-induced apoptosis. Proc. Natl. Acad. Sci. USA 94:6216. PMID: 9177197
2. Kwon, B. et al (1999) Identification of a novel activation-inducible protein of the tumor necrosis factor receptor superfamily and its ligand. J. Biol. Chem. 274:6056. PMID: 10037686
3. Gurney, A.L. et al (1999) Identification of a new member of the tumor necrosis factor family and its receptor, a human ortholog of mouse GITR. Curr. Biol. 9:215. PMID: 10074428
4. Ronchetti, S. et al (2004) GITR, a member of the TNF receptor superfamily, is costimulatory to mouse T lymphocyte subpopulations. Eur. J. Immunol. 34:613. PMID: 14991590
5. Shimizu, J. et al (2002) Stimulation of CD25(+)CD4(+) regulatory T cells through GITR breaks immunological self-tolerance. Nat. Immunol. 3:135. PMID: 11812990
6. Kanamaru, F. et al (2004) Costimulation via glucocorticoid-induced TNF receptor in both conventional and CD25+ regulatory CD4+ T cells. J. Immunol. 172:7306. PMID: 15187106
7. Hanabuchi, S. et al (2006) Human plasmacytoid predendritic cells activate NK cells through glucocorticoid-induced tumor necrosis factor receptor-ligand (GITRL). Blood 107:3617. PMID: 16397134
8. Tone, M. et al (2003) Mouse glucocorticoid-induced tumor necrosis factor receptor ligand is costimulatory for T cells. Proc. Natl. Acad. Sci, USA 100:15059. PMID: 14608036
9. Kim, J.D. et al (2003) Cloning and characterization of GITR ligand. Genes Immunol. 4:564. PMID: 14647196
10. Yu, K. et al (2003) Identification of a ligand for glucocorticoid-induced tumor necrosis factor receptor constitutively expressed in dendritic cells. Biochem. Biophys. Res. Commun. 310:433. PMID: 14521928
11. McHugh, R.S. et al (2002) CD4(+)CD25(+) immunoregulatory T cells: gene expression analysis reveals a functional role for the glucocorticoid-induced TNF receptor. Immunity 16:311. PMID: 11869690
12. Hu,, P. et al (2008) Construction and preclinical characterization of Fc-mGITRL for the immunotherapy of cancer. Clin. Cancer Res. 14:579. PMID: 18223234
13. Ko, K. et al (2005) Treatment of advanced tumors with agonistic anti-GITR mAb and its effects on tumor-infiltrating FoxP3+CD25+CD4+ regulatory T cells. J. Exp. Med. 202:885. PMID: 16186187
14. Zhou, P. et al (2007) Pivotal roles of CD4+ effector T cells in mediating agonistic anti-GITR mAb-induced immune activation and tumor immunity in CT26 tumors. J. Immunol. 179:7365. PMID: 18025180
15. Coe, D. et al (2010) Depletion of regulatory T cells by anti-GITR mAb as a novel mechanism for cancer immunotherapy. Cancer Immunol. Immunother. 59:1367. PMID: 20480365
16. Cohen, A.D. et al (2010) Agonist anti-GITR monoclonal antibody induces melanoma tumor immunity in mice by altering regulatory T cell stability and intra-tumor accumulation. PLoS One 5:10436. PMID: 20454651
17. Mitsui, J. et al (2010) Two distinct mechanisms of augmented antitumor activity by modulation of immunostimulatory/inhibitory signals. Clin. Cancer Res. 16:2781. PMID: 20460483
18. Dempke, W.C.M. et al (2017) Second- and third-generation drugs for immuno-oncology treatment-the more the better? Eur. J. Cancer 74:55. PMID: 28335888
19. Marin-Acevedo, J.A. et al (2018) Next generation of immune checkpoint therapy in cancer: new developments and challenges. J. Hematol. Oncol. 11:39. PMID: 29544515
OX40 Signaling Stimulates T Cell Activation and Promotes Tumor Regression
OX40, also referred to as TNFRSF4 and CD134, is a type I transmembrane receptor that is primarily expressed on activated CD4+ and CD8+ T cells, activated natural killer cells, NKT cells, and neutrophils.1-4 Its binding partner, OX40 Ligand (OX40L), is a type II transmembrane protein that is expressed on activated antigen-presenting cells, including B cells, macrophages, and dendritic cells (DCs), in response to inflammatory cytokines or following stimulation through the B cell receptor (BCR), CD40, or toll-like receptors (TLRs).3,5 The interaction between OX40 and OX40L delivers a robust co-stimulatory signal that enhances T cell proliferation, survival, and effector functions.3-5 Moreover, OX40 engagement can diminish the suppressive activity of regulatory T cells and enhance natural killer cell-mediated cytotoxicity and IFN-gamma secretion, particularly when antibody: Fc receptor interactions occur.2,6,7
In mouse tumor models, treatment with agonistic anti-OX40 antibodies or OX40 Ligand-Fc fusion proteins has resulted in tumor regression, largely due to increased infiltration of T cells into the tumor and enhanced expansion and activity of both CD4+ and CD8+ T cells.8-12 Additional preclinical studies have shown that combining an agonistic anti-OX40 antibody with immune checkpoint inhibitors such as anti-CTLA-4 or anti-PD-1 blocking antibodies can produce synergistic improvements in anti-tumor immune responses.13-15 Clinical evaluation of a mouse anti-OX40 agonistic antibody in patients with advanced cancers demonstrated good tolerability and induced immunostimulatory effects, similar to those observed in animal models.16 Collectively, these data support the potential of OX40-targeted therapies, either alone or in combination with immune checkpoint blockade, as promising approaches for cancer treatment.
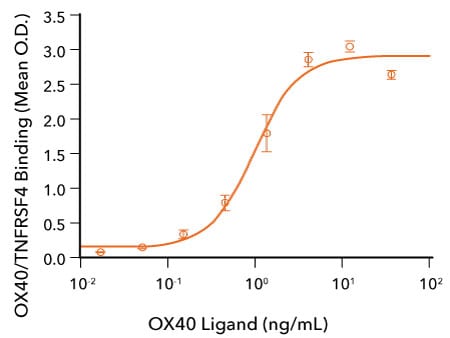
OX40 Ligand Binds to OX40/TNFRSF4. Recombinant Human OX40/TNFRSF4 (R&D Systems, Catalog # 9969-OX) was immobilized at 0.25 μg/mL and the indicated concentrations of Recombinant Human OX40 Ligand/TNFSF4 (R&D Systems, Catalog # 1054-OX) were added. Recombinant Human OX40 Ligand bound with an ED50 of 0.25-1.5 ng/mL.
References
1. Gramaglia, I. et al. (1998) OX-40 ligand: a potent costimulatory molecule for sustaining primary CD4 T cell responses. J. Immunol. 161:6510. PMID: 9862675
2. Turaj, A.H. et al (2018) Augmentation of CD134 (OX40)-dependent NK anti-tumour activity is dependent on antibody cross-linking. Sci. Rep. 8:2278. PMID: 29396470
3. Croft, M.:(2010) Control of immunity by the TNFR-related molecule OX40 (CD134). Ann. Rev. Immunol. 28:57. PMID: 20307208
4. Willoughby, J. et al (2017) OX40: Structure and function - What questions remain? Mol. Immunol. 83:13. PMID: 28092803
5. Mendel, I. & E.M. Shevach:(2005) Activated T cells express the OX40 ligand: requirements for induction and costimulatory function. Immunology 117:196. PMID: 16423055
6. Takeda, I. et al (2004) Distinct roles for the OX40-OX40 ligand interaction in regulatory and nonregulatory T cells. J. Immunol. 172:3580. PMID: 15004159
7. Aspeslagh, S. et al (2016) Rationale for anti-OX40 cancer immunotherapy. Eur. J. Immunol. 52:50. PMID: 26645943
8. Weinberg, A.D. et al (2000) Engagement of the OX-40 receptor in vivo enhances antitumor immunity. J. Immunol. 164:2160. PMID: 10657670
9. Kjaergaard, J. et al (2000) Therapeutic efficacy of OX-40 receptor antibody depends on tumor immunogenicity and anatomic site of tumor growth. Cancer Res. 60:5514. PMID: 11034096
10. Ali, S.A. et al (2004) Anti-tumor therapeutic efficacy of OX40L in murine tumour model. Vaccine 22:3585. PMID: 15315837
11. Gough, M.J. et al (2008) OX40 agonist therapy enhances CD8 infiltration and decreases immune suppression in the tumor. Cancer Res. 68:5206. PMID: 18593921
12. Piconese, S. et al (2008) OX40 triggering blocks suppression by regulatory T cells and facilitates tumor rejection. J. Exp. Med. 205:825. PMID: 18362171
13. Redmond, W.L. et al (2014) Combined targeting of costimulatory (OX40) and coinhibitory (CTLA-4) pathways elicits potent effector T cells capable of driving robust antitumor immunity. Cancer Immunol. Res. 2:142. PMID: 24778278
14. Marabelle, A. et al (2013) Depleting tumor-specific Tregs at a single site eradicates disseminated tumors. J. Clin. Invest. 123:2447. PMID: 23728179
15. Guo, Z. et al (2014) PD-1 blockade and OX40 triggering synergistically protects against tumor growth in a murine model of ovarian cancer. PLoS One 9:89350. PMID: 24586709
16. Curti, B.D. et al (2013) OX40 is a potent immune-stimulating target in late-stage cancer patients. Cancer Res. 73:7189. PMID: 24177180.
Emerging Immune Checkpoint Pathways
While traditional immune checkpoint targets such as CTLA-4 and PD-1 have transformed cancer immunotherapy, increasing attention is being directed toward alternative immune regulatory pathways outside of classic T cell checkpoints. Emerging targets such as CD47, leukocyte immunoglobulin-like receptors (LILRBs), and indoleamine 2,3-dioxygenase (IDO) play key roles in promoting immune suppression in the tumor microenvironment and are being investigated as potential drug targets.
The CD47-SIRP alpha Pathway is an Innate Immune Checkpoint
The CD47/SIRP pathway represents an innate immune checkpoint distinct from the T cell-focused checkpoints commonly targeted in cancer immunotherapy.1 CD47, also known as Integrin-associated protein (IAP) or OA3, is an atypical immunoglobulin superfamily member characterized by a single N-terminal IgV-like domain, five transmembrane regions, and a cytoplasmic tail lacking classical signaling motifs.2 Expressed ubiquitously on healthy cells, CD47 functions as a “self” marker, preventing the clearance of normal, autologous cells by phagocytes.3
Phagocytosis inhibition occurs via CD47 binding to SIRPα, an immunoglobulin superfamily member primarily found on myeloid cells like macrophages, dendritic cells, neutrophils, and monocytes.3,4 SIRPα contains an extracellular IgV-like domain that directly interacts with the IgV-like domain in CD47, followed by two IgC-like domains, a transmembrane segment, and a cytoplasmic tail featuring two immunoreceptor tyrosine-based inhibitory motifs (ITIMs).5 The interaction between CD47 and SIRPα triggers phosphorylation of the ITIMs in SIRPα, which recruits the SHP-1 and SHP-2 phosphatases, leading to dephosphorylation of downstream signaling targets to inhibit phagocytic activity.6-8
Besides SIRPα, CD47 also binds with lower affinity to SIRPγ.9 SIRPγ is expressed on human T cells and likely facilitates their transendothelial migration.9 Other proteins that interact with CD47 include the extracellular matrix protein, thrombospondin-1 and multiple integrins (αVβ3, α2bβ3, α2β1, α4β1).10-12 Notably, the thrombospondin-1/CD47 axis exerts immunosuppressive effects such as inhibiting T cell activation, promoting regulatory T cell induction from naïve or memory T cells, and negatively regulating dendritic cell activation and cytokine secretion.13-16
CD47 is frequently overexpressed in various hematologic and solid tumors and is generally associated with a poor prognosis, indicating that cancer cells exploit this pathway to escape immune clearance.13-21 Antibodies blocking CD47 enhance macrophage-mediated tumor phagocytosis in vitro and reduce tumor growth and metastasis across multiple xenograft and syngeneic mouse models.22,23 CD47 blockade also improves antigen uptake and presentation, and dendritic cell- and macrophage-mediated cytokine production, leading to increased activation of tumor-specific cytotoxic T cells.3,23-25 Preclinical studies suggest that blocking CD47 or SIRPα, particularly when combined with immune checkpoint inhibitors targeting PD-1 or CTLA-4, may produce synergistic effects against tumors.23
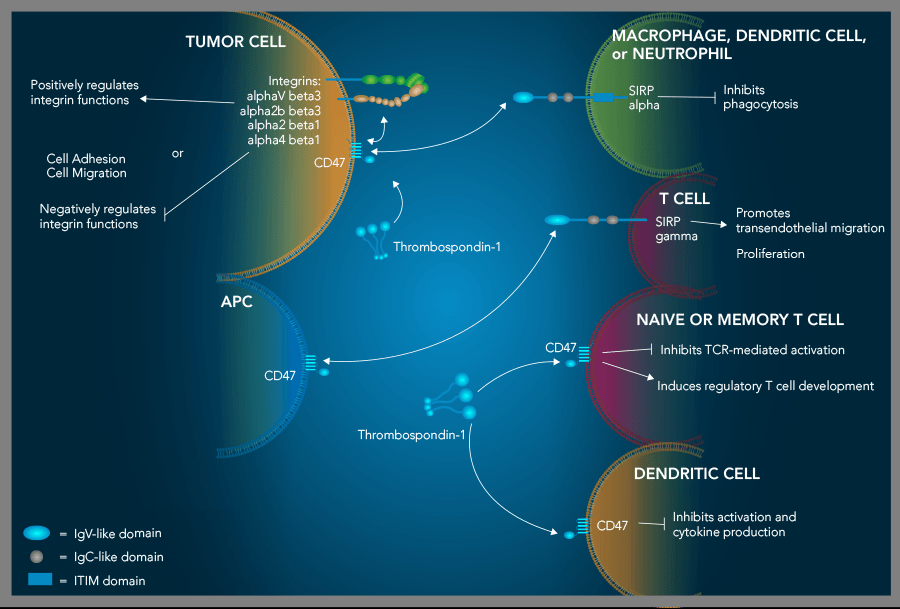
The CD47-SIRP alpha signaling pathway is a myeloid cell-specific phagocytosis checkpoint that can be exploited by tumor cells. CD47 is a ubiquitously expressed protein that serves as a marker of “self”. Binding of CD47 to SIRP alpha on myeloid cells inhibits myeloid cell-mediated phagocytosis. By overexpressing CD47, tumor cells frequently exploit this pathway, allowing them to evade detection and phagocytic destruction. In addition to its interaction with SIRP alpha, CD47 interacts with Thrombospondin-1, and this interaction also has immunosuppressive effects including the inhibition of T cell and dendritic cell activation and the generation of regulatory T cells.
SPR data for CD47:SIRP alpha interaction
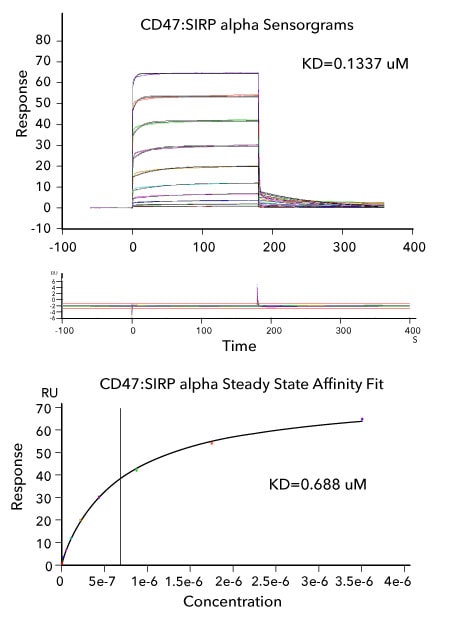
Affinity Measurements and Binding Kinetics of the CD47:SIRP alpha Interaction by Surface Plasmon Resonance. Sensorgram data of captured Avi-tag Biotinylated Recombinant Human CD47 Fc tag (R&D Systems, Catalog # AVI4670)binding to Recombinant Human SIRP alpha His tag (R&D Systems, Catalog # 9378-SA). The corresponding overlaid kinetic fits with the residual plot shown below. The concentration of Recombinant Human SIRP alpha His-tag ranged from 6.85 nM to 3.51 μM. The corresponding steady state affinity fit is shown at the bottom. The experiment was performed on a Biacore T200, GE Healthcare.
References
- Veillette, A. & J. Chen (2018) SIRP-alpha-CD47 immune checkpoint blockade in anticancer therapy. Trends Immunol. 39:173. PMID: 29336991.
- Campbell, I.G. et al. (1992) An ovarian tumor marker with homology to vaccinia virus contains an IgV-like region and multiple transmembrane domains. Cancer Res. 52:5416. PMID: 1394148.
- Matlung, H.L. et al. (2017) The CD47-SIRP-alpha signaling axis as an innate immune checkpoint in cancer. Immunol. Rev. 276:145. PMID: 28258703.
- Adams, S. et al. (1998) Signal-regulatory protein is selectively expressed by myeloid and neuronal cells. J. Immunol. 161:1853. PMID: 9712053.
- van Beek, E.M. et al. (2005) Signal regulatory proteins in the immune system. J. Immunol. 175:7781. PMID: 16339510.
- Barclay, A.N. & T. Van den Berg (2014) The interaction between signal regulatory protein alpha (SIRP-alpha) and CD47: structure, function, and therapeutic target. Annu. Rev. Immunol. 32:25. PMID: 24215318.
- Lienard, H. et al. (1999) Signal regulatory proteins negatively regulate immunoreceptor-dependent cell activation. J. Biol. Chem. 274:10542295.
- Tsai, R.K. et al. (2008) Inhibition of "self" engulfment through deactivation of myosin-II at the phagocytic synapse between human cells. J. Cell. Biol. 180:989. PMID: 18332220.
- Brooke, G. et al. (2004) Human lymphocytes interact directly with CD47 through a novel member of the signal regulatory protein (SIRP) family. J. Immunol. 173:2562. PMID: 15294972.
- Brown, E.J. & W.A. Frazier (2001) Integrin-associated protein (CD47) and its ligands. Trends Cell Biol. 11:130. PMID: 11306274.
- Barazi, H.O. et al. (2002) Regulation of integrin function by CD47 ligands. Differential effects on alpha vbeta3 and alpha 4beta1 integrin-mediated adhesion. J. Biol. Chem. 277:42859. PMID: 12218055.
- Isenberg, J.S. et al. (2008) CD47: a new target in cardiovascular therapy. Arterioscler. Thromb. Vasc. Biol. 28:615. PMID: 18187671.
- Li, Z. et al. (2001) Thrombospondin-1 inhibits TCR-mediated T lymphocyte early activation. J. Immunol. 166:2427. PMID: 11160302.
- Miller, T.W. et al. (2013) Thrombospondin-1 is a CD47-dependent endogenous inhibitor of hydrogen sulfide signaling in T cell activation. Matrix Biol. 32:316. PMID: 23499828.
- Grimbert, P. et al. (2006) Thrombospondin/CD47 interaction: a pathway to generate regulatory T cells from human CD4+CD25- T cells in response to inflammation. J. Immunol. 177:3534. PMID: 16951312.
- Doyen, V. et al. (2003) Thrombospondin 1 is an autocrine negative regulator of human dendritic cell activation. J. Exp. Med. 198:1277. PMID: 14568985.
- Rendtlew, J.M. et al. (2007) Dysregulation of CD47 and the ligands thrombospondin 1 and 2 in multiple myeloma. Br. J. Haematol. 138:756. PMID: 17760807.
- Jaiswal, S. et al. (2009) CD47 is upregulated on circulating hematopoietic stem cells and leukemia cells to avoid phagocytosis. Cell 138:271. PMID: 19632178.
- Edris, B. et al. (2012) Antibody therapy targeting the CD47 protein is effective in a model of aggressive metastatic leiomyosarcoma. Proc. Natl. Acad. Sci. USA 109:6656. PMID: 22451919.
- Willingham, S.B. et al. (2012) The CD47-signal regulatory protein alpha (SIRP-alpha) interaction is a therapeutic target for human solid tumors. Proc. Natl. Acad. Sci. USA 109:6662. PMID: 22451913.
- Liu, X.J. et al. (2017) Is CD47 an innate immune checkpoint for tumor evasion? J. Hematol. Oncol. 10:12. PMID: 28077173.
- Burugu, S. et al. (2017) Emerging targets in cancer immunotherapy. Semin. Cancer Biol. 52:39. PMID: 28987965.
- Murata, Y. et al. (2018) CD47-signal regulatory protein alpha signaling system and its application to cancer immunotherapy. Cancer Sci. 109:2349. PMID: 29873856.
- Tseng, D. et al. (2013) Anti-CD47 antibody-mediated phagocytosis of cancer by macrophages primes an effective antitumor T-cell response. Proc. Natl. Acad. Sci. USA 110:11103. PMID: 23690610.
- Liu, X. et al. (2015) CD47 blockade triggers T cell-mediated destruction of immunogenic tumors. Nat. Med. 21:1209. PMID: 26322579.
Tryptophan Metabolism Can Inhibit Immune Cell Functions
Tryptophan, an essential amino acid required for protein synthesis, is primarily metabolized through the kynurenine pathway, which modulates immune cell functions.1 The enzymes, indoleamine-2,3-dioxygenase (IDO1 and IDO2) and tryptophan-2,3-dioxygenase (TDO2) catalyze the initial, rate-limiting step in the kynurenine pathway by converting L-tryptophan into N-formyl-kynurenine, which is subsequently converted into kynurenine. This process affects immune responses by depleting tryptophan, which inhibits T cell proliferation and activity,2-5 and by producing kynurenine, an immunoregulatory metabolite that inhibits T and natural killer cell functions and proliferation, while promoting regulatory T cell differentiation.6-9
IDO and TDO are expressed across multiple cell types, including stromal, vascular, tumor, and immune cells, most notably on antigen-presenting cells.10 Expression of indoleamine-2,3-dioxygenase is strongly upregulated by inflammatory cytokines such as IFN-gamma, TNF-alpha, and IL-6.10-12 Numerous tumors also express IDO and/or TDO, contributing to immune evasion.10-12 High IDO levels in tumors correlate with increased myeloid-derived suppressor cell (MDSC) recruitment and activation, tumor aggressiveness, and resistance to immune checkpoint therapies.13 Consequently, targeting the kynurenine pathway, particularly through small-molecule IDO inhibitors, is a promising avenue under investigation for cancer immunotherapy.
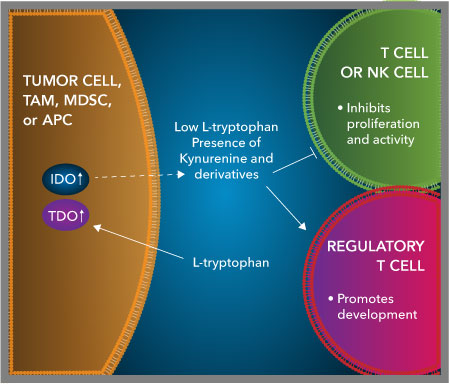
High level expression of indoleamine-2,3-dioxygenase and tryptophan-2,3-dioxygenase by tumor cells or cells in the tumor microenvironment inhibits the functions of T cells and natural killer (NK) cells. Indoleamine-2,3-dioxygenase and tryptophan-2,3-dioxygenase, two enzymes involved in the metabolism of tryptophan, are produced by tumor cells or other cells present in the tumor microenvironment, including tumor-associated macrophages (TAMs), myeloid-derived suppressor cells (MDSCs) and antigen-presenting cells (APCs). High level expression of indoleamine-2,3-dioxygenase and tryptophan-2,3-dioxygenase contribute to a favorable environment for tumor progression as they increase the rate of tryptophan degradation, leading to the depletion of tryptophan from the tumor microenvironment and the production of the immunosuppressive metabolite, kynurenine. Together, these conditions suppress T cell and natural killer (NK) cell proliferation and activity and promote the generation of regulatory T cells.
References
- Erhardt, S. et al. (2009) Pharmacological manipulation of kynurenic acid: potential in the treatment of psychiatric disorders. CNS Drugs 23:91. PMID: 19173370.
- Munn, D.H. et al. (1999) Inhibition of T cell proliferation by macrophage tryptophan catabolism. J. Exp. Med. 189:1363. PMID: 10224276.
- Mellor, A.L. et al. (2002) Cells expression indoleamine 2,3-dioxygenase inhibit T cell responses. J. Immunol. 168:3771. PMID: 11937528.
- Munn, D.H. & A.L. Mellor (2007) Indoleamine 2,3-dioxygenase and tumor-induced tolerance. J. Clin. Invest. 117:1147. PMID: 17476344.
- Lee, G.K. et al. (2002) Tryptophan deprivation sensitizes activated T cells to apoptosis prior to cell division. Immunology 107:452. PMID: 12460190.
- Frumento, G. et al. (2002) Tryptophan-derived catabolites are responsible for inhibition of T and natural killer cell proliferation induced by indoleamine 2,3-dioxygenase. J. Exp. Med. 196:459. PMID: 12186838.
- Terness, P. et al. (2002) Inhibition of allogeneic T cell proliferation by indoleamine 2,3-dioxygenase-expressing dendritic cells: mediation of suppression by tryptophan metabolites. J. Exp. Med. 196:447. PMID: 12186837.
- Della Chiesa, M. et al. (2006) The tryptophan catabolite L-kynurenine inhibits the surface expression of NKp46- and NKG2D-activating receptors and regulates NK-cell function. Blood 108:4118. PMID: 16902152.
- Yan, Y. et al. (2010) IDO upregulates regulatory T cells via tryptophan catabolite and suppresses encephalitogenic T cell responses in experimental autoimmune encephalomyelitis. J. Immunol. 185:5953. PMID: 20944000.
- Ye, Z. et al. (2019) Role of IDO and TDO in cancers and related diseases and the therapeutic implications. J. Cancer 10:2771. PMID: 31258785.
- Marin-Acevedo, J.A. et al. (2018) Next generation of immune checkpoint therapy in cancer: new developments and challenges. J. Hematol. Oncol. 11:39. PMID: 29544515.
- Burugu, S. et al. (2017) Emerging targets in cancer immunotherapy. Semin. Cancer Biol. 52:39. PMID: 28987965.
- Holmgaard, R.B. et al. (2015) Tumor-expressed IDO recruits and activates MDSCs in a Treg-dependent manner. Cell Rep. 13:412. PMID: 26411680.
LILRB1/LILRB2
The leukocyte immunoglobulin-like receptors (LILRs), also referred to as immunoglobulin-like transcripts (ILTs), comprise a family of immunomodulatory receptors expressed across various hematopoietic cells, including dendritic cells, monocytes, macrophages, granulocytes, natural killer (NK) cells, as well as T and B lymphocytes.1 The LILR family can be divided into two subgroups: the activating LILRA receptors (LILRA1-6) and the inhibitory LILRB receptors (LILRB1-5). Activation mediated by LILRA receptors occurs via immunoreceptor tyrosine-based activation motifs (ITAMs), promoting immune cell stimulation, whereas LILRB receptors transmit inhibitory signals through immunoreceptor tyrosine-based inhibitory motifs (ITIMs), dampening immune responses.
Notably, LILRB receptors are unique to humans and primates, with murine counterparts limited to paired immunoglobulin-like receptor B (PirB) and gp49B1.2 Ligand interactions for human LILRB1/ILT2 and LILRB2/ILT4 include both classical (HLA-A, -B, -C) and non-classical (HLA-E, -F, -G) MHC class I molecules. Other ligands reported include S100A8/A9 and the viral MHC mimic UL18 for LILRB1/ILT2, and angiopoietin-like proteins (ANGPTLs), CD1d, myelin-associated glycoprotein (MAG), oligodendrocyte myelin glycoprotein (OMgp), oligomeric beta-amyloid (Aβ), and reticulon 4 (RTN4/NOGO) for LILRB2/ILT4.2 Unlike LILRB2, LILRB1 binding to HLA ligands is dependent on β2-microglobulin.3,4 Furthermore, LILRB1/ILT2 exhibits higher affinity for HLA-G compared to classical HLA class I molecules, whereas LILRB2/ILT4 preferentially binds ANGPTL proteins over HLA-G.2-4 ANGPTLs have also been identified as ligands for LILRB4 and LILRB5, and classical HLA class I molecules are reported to interact with LILRB5; however, ligands for LILRB3 remain unknown.2,5
Given their inhibitory functions, LILRB receptors are classified as immune checkpoint molecules. Ligands such as HLA-G that engage LILRB1 are frequently overexpressed on tumor cells, contributing to immune evasion by suppressing the activity of NK cells, dendritic cells, monocytes/macrophages, T cells, and B cells.1,6,7 Moreover, LILRBs are upregulated not only on various cancer cells but also on immune populations within the tumor microenvironment, including tumor-associated macrophages (TAMs) and myeloid-derived suppressor cells (MDSCs), where they facilitate tumor progression, angiogenesis, and metastasis.7 In preclinical models, blockade of LILRB2 in Lewis lung carcinoma resulted in enhanced anti-tumor immunity, decreased frequencies of granulocytic MDSCs and regulatory T cells, and suppressed tumor growth both as a monotherapy and synergistically when combined with anti-PD-L1 antibodies.8 Collectively, these findings support the potential of LILRB receptors as promising targets for cancer immunotherapy.
Leukocyte immunoglobulin-like receptors mediate immune cell stimulatory and immune cell inhibitory effects. Members of the leukocyte immunoglobulin-like receptor family belong to the LILRA or LILRB subfamilies, which have immune cell stimulatory or inhibitory effects, respectively. LILRA and LILRB subfamilies bind to a range of different ligands including MHC class I molecules, angiopoietin (ANGPT) and angiopoietin-like (ANGPTL) proteins, and other proteins as shown in the graphic and described in the text. Interactions between the LILR family proteins and their ligands either promote (LILRA; shown on the left) or inhibit (LILRB; shown on the right) immune cell activation. LILRBs in particular are being investigated as potential therapeutic targets due to their immunosuppressive effects.
Human Angiopoietin-like 7 (ANGPTL7) Binds to LILRB2/ILT4. Recombinant Human LILRB2/ILT4 (R&D Systems, Catalog # 8429-T4) was coated on a plate at 2 μg/mL and the indicated concentrations of Recombinant Human Angiopoietin-like 7 (R&D Systems, Catalog # 914-AN) were added. Recombinant Human ANGPTL7 bound with an ED50 of 25-150 ng/mL.
References
- van der Touw, W. et al. (2017) LILRB receptor-mediated regulation of myeloid cell maturation and function. Cancer Immunol. Immunother. 66:1079. PMID: 28638976.
- Kang, X. et al. (2016) Inhibitory leukocyte immunoglobulin-like receptors: Immune checkpoint proteins and tumor sustaining factors. Cell Cycle 15:25. PMID: 26636629.
- Allen, R.L. et al. (2001) Leukocyte receptor complex-encoded immunomodulatory receptors show differing specificity for alternative HLA-B27 structures. J. Immunol. 167:5543. PMID: 11698424.
- Willcox, B.E. et al. (2003) Crystal structure of HLA-A2 bound to LIR-1, a host and viral major histocompatibility complex receptor. Nat. Immunol. 4:913. PMID: 12897781.
- Zhang, Z. et al. (2015) The leukocyte immunoglobulin-like receptor family member LILRB5 binds to HLA-class I heavy chains. PLoS One 10:129063. PMID: 26098415.
- Barkal, A.A. et al. (2017) Engagement of MHC class I by the inhibitory receptor LILRB1 suppresses macrophages and is a target of cancer immunotherapy. Nat. Immunol. 19:76. PMID: 29180808.
- Zhao, J. et al. (2019) The MHC class I-LILRB1 signalling axis as a promising target in cancer therapy. Scand. J. Immunol. 90:12804. PMID: 31267559.
- Chen, H-M. et al. (2018) Blocking immunoinhibitory receptor LILRB2 reprograms tumor-associated myeloid cells and promotes antitumor immunity. J. Clin. Invest. 128:5647. PMID: 30352428.
Tailor-Made Reagents
From flexible sizes, formats, and formulations to fully customized products, we specialize in meeting your unique needs.
Immune Checkpoint Targets eBook
Explore comprehensive insights into the immune checkpoint targets shaping the future of cancer immunotherapy. Download our recent eBook to learn more about the roles these molecules play in regulating immune responses.
A Look Inside a Tumor Wall Poster
The tumor microenvironment (TME) plays a central role in inhibiting anti-tumor immune responses. Request this poster to learn about the key mechanisms by which Tregs, MDSCs, TAMs, and tumor-derived exosomes promote immunosuppression in the tumor microenvironment.
Immune Checkpoint Targets Wall Poster
Decorate your lab with our recent poster showing the immune checkpoint ligand-receptor interactions being investigated as potential targets for cancer immunotherapy. It serves as a great reference tool and is sure to brighten up your lab.
